Invertebrate Anatomy OnLine
Saccoglossus kowalevskii ©
Acorn Worm
5jul2006
Copyright 2001 by
Richard Fox
Lander University
and
Edward Ruppert
ClemsonUniversity
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises, a glossary, and chapters on supplies and laboratory techniques are also available at this site. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Hemichordata P, Enteropneusta C, Harrimaniidae F (Fig 9-26, 27-12)
Hemichordata P
Hemichordata consists of Pterobranchia and the Enteropneusta, the acorn worms and sea angels, respectively. About 90 species are known and all are marine and benthic.
Hemichordates have the three body divisions and three-part coelom characteristic of the ancestral deuterostomes. The protosome at the anterior end contains the protocoel, the middle mesosome contains the mesocoel, and the posterior metasome has the metacoel. The heart/kidney complex is a combination excretory and nutrient delivery system consisting of a pressurized hemal system and metanephridium. Pharyngeal gill slits are present and a median dorsal diverticulum of the gut, suggestive of the chordate notochord and known as the stomochord, supports the heart/kidney.
Hemichordates are of considerable phylogenetic interest because of their evolutionary connections with the ancestral deuterostomes, echinoderms, chordates, and perhaps lophophorates.
Enteropneusta C
Seventy hemichordate species are enteropneusts, or acorn worms (Fig 27-1). These are large worms with elongate, bodies that inhabit soft marine sediments where they may be deposit or suspension feeders. Numerous pharyngeal gill slits are present in the anterior gut.
The body is divided into the three regions thought to have been present in the ancestral deuterostomes but in enteropneusts they have specialized names. The protosome is known as the proboscis, the mesosome is the collar, and the metasome is the trunk. The mouth is located between the proboscis and collar and the trunk is at the extreme posterior end of the trunk. Enteropneusts are gonochoric with external fertilization, radial cleavage, and enterocoely.
Laboratory Specimens
Saccoglossus kowalevskii (= Dolichoglossus) is a small acorn worm common in shallow, intertidal mudflats on the coasts of the eastern United States and Europe (Fig 27-1B,F). It constructs and inhabits helical burrows on silty-sand, low-energy, intertidal flats. Saccoglossus feeds on particulate organic matter at the sand surface which it collects using its elongate ciliated proboscis.
These instructions were written for Saccoglossus but can be used, with a little modification, for Balanoglossus (Ptychoderidae) and other enteropneusts. Parenthetical comments refer to Balanoglossus. Dissection of living specimens is preferable to preserved and the color descriptions used here refer to living specimens.
External Anatomy
Obtain a living specimen in a 10-cm glass culture dish of seawater. Study the worm with the dissecting microscope.
Body Regions
The wormlike body is divided into a long, pointed, anterior proboscis (the proboscis is short, blunt, and acorn-shaped in Balanoglossus, Fig 27-1A), a short collar, and a long posterior trunk (Fig 1, 27-1B). The proboscis is connected to the collar by a slender, dorsal, reinforced proboscis stalk. Saccoglossus reaches 15-20 cm in length and most of this length is trunk. The trunk is fragile and, in the laboratory, usually is not present in its entirety, the posterior end having been lost during collecting. The body is permanently coiled into a helix and inhabits a helical burrow (Fig 27-1F).
Epidermis
The entire animal is covered with a multiciliated, glandular, sensory, monolayered, columnar epithelium entirely lacking a cuticle or exoskeleton (Fig 27-2). The integument is fragile and, unlike that of other animals, offers little mechanical protection against infection or damage. The epidermis secretes mucus and 2, 5-dibromophenol, a bromine compound that smells like iodine and is thought to be an antibiotic to protect the soft cuticle-free body from infection. An alternative hypothesis is that the phenol prevents bacterial growth on the walls of the burrow thus reducing competition for oxygen. An iodine-like odor is characteristic of acorn worms and is easily detected when digging or handling these worms.
