Invertebrate Anatomy OnLine
Katharina tunicata ©
Katy Chiton
24may2007
Copyright 2003 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises can be accessed by clicking on the links on the left. A glossary and chapters on supplies and laboratory techniques are also. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Mollusca P, Eumollusca, Polyplacophora C, Neoloricata O, Ischnochitonina sO, Mopaliidae F (Fig 12-125)
Mollusca P
Mollusca, the second largest metazoan taxon, consists of Aplacophora, Polyplacophora, Monoplacophora, Gastropoda, Cephalopoda, Bivalvia, and Scaphopoda. The typical mollusc has a calcareous shell, muscular foot, head with mouth and sense organs, and a visceral mass containing most of the gut, the heart, gonads, and kidney. Dorsally the body wall is the mantle and a fold of this body wall forms and encloses that all important molluscan chamber, the mantle cavity. The mantle cavity is filled with water or air and in it are located the gill(s), anus, nephridiopore(s) and gonopore(s). The coelom is reduced to small spaces including the pericardial cavity containing the heart and the gonocoel containing the gonad.
The well-developed hemal system consists of the heart and vessels leading to a spacious hemocoel in which most of the viscera are located. The kidneys are large metanephridia. The central nervous system is cephalized and tetraneurous. There is a tendency to concentrate ganglia in the circumenteric nerve ring from which arise four major longitudinal nerve cords.
Molluscs may be either gonochoric or hermaphroditic. Spiral cleavage produces a veliger larva in many taxa unless it is suppressed in favor of direct development or another larva. Molluscs arose in the sea and most remain there but molluscs have also colonized freshwater and terrestrial habitats.
Eumollusca
Eumollusca is the sister taxon of Aplacophora and includes all molluscs other than aplacophorans. The eumolluscan gut has digestive ceca which are lacking in aplacophorans, the gut is coiled, and a complex radular musculature is present.
Polyplacophora C
Polyplacophora (=bearer of many shells) includes 800 species of chitons, or coat-of-mail molluscs. Chitons have elongate, bilaterally symmetrical, dorsoventrally flattened bodies with an oval outline. The shell covers most of the dorsal surface and consists of eight overlapping calcareous plates. The broad, oval, flat foot is used for attaching to and creeping slowly over hard substrata. The mantle cavity is lateral and houses numerous gills. The head is small, lacking macroscopic sense organs and the nervous system consists of a circumenterric ring and longitudinal cords, mostly without ganglia.
Chitons are microphagous marine browsers living on rocks, often in shallow water. Chitons are adapted for life attached to hard surfaces on turbulent shores although many live in quieter deeper water. A well-developed radula is present but a crystalline style is absent. Digestion is extracellular. Chitons are gonochoric with external fertilization. The hatching stage is a trochophore larva but no veliger develops from it. Some species brood the young in the mantle cavity. This is one of the most ancient extant molluscan taxa.
Neoloricata O
Neoloricata is the only extant chiton higher taxon. The valves have articulamenta, which are lacking in the extinct Paleoloricata.
Ishnochitonina sO
Most chitons belong to this taxon in which most or all of the tegmentum is exposed.
Laboratory Specimens
1. Katharina tunicata is a common, large, shallow-water chiton of the northeastern Pacific, occurring from the Aleutian Islands to southern California (Fig 12-7A). It inhabits the intertidal zone of rocky shores. Preserved Katharina are available from biological supply companies. This is one of the rare instances in which preserved specimens are preferable to living for dissection in an introductory laboratory course. Dissection of living specimens is difficult.
External Anatomy
Dorsal Surface
Study the dorsal surface of a preserved chiton in a dissecting pan of tapwater. Note the bilateral symmetry and dorsoventral flattening. The animal is oval in dorsal view. The dorsal surface is arched, or convex, and the ventral surface is flat Fig 1, 12-6). The long axis coincides with the antero-posterior axis. There is no torsion or coiling.
The body wall covering the dorsum is the mantle (Fig 1). The mantle surrounds and secretes the shell, which, in all polyplacophorans, is composed of eight overlapping, calcareous plates, or valves. The valves are numbered from anterior to posterior and each valve overlaps the one posterior to it (Fig 12-6). Look at the valves at the two ends of the series and determine which is anterior. Division of the shell into several articulated plates provides flexibility and allows the animal to adjust its shape to irregularly contoured surfaces, such as rocks.
The mantle overlaps the lateral margins of the valves to varying degrees in different species. In Katharina most of the surface of each valve is covered by the mantle (Fig 12-7A) and very little of the valve is visible without removing the mantle. In Cryptochiton the entire surface of the shell is covered by the mantle and is not visible at all (Fig 12-7A). In most chitons, however, a relatively large, rectangular area of each valve is exposed (Fig 12-7B, 12-6C), more than in Katharina or Cryptochiton.
Figure 1. Dorsal view of the Pacific chiton, Katharina tunicata. Chiton11L.gif
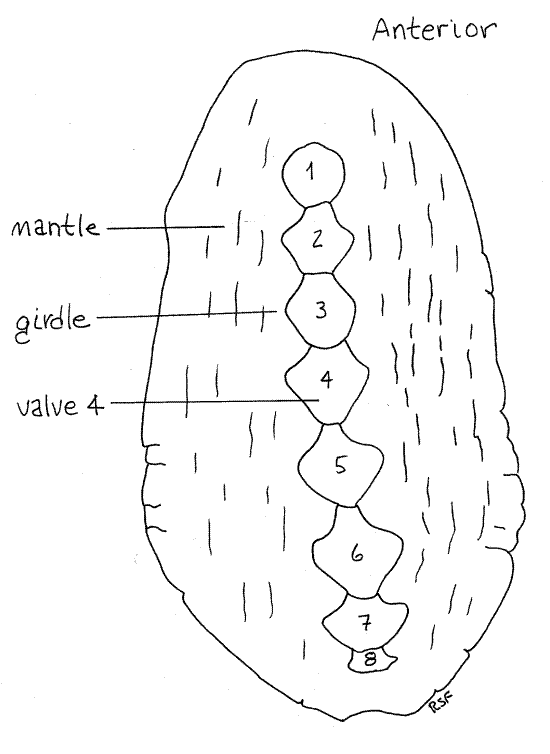
The lateral region of the mantle is thickened to form a girdle in which the shell is embedded (Fig 1, 12-6A). The girdle is tough and leathery and covered with a thick chitinous cuticle. The girdle often bears chitinous or calcareous spines or scales (Fig 12-6C). In Katharina, the girdle appears naked and smooth and is glossy black, although scattered microscopic spicules are present.
Venter
Turn the chiton over and look at the ventral surface (Fig 2, 12-8A). The central region is the flat, oval, muscular foot. The small head is anterior to the foot and is separated from it by a groove. The mantle cavity borders most of the ventral surface.
Figure 2. Ventral view of the chiton, Katharina. several gills have been removed from the posterior right mantle cavity to reveal the gonopore and nephridiopore. The stumps are indicated in the drawing. Chiton12La.gif
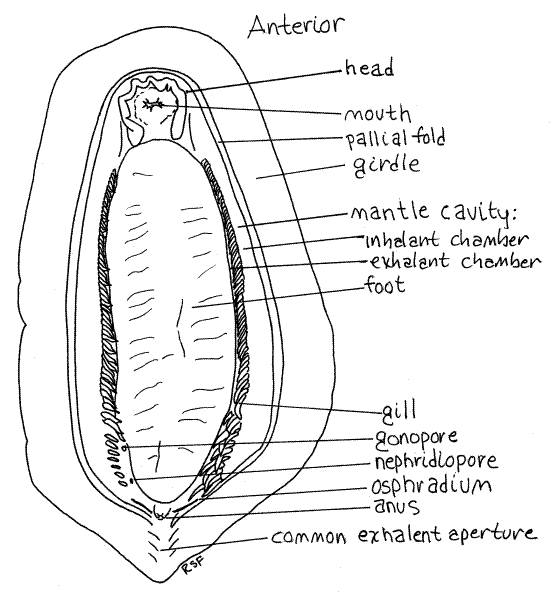
Head
The head is small and indistinct. It is located at the anterior end of the venter, immediately anterior to the much larger foot. A ridge of tissue partially surrounds the mouth. Unlike that of most bilaterans, the chiton head lacks a concentration of sense organs and there are no eyes or tentacles.
Foot
The foot dominates the ventral surface. It is muscular and its epithelium is abundantly supplied with mucus glands to assist in its role of gripping hard substrata. When dislodgement, by predator or waves threatens, the chiton seals the girdle margins to the substratum and lifts the inner margin of the girdle slightly, thereby creating suction that helps hold the animal to the substratum. Under these conditions it is almost impossible to remove a large chiton from a rock without damaging the animal (or the rock).
Mantle Cavity
The mantle cavity is a conspicuous groove encircling the body between the foot and girdle (Fig 2, 12-8A). It is best developed laterally. A row of 50-60 small gills is located in each lateral mantle cavity. (Some species have as few as six gills one each side, in which case they are found in the posterior mantle cavity.) The ciliated epithelium of the mantle cavity and gills generates a posteriorly directed flow of water that ventilates the gills.
Gills
Without removing it from the animal, look at a single gill with high power of the dissecting microscope. It consists of a flat, central axis that attaches by its base to the roof of the mantle cavity so the pointed apex of the gill hangs down into the cavity (Fig 12-8A). The axis bears numerous flat, closely spaced filaments attached perpendicularly to both sides (Fig 12-8B). Because there are filaments on both sides of the axis, the gill is said to be bipectinate.
>1a. With fine forceps grasp a gill at its base and twist it off the body wall. Place it in a drop of tapwater on a slide (seawater or magnesium chloride if your specimen is alive). Do not use a coverslip. Examine the gill with high power (ca 30-40X) of the dissecting microscope. It will probably be advantageous to use transmitted light for this (use the substage lamp or the compound microscope). Find the central axis and the filaments arising from its two broad surfaces. The flat surfaces of the filaments bear the lateral cilia which generate the respiratory current. Frontal cilia on the edges of the filaments remove debris from the gills. (If your animal is alive these cilia may be beating.) Look for the afferent and efferent blood vessels on the medial and lateral borders respectively (Fig 12-8B). <
Inhalant and Exhalant Chambers
Each row of gills separates its mantle cavity into a lateral inhalant chamber (canal) and a medial exhalant chamber (canal) (Fig 2, 12-8A). The respiratory current enters the mantle cavity along the sides, flows posteriorly in the inhalant chamber, crosses the gills, enters the exhalent chamber, and continues posteriorly to exit via a common exhalant aperture (canal) that crosses the posterior girdle.