>1a. Sniff the dish, or your hands if you have handled the worm, and see if you can detect a medicinal odor similar to that of iodine. If you handle the worm your hands will retain this odor for several hours but is not unpleasant. <
Figure 1. View of the left side of Saccoglossus kowalevskii. Hemi28La.gif
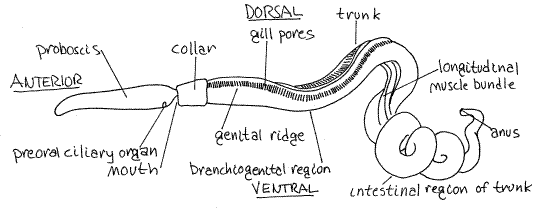
Proboscis
Relocate the anterior, pale cream, white, yellowish or pink proboscis. It is very mobile and contains abundant muscles. Watch for retrograde waves of peristalsis passing along it from anterior to posterior generated by the abundant proboscis musculature. The proboscis is the burrowing and feeding organ (Fig 27-4A-D).
>1b. Place a healthy worm on the surface of silty sand in a large dish of seawater. Observe the activity of the proboscis, watching for waves of contraction. Do they begin anteriorly or posteriorly? <
Place your specimen in a small, wax-bottom dissecting pan of isotonic magnesium chloride (or tapwater if preserved) and allow a few minutes for the animal to relax. Place the pan on the stage of the dissecting microscope.
The proboscis is homologous to the protosome of the ancestral deuterostome. It is the anteriormost body region and its coelomic space is the unpaired protocoel. Within the proboscis is the heart-kidney complex and elements of the hemal and nervous systems.
The proboscis epithelium is ciliated and secretes mucus and amylase. On the posterior wall of the proboscis, facing the collar, is a large U-shaped patch of ciliated secretory epithelium known as the preoral ciliary organ (Fig 1, 27-4E). The protocoel opens to the exterior by a small obscure proboscis pore located on the left side of the proboscis stalk near the collar (Fig 27-3A). The pore may be difficult to find. Look for it at the base of the stalk just before the stalk joins the collar. You must deflect the lip of the collar and gently probe the surface of the stalk and open the pore, before you will see it. The protocoel is connected to the proboscis pore by a proboscis pore canal that passes through the muscle of the body wall.
Proboscis Stalk
The slender proboscis stalk connects the proboscis with the collar and is reinforced by an internal proboscis skeleton (Fig 1, 27-4E). Push gently at the stalk with your nadel to demonstrate its relatively rigid consistency.
Collar
The collar is orange and short, about as wide as long (Fig 1, 27-4E). The very large, open mouth is on the anterior wall of the collar below the stalk and opposite the preoral ciliary organ. The mouth occupies almost the entire anterior wall of the collar.
It opens into the buccal cavity in the interior of the collar (Fig 27-3B).
The collar is homologous to the mesosome of the ancestral coelomate and contains paired mesocoels (Fig 27-3A). They, like the protocoel, connect to the exterior by ducts and pores but these are not visible in gross examination.
>1c. If your specimen is alive, pipet a drop of carmine/seawater suspension, fine chalk dust, or colloidal carbon (India ink in seawater) over the surface of the proboscis and collar. Watch the particles with magnification and describe their motion. Proboscis cilia move particles posteriorly and the stalk cilia sweep them ventrally into the mouth (Fig 27-4E). <
Trunk
Posterior to the collar is the long pink, brownish, or orange trunk (Fig 1, 27-1B). It is homologous to the metasome of the early deuterostomes and contains the paired metacoelomic compartments. It is long and accounts for most of the length of the worm. In Saccoglossus it is helical but that is not the case with most acorn worms.
In most species, the trunk is divisible into three major regions. In order, from anterior to posterior, are the branchiogenital region, the hepatic region, and the intestinal region (Fig 27-1A) but Saccoglossus has no externally recognizable hepatic region.
The branchiogenital region bears the gill pores and gonads (Fig 1). The approximately 80 pairs of external, dorsolateral gill pores are easily observed with a dissecting microscope. The number varies with species and increases with age (and size). The gill pores are the external openings of the internal gill slits.
The numerous, paired, pouchlike gonads are visible externally forming two long rounded genital ridges on the dorsolateral body wall. (They are flat genital wings in Balanoglossus.) The extent of the genital ridges roughly coincides with the region of the gill pores. At maturity, each gonad opens to the exterior via its own tiny dorsolateral gonopore (Fig 27-3A). These pores develop with the gonads and are not always present.