The anus is on the roof of the mantle cavity at the posterior end of the foot (Fig 2, 12-8A). Water exiting the mantle cavity flows over the anus just before it leaves the animal.
Nephridiopores
The two nephridiopores open near the posterior end of the lateral mantle cavities but are usually very hard to find, especially in preserved specimens (Fig 2, 12-8A). Their position varies with species but they are always in the posterior end of the exhalant chamber near the largest (or one of the largest) gill. In Katharina the nephridiopores are at a level about halfway between the last gill and the penultimate gill. They are not in the gill row, rather are medial to it, in the roof of the exhalant chamber midway between the gills and the edge of the foot. Each is a tiny, longitudinal slit in the tough integument of the mantle roof. Use a bent microneedle to verify the existence of the opening in the integument but be careful that you do not accidentally manufacture a new opening by probing too hard. The integument of the roof of the mantle is tough and will resist probings where there is no pore. Probe suspected positions gently until the needle slips into an opening. When you think you have found a nephridiopore on one side, see if there is a similar opening in the same position on the other side.
Gonopores
In preserved material the gonopores are usually almost as obscure as the nephridiopores and their position also varies with species. They are always anterior to the nephridiopores (Fig 2, 12-8A). In Katharina they open on the top of an inconspicuous, low papilla on the roof of the exhalant chamber of the mantle cavity. They are a little closer to the gills than to the edge of the foot. These pores are in the vicinity of gills 4-7 (counting from the posterior end of the row) and under shell plates 6-7. The integument in the vicinity of the pore is minutely wrinkled. If you find a wrinkled area in approximately the right position, use a needle to demonstrate the existence of a pore in the center of the suspected area. Again, it is a good idea to look at the other side and see if there is a similar opening in the equivalent position.
Osphradia
The two so-called osphradia are areas of sensory tissue in the roof of the posterior mantle cavity. Each is a short, straight, dark, oblique line extending laterally and anteriorly from the anus toward the lateral mantle cavity (Fig 2). They are easily seen in living animals but not in preserved. They are downstream of the gills. Their function is not understood but they are presumed to be sensitive to silt and are perhaps chemosensory.
Shell
Study the shell of your specimen supplemented by a dried and disarticulated set of valves (Fig 3, 12-6). Each of the eight valves is arched dorsally and in most chitons overlaps the valve posterior to it and is embedded in the girdle.
Each valve is composed of four layers, one on top of the other. The tegmentum is the conspicuous layer exposed at the surface and surrounded by the surrounding girdle. It is the part of the valve visible in an intact animal. The tegmentum is composed chiefly of the protein conchiolin but also contains pigments and a small amount of calcium carbonate. The tegmentum is secreted by the medial edge of the girdle and this part of the girdle is attached to the edge of the tegmentum. The tegmentum is penetrated by epidermally lined canals which may be secretory or sensory (Fig 12-10, more later).
In Katharina the tegmentum is thick, dark, and relatively soft. It can be scraped away easily with a needle. Do this with your preserved specimen to expose the next layer, the hard, white, calcareous articulamentum, beneath it. The soft tegmentum is often worn away naturally and, if it is, you can see the articulamentum without scraping.
The articulamentum lies under the tegmentum and extends well beyond it under the neighboring girdle and the adjacent anterior valve (Fig 3, 12-6B). The articulamentum, which is entirely calcareous, is not visible in intact specimens. Use a teasing needle (not your microneedle) to push the girdle away from the tegmentum. This will expose the underlying white articulamentum. Note that the articulamentum extends far beneath the girdle. The articulamentum is secreted by the underlying mantle epidermis which you cannot yet see. Look at a dried valve for an unobstructed view of the articulamentum.
Figure 3. Representative valves of the shell of Katharina. Chiton13La.gif
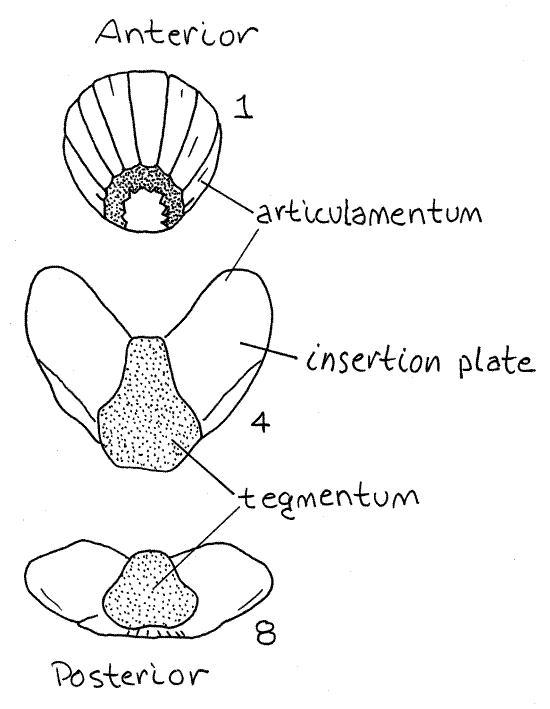
A very thin, inconspicuous organic periostracum covers the upper surface of the tegmentum and a second calcareous layer, the hypostracum, lies below the articulamentum.
The valves should be removed at this point in preparation for study of the internal organs. This is relatively easy to do with a preserved animal as the muscle attachments are much weaker than those of living chitons. Removing the valves is very difficult in living specimens.