The middle of the trunk is called the hepatic region because the gut epithelium here is thought to function in intracellular digestion as does that of the liverlike digestive ceca of other animals. In Balanoglossus and Ptychodera, but not Saccoglossus, this epithelium forms gut diverticula which protrude from the dorsolateral surface of the trunk as conspicuous papillae (Fig 27-1A, 27-3B). These hepatic ceca, as they are known, are easily seen in complete specimens of Balanoglossus where they cover the dorsolateral aspect of the hepatic region of the trunk. Unfortunately, the hepatic and intestinal regions are often missing due to the tendency of the trunk to break. In Saccoglossus, the gut epithelium of the hepatic region does not form diverticula and gives no external indication of its existence.
The midregion of the trunk of Saccoglossus bears two conspicuous bundles of longitudinal muscles lying side by side along the ventral midline (Fig 1). These are the muscles that hold the animal in its characteristic helical shape.
The intestinal region of the trunk is fragile and thin-walled but is often missing in laboratory specimens as it is frequently lost when the worms are dug from the mud flat. It contains the intestine which ends with the terminal anus at the posterior end of the body. There is no postanal tail in adult enteropneusts although there is in larval Saccoglossus and adult pterobranchs. A postanal tail is another chordate apomorphy.
Internal Anatomy
The absence of cuticle and the paucity of connective tissue in the body wall make it easy to open the body cavities but increase the likelihood of doing damage in the process. Most of the dissection can be accomplished with a 000 insect pin but you will need a sharp scalpel or razor blade to make cross sections.
" Begin by cutting the animal into anterior and posterior sections. Use a sharp scalpel or razor blade to make a smooth transverse section across the branchiogenital region about 1 cm posterior to the collar. Set the posterior end aside for the time being and devote your attention to the anterior portion.
" Orient the proboscis with its ventral side up in the center of a small dissecting pan. Use the stalk and gill pores, both of which are dorsal, as landmarks. Anchor the tip of the proboscis near the middle of the pan with a 000 stainless steel insect pin inserted at a 45 ° angle. Arrange the body so it extends straight back from the proboscis and anchor it with two pins in its right and left sides. Do not pull on the pinned proboscis or the fragile tissues will tear.
Beginning at the anterior edge of the collar (at the mouth), use a scraping motion of the point of the insect pin to make a longitudinal midventral incision posteriorly through the collar to the truncated posterior end of the worm fragment. In this manner cut through the body wall into the lumen of the gut. Deflect (carefully) and pin the cut edges of the body but do not try to stretch the tissue as you pin it. This incision opens the gut.
Open the proboscis using the same technique beginning about 1 cm anterior to the stalk by making a longitudinal midventral incision with a pin. Cut posteriorly toward the collar and stop at the proboscis stalk. This incision opens the proboscis coelom (protocoel). Deflect the cut sides of the proboscis, pinning them as necessary (Fig 2).
Enteropneust muscles are myoepithelial cells originating from the mesothelium. They occupy most of the space between the epidermis and the gut epithelium, filling the coelom. The muscles of the proboscis are responsible for the extension, contraction, and peristalsis exhibited by that region.
Proboscis
The space inside the proboscis is the protocoel, or proboscis coelom, but it is largely occupied by muscle fibers which give it a spongy appearance (Fig 2, 27-5A). Although enteropneust coeloms are visible in histological sections, they are often difficult to demonstrate in gross dissection because of the tendency of the mesothelium to differentiate into muscles and other tissues which then occupy most of the cavity.
Figure 2. The anterior end of Saccoglossus opened by a midventral incision to reveal the interior of the protocoel and the anterior gut of the collar and anterior trunk. The dotted line indicates the position of the proboscis skeleton. Hemi29La.gif
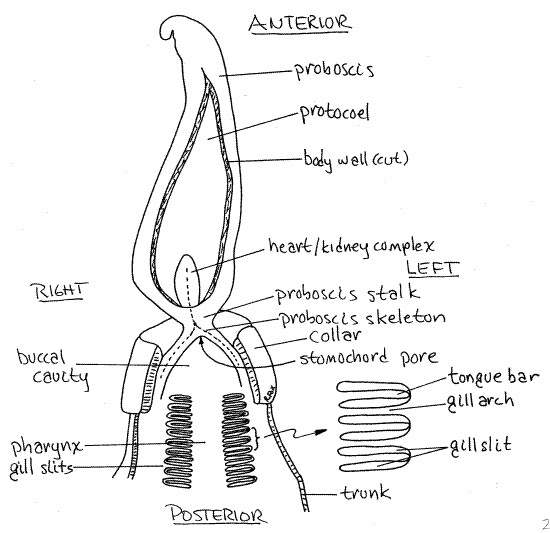
The heart-kidney complex is at the posterior end of the protocoel (Fig 2, 27-3A). This rather large, white or yellowish (in life), heart-shaped organ is composed of a ventral glomerulus, a central heart and its surrounding pericardium with podocytes, the anterior tip of the stomochord, and the anterior end of the proboscis skeleton (Fig 27-5A). The glomerulus is the only part you can see.