Esthetes
The thickness of the tegmentum is crossed by tiny canals that extend almost to its surface (Fig 12-10). The canals are occupied by sense organs called esthetes. These originate as outgrowths of the mantle margin which extend between the tegmentum and articulamentum and do not pass through the articulamentum. The largest esthetes are photoreceptors, often equipped with cornea, lens, retina, and pigmented layer. The tegmentum over the end of the canal is thin and serves as a cornea. Adult chitons have no eyes on the head but, because of the esthetes, are sensitive to light. (The chiton trochophore larva, by the way, has a pair of eyes.) Look at the surface of the tegmentum with high magnification of the dissecting microscope. You should be able to see numerous tiny spots which are the corneas of the esthetes. They are sometimes easy to see, sometimes not so easy. If you have trouble finding them look at other valves, especially the first where their density is highest. It may help to brush the tegmentum with a toothbrush. If one of the valves has an area where the tegmentum is partially eroded, you may be able to see the canals extending across the tegmentum to the esthetes. Look at an exposed area of articulamentum and note that it is NOT perforated by these canals.
Esthetes are easiest to see in young individuals because their valves are less fouled by debris or other organisms. Because of interference by increasing amounts of fouling on the valves, older individuals rely less on photoreception and are not as responsive to light intensities.
& Use a sharp scalpel to make 2-4 deep slits radiating outward across the surface of valve 1. Make these cuts from the edges of the tegmentum completely through the girdle and across the underlying articulamentum. To avoid damage to the body cavity and its organs, do not cut through areas of the girdle that are not underlain by shell. Grasp the valve by its exposed posterior edge with heavy forceps and pull posteriorly. The valve should slip easily out of the pocket that contains it. If it does not, extend the slits or make more of them but do not damage the valve or the soft tissues below it. With a pencil, label its ventral surface with a "1" and set it aside.
Move to the second valve and, on each side, make a single, deep, transverse incision through the girdle to the articulamentum. Free the tegmentum from the girdle (Fig 1). Grasp the valve with strong forceps and pull posteriorly to remove it as before. Once removed, label it with a "2". Note that this valve is strongly arched dorsally and that it has two large, anterolateralinsertion plates that in life extend under the adjacent anterior valve and the girdle. They are composed entirely of articulamentum and hypostracum and are not overlain by tegmentum. Plates 2-7 are similar to each other and the same methods are used to remove them. Label each valve before removing the next one.
Remove the eighth valve by making two posterolateral incisions to the margin of the articulamentum. Pull this valve straight up to remove it. Label it.
Arrange the valves on a piece of paper as they would be in an intact chiton. Since each valve overlaps the one behind it is best to start with valve 8 and proceed anteriorly.
>1b. (Do not do the following if you or your instructor wish to keep the valves intact.) Place one of the valves in a small culture dish of household bleach. Place a second valve in a dish of 10% HCl. Label the two dishes. Look at the valves at the end of the laboratory period and compare the effects of the two solutions on the tegmentum and articulamentum. Which was altered the most by treatment with bleach? ____________________ With acid? __________________ What components of the valve would you expect to be removed by bleach? ____________________ By acid? ______________________ What conclusions would you draw from this about the composition of the two parts of the valve?
Internal Anatomy
Removal of the valves has exposed the dorsal body wall which is much thinner here than in the region of the girdle. It consists chiefly of the thin epidermis and underlying muscles and connective tissue. Inside it is a cavity, the hemocoel in which are located most of the internal organs. Be very careful that you do not damage the body wall, hemocoel, or organs until instructed to do so.
Valve Musculature
Much of a chiton's musculature is associated with the valves and can be seen through the dorsal body wall after the valves have been removed. Some of the muscles you see run from valve to valve to hold the valves together and change their orientation with respect to each other, whereas others run from the valves to the foot, girdle, or mantle. Most conspicuous are seven pairs of transverse muscles that form high ridges across the top of the body between the valves.
Muscles attach to the valves through the mediation of tonofilaments and microfilaments running through the intervening epidermis and attaching to the shell. You broke these connections when you removed the valves. These connections are vastly weakened in preserved specimens and this is why their valves are so much easier to remove.
The valve muscles may be either intrinsic, originating and inserting on adjacent valves, or extrinsic, inserting on a valve but originating in the body. In general these muscles are repeated serially and each valve has a set of them but some of the muscles are reduced or absent on the eighth and sometimes the first valve.
Intrinsic Valve Muscles
Four sets of intrinsic valve muscles are present. 1. The rectus muscles are longitudinal muscles clearly visible through the body wall near the dorsal midline. They originate on the anterior midline of one valve and run anteriorly to insert near the midline of the next valve. 2. The oblique muscles run diagonally anterolaterally from the anterior midline of one valve to a lateral insertion below the preceding valve. 3. A lateral longitudinal muscle makes an unbroken oval ring around the periphery of the shell. It is composed of dorsal and ventral fibers. Its dorsal fibers originate on the ventrolateral surface of the insertion plate of one valve and insert on the ventral lateral surface of the adjacent preceding valve. The ventral fibers, however, are not attached to the shell and form an unbroken ring of muscle around the edge of the shell. This is the enrollment muscle that enables chitons to roll into a ball. 4. The transverse muscles, which were mentioned earlier, are thick pads of muscle in the overlap between two adjacent valves. They form the seven transverse ridges arching across the dorsal body wall that are so conspicuous after the valves are removed. Their fibers are oriented more or less dorsoventrally and run from the dorsal surface of the anterior insertion plate of one valve to the ventral surface of the posterior edge of the adjacent preceding valve.