The hemal system and excretory system function together as a nutrient delivery system for the tissues of the proboscis. Contraction of the heart forces an ultrafiltrate of the blood across the walls of the glomerulus into the protocoel to form the primary urine. Nutrients, such as sugars and amino acids, in the primary urine are absorbed by the proboscis muscles and other tissues. Following this absorption the ultrafiltrate has become the final urine which is then excreted through the proboscis pore.
Collar and Trunk
Immediately posterior to the heart/kidney complex, notice the transparent, Y-shaped proboscis skeleton (Fig 2) embedded in the dorsal wall of the collar (it is dark gray in Balanoglossus). Because part of it is obscured, it will appear to you as a "V" rather than a "Y". The exposed, open arms of the "Y" point posteriorly and form ridges bordering the mouth, whereas its stem extends anteriorly into the stalk and heart-kidney and is hidden from view.
Gently push one arm of the proboscis skeleton with the tip of your nadel to test its strength and consistency. It is composed, in part, of a cartilage-like connective tissue known as chondroid. The skeleton continues anteriorly through the stalk into the heart-kidney complex as a single rod, which you can feel by pushing the stalk with your nadel.
The proboscis skeleton attaches the proboscis to the collar and supports the heart-kidney complex. Muscles attached to the arms of the Y pull the proboscis against the collar when they contract.
Digestive and Respiratory Systems
The digestive system consists of mouth, buccal cavity, pharynx with gill pores, esophagus, intestine divided into hepatic and posthepatic regions, and anus. The mouth and buccal cavity are in the collar, whereas the remaining regions are in the trunk.
Buccal Cavity
The mouth opens into the short buccal cavity in the interior of the collar (Fig 2). The stomochord is a dorsal evagination of the anterior end of the buccal cavity, which extends through the stalk into the proboscis.
A tiny stomochord pore is in the anterior dorsal wall of the buccal cavity just posterior to the intersection of the two arms of the proboscis skeleton (Fig 2). Although usually not discernible in gross dissection, it is the point where the gut lumen evaginates into the hollow stomochord (Fig 27-5A). The stomochord has been considered by some zoologists to be homologous to the chordate notochord.
Pharynx
Posterior to the buccal cavity the gut enters the trunk and becomes the pharynx which is easily recognized by the presence of conspicuous U-shaped pharyngeal gill slits in its walls (Fig 2). The gill slits are long narrow, parallel openings occurring in two rows dorsolaterally on the pharyngeal walls.
Each slit opens into its own pouch, or atrium, which itself opens to the exterior via a gill pore on the surface of the animal (Fig 3). The gill pores are visible externally (Fig 1). Adjacent gill slits are separated from each other by gill arches (interbranchial septa). The ciliated epithelium of the gill slit generates the water current that enters the mouth and exits through the gill slits, atria, and gill pores.
Look closely at a gill slit with moderate magnification (25-30X) and open it gently with a nadel. The gill slits are U-shaped. The center of the "U" is occupied by a vertical tongue bar that descends ventrally from the dorsal edge of the slit to the ventral edge (Fig 2 inset). From this viewpoint, tongue bars look very much like the gill arches that separate adjacent slits. Unlike the arches, tongue bars do not connect with the ventral border of the slit although they appear to do so. Gill arches are columns of tissue that are attached at both ends whereas tongue bars are only attached dorsally, although they appear to be attached at both ends. Gill arches are partitions that separate adjacent gill slits from each other. Tongue bars, on the other hand, simply subdivide a gill slit into anterior and posterior halves. The tongue bar provides surface area for cilia and contains most of the blood supply to the gill, including a branchial plexus of capillaries.
Figure 3. A cross section through the branchiogenital region of the trunk of Saccoglossus. The section is slightly oblique so that on the left it passes through the middle of a gill slit and the tongue bar whereas on the right it passes through the gill arch (septum) between two gill slits. Hemi30La.gif
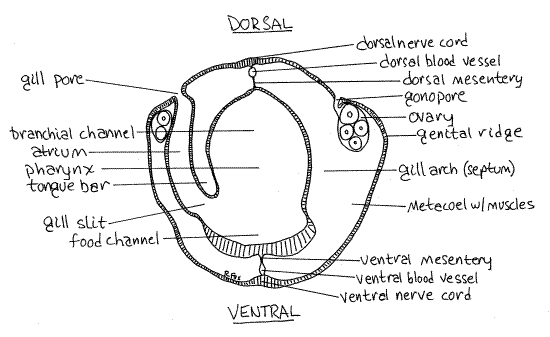
Push gently at the tongue bar with your nadel to demonstrate that it is attached dorsally but not ventrally. Push on the gill arch to demonstrate that it is attached at both ends.
With a nadel lift the tongue bar out of the slit and push it toward the middle of the pharynx to reveal the atrium dorsal and lateral to the slit. Each slit opens laterally into a water-filled internal pouch, which is the atrium (Fig 3). The atria in turn open to the exterior via the gill pores which you saw earlier on the dorsal surface of the trunk. Note that the outer openings of the gills, on the surface of the animal, are called gill pores whereas the inner openings, in the gut wall, are the gill slits.
This arrangement of gill slits, atrium, and gill pores is similar to that found in cephalochordates and urochordates in which the gill slits open from the pharynx into an internal water space, the atrium, and then to the exterior. In vertebrates a similar situation is seen in larval anuran amphibians (tadpoles) in which the gills open into a surrounding water jacket which then opens to the exterior via a single opening.
In all these animals the water space between the gill slits and gill pores is an epidermally-lined invagination of the body surface. In enteropneusts each gill has its own atrium whereas in the cephalochordates and urochordates they share a common atrium.
" Retrieve the posterior portion of the worm which you set aside earlier.
Make a cross section using a sharp scalpel, about 2 mm from the anterior (cut) end. The result is a thin slice of the branchiogenital region which will give you a different view of the pharynx and its gills (Fig 3). Observe the cut surfaces of the slice with high power of the dissecting microscope and look for the features of the gills described above. Look for the pharynx lumen, gill slits and gill arches, tongue bars, atria, and gill pore. Be sure you understand the physical relationships between these structures and how they function.
Notice that in cross section the pharynx is incompletely divided into two channels. Dorsally there is a large branchial channel (Fig 3, 25-5B) associated with the gills and through which water flows. Ventral to it is the smaller food channel, which transports food posteriorly through the pharyngeal region to the esophagus and intestine. The division between digestive and branchial regions is indistinct in Saccoglossus but in Balanoglossus they are separated by high lateral ridges. Save the cross section for later use.
The primary function of the gill slits of enteropneusts is to allow the water from the feeding current to escape from the gut lumen, thus concentrating the food. In addition they have a respiratory function and apparently also serve as filters to retain fine particles as water exits the pharynx.
" Orient the severed trunk portion of the animal on the wax with its ventral surface down. Pin it in place and open it with a middorsal incision, made with an insect pin, similar to the midventral incision made earlier on the anterior end of the animal. Begin at the truncated (anterior) end of the trunk and cut posteriorly into the abdominal region. Use pins to hold the incision open.
Esophagus
Immediately posterior to the pharynx the gut is the esophagus. Anteriorly its walls are thin and its lumen spacious. This is followed by a region with thick folded walls and restricted lumen and then by a third region in which the walls are thin and the lumen expanded.
Saccoglossus kowalevskii has about five pairs of simple, unciliated pores leading from the lumen of the esophagus to the exterior. They are presumably serially homologous to the pharyngeal gill slits and perhaps serve to eliminate whatever water remains in the food mass. Posterior to the pharynx the gut has no respiratory function and it is likely to be filled with concentrated particulate material from which much of the water has been removed.
Intestine
The intestine extends from the posterior end of the esophagus to the anus. The dorsal epithelium of the anterior end of the intestine is modified to form liver-like hepatic tissue and is usually dark brown or greenish. This specialized epithelium is involved in storing food molecules following intracellular digestion. (In some species, such as Balanoglossus, but not Saccoglossus, the gut wall forms dorsal diverticula, the hepatic ceca, which appear at the body surface as papillae and define the hepatic region of the trunk (Fig 27-3B). These ceca are lined by an epithelium similar to the hepatic tissue of Saccoglossus and with the same function. In Saccoglossus there are no such diverticula or papillae and the hepatic region usually cannot be recognized externally.) It is not necessary to open the abdominal region all the way to the anus.