Extrinsic Valve Muscles
Extrinsic valve muscles run from various places on the ventral surface of the valves to other parts of the body. Each valve has a pair of strong anterior lateropedal and posterior lateropedal muscles that originate on the valve and insert on the foot. Other muscles, the inner mantle muscles, run from the valves to the thick girdle lateral to the mantle cavity. Radial muscles extend from the lateral edges of each valve into the girdle. The girdle also contains longitudinal muscle fibers that are not associated with the valves.
Body Cavity
& Use strong scissors to remove the medial pieces of girdle remaining after removal of the valves. Do not try to remove the still-intact lateral girdle. This will completely expose the tough, thin, translucent dorsal body wall. Refer to Figure 4, then follow the instructions to very carefully remove the dorsal body wall and expose the hemocoel. Some of the structures in the hemocoel adhere tightly to the body wall and may be inadvertently removed if you are not careful.
& Use fine scissors to make a small anterior opening in the body wall as far to the side as the girdle will permit. Lift the body wall with forceps and carefully cut posteriorly along the side. Look inside the lengthening opening to be sure you do not cut any organs. Gently insert a teasing needle into the opening and use it to separate the organs from the body wall without damaging the organs. It will be necessary to cut the muscles associated with the body wall. Keep the incision as far laterally as the girdle permits. When, cutting posteriorly, you reach the level of valve 7, be particularly careful of the thin, membranous pericardium and the heart which adhere tightly to the posterior end of the body wall (Fig 12-8C).
When all organs are freed, make a similar longitudinal, lateral incision on the opposite side, connect it anteriorly and posteriorly with the existing incision, and carefully remove the dorsal body wall and muscles.
The large space you have uncovered is the functional body cavity of the animal but it is not a coelom and is not lined with mesothelium or peritoneum. Instead it is part of the hemocoelof the hemal system and in life it contains blood (Fig 4, 12-9A).
Viewed dorsally, the major landmarks in the hemocoel are the large gonad in the middle region where it may cover much of the digestive system, the digestive ceca, and the pericardium and heart (Fig 4, 12-8C, 12-9A). The organ systems will be studied in the order in which they are most conveniently exposed by a dorsal dissection.
Hemal System
Chitons have a hemal system consisting of a heart, vessels, and an extensive system of unlined sinuses that make up the hemocoel.
The pericardial cavity is a coelomic space surrounding the heart. It is a membranous sac occupying the posterior end of the body cavity Fig 4). It extends from one side of the hemocoel to the other but is very thin and flattened. It occupies the space under the last two shell valves. The pericardial cavity is enclosed by the pericardium, a type of peritoneum. The pericardium and the wall of the heart are closely associated and difficult to distinguish from each other in reserved specimens.
Figure 4. Dorsal dissection of the chiton, Katharina. Chiton14La.gif
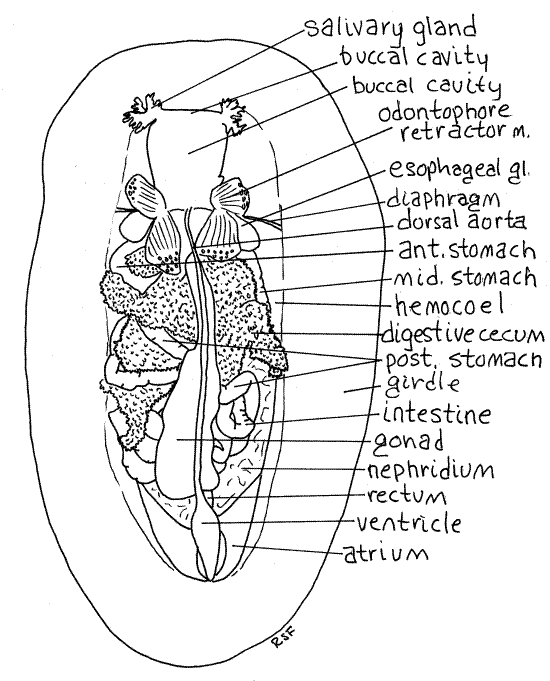
The heart consists of a single median ventricle and the two lateral atria draining into it (Fig 4, 12-8C, 12-9A). The ventricle is a flattened tube lying on the dorsal midline. It ends blindly at the posterior end of the hemocoel. At this position its pointed tip is attached to the connective tissue of the body wall. Anteriorly the ventricle narrows to become the dorsal aorta, which carries blood from the heart to the hemocoel. The aorta extends anteriorly on the dorsal midline over the gonad to the head where it empties into the hemocoel. Blood from the hemocoel goes to the foot, gills, and finally returns to the atria. There is a crescent-shaped atrium on either side of the ventricle. Each empties into a side of the ventricle. The atria receive oxygenated blood from the gills.
The hemocoel has several subdivisions, the two largest of which are the perivisceral hemocoel which is the space, already noted, surrounding the viscera of the posterior 4/5 of the body and the cephalic hemocoel which surrounds the tissues in the anterior fifth of the body. The cephalic hemocoel appears to be continuous with the perivisceral hemocoel but in reality is separated from it by a thin transverse, vertical partition, the diaphragm. The diaphragm is inconspicuous.