Reproductive System
Enteropneusts are gonochoric and have numerous pairs of saclike gonads in low dorso-lateral genital ridges on the branchiogenital region of the anterior trunk. The genital ridges are composed of numerous separate pouchlike gonads (Fig 3, 27-3A). Each gonad is a sac which bulges into the trunk coelom and opens to the exterior by a tiny dorsal pore that usually cannot be seen. In life, testes are pale orange whereas ovaries are gray or lavender. The genital ridges may be swollen in your specimen if it is reproductively active.
>1d. If the ridges are swollen, open one of them and look for gametes. The eggs of Saccoglossus are large and yolky. (Those of Balanoglossus are small, with little yolk.) Eggs are easily seen in damaged or dissected genital ridges if your animal is a female but individual sperm cannot be seen with a dissecting microscope. Make a wet mount of the gonad contents and examine it with a compound microscope. Sperm are tiny moving flagellated cells, eggs are large spheres. <
Development is nearly direct in Saccoglossus and other Harrimaniidae and includes a short-lived lecithotrophic larva (Fig 27-8A-D) whereas Balanoglossus and its relatives have a long-lived planktotrophic tornaria larva (Fig 28-E-F).
Find gonads in the cross section you made earlier.
Hemal System
The hemal system is well developed and includes contractile dorsal and ventral longitudinal vessels and with various connecting vessels, a pericardium partially surrounding a heart, a glomerulus with podocytes, and a rich branchial blood supply to the pharyngeal gills (Fig 27-5A). Very little of this is demonstrable in gross dissection. The dorsal and ventral blood vessels can sometimes be seen in the cross section of the pharynx (Fig 3). They are in the dorsal and ventral mesenteries above and below the gut respectively. The blood is colorless and essentially acellular.
The heart is located in the protocoel on the dorsal surface of the stomochord (Fig 27-5A). The heart itself is not contractile. The entire heart complex and stomochord are covered with the mesothelium of the protocoel. The dorsal surface of the heart is surrounded by a contractile muscular fold of this mesothelium known as the pericardium.
The dorsal blood vessel carries blood anteriorly, to the heart, whereas blood in the ventral blood vessel flows posteriorly away from the heart (Fig 27-5A). The ventral vessel exits the heart as two vessels which encircle the mouth and then coalesce to form the single ventral blood vessel in which blood flows to the body. The dorsal and ventral blood vessels connect to numerous minor vessels leading to and from sinuses in the tissues.
Excretory System
The excretory system is a metanephridial kidney intimately associated, both functionally and spatially, with the heart and pericardium. The anterior end of the heart is folded to form numerous diverticula collectively known as the glomerulus. The peritoneum of the glomerulus is equipped with podocytes. Blood flowing anteriorly in the dorsal blood vessel enters the posterior end of the heart. Contractions of the pericardium push blood against the inner wall of the glomerulus and force serum across the pericardium of the glomerulus to form an ultrafiltrate (primary urine) in the lumen of the protocoel.
Cells in and surrounding the protocoel may modify the ultrafiltrate by removing such materials as glucose and amino acids for their own nourishment and prevent their being lost through the proboscis pore. You recall that the protocoel connects to the exterior via a proboscis duct, which is a metanephridium, and pore. An important, perhaps the original, function of the kidney is to provide tissues with nutrients.
The heart-kidney complex is similar to the nephrons of your own kidney, which are also metanephridia. Recall what you know of the vertebrate nephron. The hemichordate glomerulus is comparable to the vertebrate glomerulus, the protocoel is comparable to Bowman's capsule (which is also a coelomic space), and the proboscis pore canal is the metanephridial tubule. In addition, the hemichordate heart-kidney complex and protocoel pore are homologous to the axial gland, axial sinus, and madreporite of echinoderms.
Nervous System
The nervous system cannot be studied in gross dissection. It includes dorsal and ventral longitudinal cords, two nerve rings, and a nerve plexus at the base of the epidermis. Most of it is solid and intraepidermal (Fig 3, 27-5A). No brain-like ganglionic mass is present in hemichordates.
A short region in the collar, known as the collar cord, is deeper (subepidermal) and is more or less hollow, at least in some species, and it is dorsal. The similarity between the collar cord and the dorsal hollow nerve cord of chordates is often cited, along with the pharyngeal gill slits and stomochord, as evidence of close phylogenetic relationships between hemichordates and chordates.
The dorsal and ventral nerve cords may be visible in your cross section (Fig 3). Both are thickened areas of epidermis lying on their respective midlines.
Sensory System
The preoral ciliary organ may be chemosensory but there is as yet no evidence supporting that hypothesis. Isolated photoreceptive cells are scattered through the epidermis. Light striking them elicits burrowing behavior in some species and increased activity in others.
>1e. Place a dark-adapted specimen on the surface of fine sediment in a large finger bowl of seawater and test the effects of a focused beam of light on the animal. Look for changes in behavior as a result of the light. See if it makes a difference if the light is directed onto the proboscis or trunk. <
Feeding Behavior
Saccoglossus is a surface deposit feeder that uses its ciliated proboscis to gather flocculent organic material and a certain amount of fine inorganic particles from the surface of the sediment surrounding the opening of its U-shaped burrow. The glandular epithelium of the proboscis secretes mucus to which these materials adhere. Cilia on the proboscis move the mucus and its trapped particles posteriorly, over the preoral ciliary organ, and into the gaping mouth. Undesirable particles can be rejected by drawing the ventral edge of the collar over the mouth so the particles pass posteriorly beyond the mouth (Fig 27-4E). At intervals Saccoglossus backs up so the anus emerges from the posterior end of the burrow to deposit a coil of sand and undigested material known as a fecal cast. Those of Saccoglossus are tiny but larger acorn worms make fecal casts that are large and characteristic features of intertidal, silty sand, tidal flats (Fig 24-4F).
The gut wall almost entirely lacks musculature but its epithelium is heavily ciliated and food is transported by ciliary currents. The mouth is followed by the buccal cavity, pharynx, esophagus, intestine, and anus, in that order. Digestion takes place in the intestine, posterior to the pharyngeal region. The epithelium of this region is secretory and absorptive. Weak peristaltic waves move food in the region immediately posterior to the pharynx.
Food and mineral particles, entrapped in mucus, are moved posteriorly through the pharynx in the ventral food channel. Once beyond the pharynx and into the hepatic region, food particles are phagocytized by liver cells and digested intracellularly. There is no evidence of extracellular digestion in enteropneusts. Mineral particles continue their posterior progress and are eventually incorporated in fecal casts deposited at the sediment surface and resembling diminutive coils of rope.
References
Balser EJ . 1987. Structural and functional analysis of the proboscis complex of Saccoglossus kowalevskii (Enteropneusta). MS thesis, Clemson Univ. Clemson, South Carolina. 71p.
Balser EJ, Ruppert EE . 1990. Structure, ultrastructure and function of the heart-kidney complex of Saccoglossus kowalevskii (Hemichordata: Enteropneusta). Acta Zool. 71:235-249.
Barrington EJW . 1965. The Biology of Hemichordata and Protochordata. Oliver and Boyd, Edinburgh. 176p.
Barrington EJW, Jefferies RPS . 1975. Protochordates. Academic Press, New York. 361p. (Symposia of the Zoological Society of London 36)
Benito J, Pardos F. 1997. Hemichordata, pp15-101 in Harrison FW, Ruppert EE (eds), Microscopic Anatomy of Invertebrates, vol 15, Hemichordata, Chaetognatha, and the Invertebrate Chordates. Wiley-Liss, New York. 537 pp.
Bullough WS . 1958. Practical Invertebrate Anatomy (2 nd ed). MacMillan, London. 483p.
Hyman LH . 1959. The Invertebrates: Smaller Coelomate Groups, vol. V. McGraw-Hill, New York, 783p.
Kleinholz LH. 1950. Saccoglossus kowalevskii, pp 547-549 in Brown FA (ed). Selected invertebrate types. Wiley, New York. 597 pp.
Ruppert EE, Fox RS. 1988. Seashore animals of the southeast. Univ. South Carolina Press, Columbia, 429 .
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Supplies
Dissecting microscope
Isotonic magnesium chloride
10-cm glass culture dish
Seawater
Small dissecting pan made from an anchovy or sardine can
Living or preserved acorn worm
#000 stainless steel insect pins
Dissecting set with microdissecting tools
Slides and coverslips
Compound microscope
Carmine-seawater suspension, or similar suspended particles