The pattern of blood flow in chitons is straightforward. Blood flows anteriorly from the ventricle via the dorsal aorta to the cephalic hemocoel. On its way the aorta gives off transverse vessels to the gonad and valve muscles. The aorta penetrates the diaphragm and ends by opening into the cephalic hemocoel. From the cephalic hemocoel blood enters the visceral artery, which extends posteriorly, penetrating the diaphragm once again, to open into the perivisceral hemocoel. Here the blood bathes the visceral organs and then flows to sinuses of the pedal hemocoel in the foot. The blood, now deoxygenated, eventually enters the afferent branchial vessels to the gills, is oxygenated, and then leaves via the efferent branchial vessels to the atria. The atria empty into the ventricle whose contractions pump blood to the dorsal aorta.
Reproductive System
Most chitons are gonochoric but not sexually dimorphic. The single large, median gonad, either ovary or testis, occupies much of the space in the hemocoel (Fig 4, 12-8C, 12-9A). Like the heart, the gonad occupies a derivative of the coelom. Near its posterior end two inconspicuous gonoducts leave its dorsal surface and extend, one on each side, along the anterior border of the pericardium to the lateral body wall. Each gonoduct penetrates the body wall and opens to the exterior via a gonopore in the mantle cavity.
Gametes are shed from the gonopores into the exhalant chamber of the mantle cavity. In most species they are then carried to the sea by the exhalant respiratory current. There is no copulation and fertilization is external. The yolky eggs undergo spiral cleavage to produce a planktonic trochophore larva that settles to the bottom as it metamorphoses. There is no veliger larva.
Excretory System
& Gently lift the gonad, free it from the pericardium, dorsal aorta, and gonoducts, and remove it. This will expose the middle region of the hemocoel which, now that the gonad is gone, is dominated by the coiled mass of the intestine (Fig 4, 12-8C).
The excretory system of chitons consists of two branched metanephridia, or kidneys. Although they are large, the metanephridia are difficult to see in preserved animals (Fig 12-9A). They are about the same color as the floor of the hemocoel. In preserved material they are collapsed and lie flat against the floor giving it a grainy appearance. Each is a large sac extending for most of the length of the hemocoel.
Each nephridium is an elaborate tube extending from the pericardial cavity to the exterior and is bathed in blood. Each begins at a ciliated nephrostome, which you will probably not see, that opens from an anterolateral corner of the pericardial cavity. The nephrostome opens into the renopericardial canal that leads to the lumen of the nephridium (Fig 12-9A). A duct exits the nephridium and runs to a nephridiopore on the roof of the mantle cavity (Fig 12-8C).
Ultrafiltration of the blood produces primary urine in the pericardial cavity. Cilia move it into the nephrostome, through the renopericardial canal, to the lumen of the metanephridium. The nephridium is immersed in the blood of the hemocoel and the composition of the primary urine is modified by exchange with the blood across the walls of the metanephridium. As it moves along the metanephridium and undergoes modification, the primary urine gradually becomes the final urine which exits into the exhalant chamber via the nephridiopore.
Digestive System
Chitons are microphagous browsers that use a rasplike radula to scrape microalgae and associated organisms from the surface of rocks. The scrapings are mixed with mucus and moved into the gut by retraction of the radula (Fig 12-2). Further passage through the gut tube is accomplished by cilia and not by muscular peristalsis. The mouth and anus are located at opposite ends of the animal and the long, much-coiled gut tube with its associated organs is arrayed between them in the hemocoel.
The buccal mass fills most of the anterior hemocoel in the head under valve 2 (Fig 4, 12-8C). It consists of the buccal cavity, pharynx, radula, odontophore, and pharyngeal glands. The myriad small muscles associated with the buccal mass are visible lateral and ventral to it. Most of these muscles are associated with the radula or its supporting structure, the odontophore, and serve to protract or retract the radula. There are 82 muscles in this complex. The odontophore retractor muscles are the most conspicuous (Fig 4). Muscles of the buccal mass may contain myoglobin and will be red or orange in living specimens.
Buccal Cavity
Relocate the mouth at the center of the head on the ventral surface. It opens into a vertical tube, the buccal cavity, directly above it and visible in the hemocoel (Fig 4). The buccal cavity is located below valves 1-2 and is the dorsalmost structure in this region. It is a wide, thin-walled sac lying on top of other parts of the anterior digestive system. A conspicuous cluster of cordlike muscles extends from under each posterolateral corner of the buccal cavity. Posteriorly the buccal cavity narrows to become the short esophagus (Fig 4, 12-8C). The posterior region of the buccal cavity (between the radular sac and the esophagus) is sometimes referred to as the pharynx.
& With fine forceps lift the dorsal wall of the buccal cavity and use fine scissors to open it with a median, longitudinal incision. Extend the incision as far anteriorly as possible. Be careful handling the delicate tissues of the gut. Reflect the walls of the buccal cavity and pin them aside with a pair of #000 insect pins so you can look into the lumen. Use a needle to demonstrate the continuity between the mouth and buccal cavity.
Two large salivary glands (= buccal glands) open into the anterior buccal cavity. These branched, yellowish glands project into the hemocoel from the anterolateral corners of the buccal mass and secrete mucus into the gut lumen.
The subradular sac and the radular sac are large pouches extending posteriorly from the buccal cavity, one above the other. The radula and odontophore are between the two sacs.
Radular Sac
The radular sac is the more dorsal, and most accessible, of the two sacs (Fig 12-8C). It connects with the buccal cavity by a narrow slit in the floor of the cavity. Look through this slit to see the anterior end of the radula in the radular sac (Fig 12-8C). The radula is a conspicuous and unmistakable feature of this part of the gut. It resembles a zipper.
& Begin anteriorly and open the radular sac with a median longitudinal incision through the floor of the buccal cavity and roof of the radular sac. Make the incision with fine scissors. Do not extend the incision beyond the posterior end of the buccal cavity at this time, even though the radula and radular sac are much longer.
Reflect the roof and walls of the radular sac and pin them aside with two #000 insect pins if needed. Examine the radular sac with magnification and notice the numerous muscles associated with its walls. The radula is a long ribbon composed of successive transverse rows of amber or black, chitinous teeth, some of which are capped with an iron compound known as magnetite.
Radular teeth are secreted far to the rear in the posterior radula sac, which you have not yet seen. The radula grows forward so the oldest, hardest, and largest teeth are at the anterior tip. This is the end that is extended from the mouth and used to scrape algae from rocks. The more posterior teeth are not yet in use.
The radula is supported by an odontophore (= tooth bearer) beside and slightly ventral to the anterior end of the radular sac. The odontophore is composed of two elongate, connective tissue bars called bolsters. The bolsters diverge laterally from each other like the two arms of a "V". The anterior ends of the bolsters approach each other near the midline but do not touch. They are connected to each other by muscle and connective issue. Numerous muscles are associated with the bolsters.
Subradular Sac
The subradular sac is the more ventral of the two buccal sacs. It contains the two sensory subradular organs. At present it is hidden by the buccal cavity and radular muscles and cannot be seen. It cannot be exposed without damaging other parts of the digestive system and the nervous system. Postpone dissecting the subradular sac until after you have completed the study of the nervous system. During feeding the subradular sac is everted through the mouth to place the subradular organs in contact with the substratum, which they test for the presence of food. You will study the radular and subradular sacs in more detail later.
Esophagus
At the level of the second transverse muscle the buccal cavity ends and the gut tube narrows, turns to the left, and becomes the short esophagus (Fig 12-8C). A pair of large, hollow, ovoid esophageal glands (= pharyngeal glands, sugar glands) lie close beside the posterior buccal cavity at the junction of the buccal cavity with the esophagus (Fig 4, 12-8C). They extend from near the middle of valve 2 to the middle of valve 3 and end at the posterior end of the esophagus. They are hollow diverticula of the esophagus. Secretory cells in the walls of the gland produce digestive enzymes, including amylase and other carbohydrases that are released into the gut lumen.
Stomach
Posterior to the esophagus the gut tube widens markedly to become the large stomach. The stomach has thin translucent walls and most of it is obscured by the large digestive ceca. The stomach is a large, asymmetrical sac located under valves 3 and 4. It spirals around the posterior end of the radular sac (which is very long) and in dorsal view appears to be three separate chambers with the intervening connections hidden by the digestive ceca (Fig 4).
Two large digestive ceca open into the stomach (Fig 12-8C). The anterior digestive cecum is the smaller and lies around the stomach. The larger posterior digestive cecum is associated with the coils of the intestine and is mostly posterior to the stomach. They are irregularly shaped and fill most of the space around the stomach and between the loops of the intestine. The digestive ceca produce digestive enzymes and are responsible for hydrolysis and absorption. Digestion, which is almost entirely extracellular, takes place in the ceca, stomach, and anterior intestine.
& Carefully remove the digestive ceca but do not damage the stomach or other organs.
The stomach consists of three distinct regions. The anterior stomach is on the left, immediately posterior to the left esophageal gland (Fig 4). The esophagus opens into its anteromedial corner. This part of the stomach is a short wide tube with its long axis oriented transversely. Open it along its long axis with your fine scissors. The second region, the middle stomach, is on the far right with its long axis oriented longitudinally. Open it along its long axis. The posterior stomach is on the left, posterior to the anterior stomach. Its long axis is oblique. Open it also. With a blunt probe explore the connections between the regions. The anterior stomach is continuous with the middle stomach, which is continuous with the posterior stomach. Note the longitudinal central core around which the stomach winds. You will later open this core and find the radula inside. The posterior stomach slopes diagonally from the left to the right and when it arrives at the right side of the body becomes the anterior intestine.
Intestine
The stomach is continuous with the long and much coiled intestine which fills much of the space in the middle and posterior hemocoel (Fig 4, 12-8C). The intestine consists of an anterior intestine and posterior intestine separated by a sphincter valve. The valve can be seen if this region of the intestine is opened. The anterior intestine is involved in extracellular digestion whereas the posterior intestine is responsible for formation of fecal pellets. After making a series of loops and turns, the intestine extends straight to the anus in the posterior mantle cavity. On the way the intestine widens almost imperceptibly under the pericardium to become the rectum. The epithelium of the posterior intestine and rectum are ciliated.
Radular Sac, Radula, and Subradular Sac
& Finish opening the radular sac by extending the median longitudinal incision in the floor of the buccal cavity. Your incision will pass through the esophagus and much of the stomach before it reaches the end of the sac. Remove the radula from the sac by cutting the muscles that hold it in place.
>1c. The radula is much longer than you probably expected. Use a centimeter rule to measure and record the length of the radula in millimeters. Compare the length of the radula with the total length of the animal, which you measured earlier. What is the ratio of radula length to the length of the chiton? <
>1d. Place the radula on a slide, without water or coverslip, and place it on the stage of the dissecting microscope. Use your fingers to flatten the radula on the slide and study it at about 15-25X. You can now see its teeth clearly. They are arranged in transverse rows but it is not always easy to distinguish successive rows from each other. In all chitons each transverse tooth row has 17 teeth consisting of a median tooth flanked by eight teeth on each side. The second tooth on each side of the median tooth is much larger than the others, has three cusps, and is capped with black magnetite. These special teeth form two conspicuous, longitudinal rows that are easily seen. <
>1e. Place the radula on something warm, such as the transformer or housing of your microscope lamp, to dry it. When it is dry, move it to a piece of white paper and place a small magnet above the radula and observe what happens. Place the magnet below the paper and see if you can move the radula across the paper with the magnet. Do these observations support the claim that the teeth are capped with an iron compound? Why might iron-capped teeth be adaptive for chitons?
>1f. AFTER you have completed the dissection of the radula, find the subradular sac. It is a large soft pouch opening from the posterior wall of the buccal cavity ventral to the radular sac. You will have to cut the many small muscles of the buccal mass that obscure your view of the tissues ventral to the buccal cavity. Once the muscles are cut, roll the anterior gut partly over on its side and look beneath it. The subradular sac is a wide pouch between the anterior end of the radular sac and the floor of the hemocoel. Look into the buccal cavity and find the internal opening of the mouth. Look on the posterior wall of the buccal cavity between the opening to the radular sac and the mouth. You will see the large opening to the subradular sac. Insert a probe or needle into the opening and see if it enters the sac you identified in side view as the subradular pocket. <
Nervous System
The nervous system consists of a circumenteric ring and two pairs of longitudinal nerve cords as well as cords serving the buccal and pharyngeal region of the gut (Fig 12-9B). The system is tetraneurous, there being two visceral longitudinal cords, to serve the gut and hemocoel, and two pedal longitudinal cords, to serve the foot. Few ganglia are present and those are weakly developed. The nerve cell bodies are in the nerve cords instead of ganglia. The only ganglia are those associated with the subradular organs and the buccal region. Since the cords contain cell bodies, axons, and dendrites they are referred to as cords, rather than nerves (a nerve is a bundle of axons).
The nervous system of preserved animals is pale creamy white, sometimes with tiny white spots apparent through the surface. Study of the nervous system is beyond the scope of this exercise and, furthermore, much of the system will have been destroyed during dissection of the digestive system.
Behavior
<1g. If small, living chitons, such as Chaetopleura apiculata from the east coast of North America, are available in the laboratory, they can be used for a study of the external features of living animals. With a minimum of disturbance, place one on a glass slide in a 4" culture dish of seawater. The chiton should be extended with its ventral surface in contact with the slide. Push the animal gently, but firmly, against the surface of the slide to encourage it to attach to the slide. It may insist on rolling into a ball (what muscles does it use for this?) and, if it does, you will not be able to use it. Put the dish on the stage of the dissecting microscope and allow the animal time to attach to the slide then study the dorsal surface with low magnification.
Find the shell, with its eight valves, and the girdle surrounding it. With higher magnification examine the dorsal surface of the girdle looking for small chitinous scales or tiny spicules.
With a fine pipet place a drop carmine/seawater suspension in the water around the anterior end of the chiton. Watch closely for movement of the particles. Can you tell where they enter the mantle cavity? Can you see particles leaving by the exhalant current?
If the animal is firmly attached, gently turn the slide over so you can see the ventral surface. Support each end of the slide with a short piece of thick glass rod so the chiton is suspended above the bottom of the dish. Look at the ventral surface and find the head, mouth, foot, girdle, and mantle cavities. Look for gills in the mantle cavities and the anus in the posterior.
Squirt a cloud of carmine/seawater under the slide at the anterior end of the chiton and look once more for the respiratory current. See if you can find the raised areas of the girdle where the water enters the inhalant chamber and look for water leaving the posterior mantle cavity. What seems to happen to the carmine particles? Do they clear the gills and exit the mantle cavity with the respiratory current or do they get tangled in mucus and leave in clumps?
Look at the mouth and try to identify the toothy, rasplike radula. Chaetopleura usually is not enthusiastic about browsing on glass slides but it may occasionally extend its radula. <
>1h. Use a paintbrush to apply a coat of India ink to the surface of a plate of glass or a microscope slide and let the ink dry thoroughly. Place the slide in a dish of seawater and put a small living chiton, such as Chaetopleura, on it, letting it attach and crawl over the surface. If the chiton makes feeding motions with its radula, it will scrape the ink away, leaving a characteristic pattern on the glass. Remove animal from the glass and the glass from the dish. Gently rinse the slide with tapwater and let it dry. Examine the markings on slide with high power of the dissecting microscope. Use the compound microscope if you need additional magnification. <
References
Abbott DP . 1987. Observing Marine Invertebrates. Stanford Univ. Press, Stanford. 380p.
Hyman LH . 1967. The Invertebrates, Mollusca, vol. VI. McGraw-Hill, New York. 792p.
Purchon RD. 1977. The Biology of the Mollusca 2nd ed. Pergamon, Oxford. 560p.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Supplies
Dissecting microscope
Compound microscope
Dissecting pan
Dissecting set with microdissecting tools
Preserved chitons
Dried chiton valves
6 cm Carolina culture dishes
bleach
10% hydrochloric acid
Small magnet
#000 stainless steel insect pins