Invertebrate Anatomy OnLine
Cassiopeia xamachana ©
Upside-down Jellyfish
30jun2006
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises can be accessed by clicking on the links to the left. A glossary and chapters on supplies and laboratory techniques are also available. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Cnidaria P, Medusozoa, Scyphozoa C, Rhizostomeae O, Cassiopeidae F (Fig 7-56, 7-75)
Cnidaria P
The cnidarian body consists of a central blind sac, the coelenteron (= gastrovascular cavity), enclosed by a body wall comprising two epithelia, the outer epidermis and the inner gastrodermis (Fig 7-1, 7-2). A gelatinous connective tissue layer, the mesoglea, lies between the two epithelia. The mouth opens at one end of the coelenteron and marks the oral end. The mouth is at the tip of a process, the manubrium that elevates it above the oral surface. The opposite pole is the aboral end. The imaginary line connecting the oral and aboral poles is the axis of symmetry around which the radial symmetry of the body is organized. The mouth is usually surrounded by one or more circles of tentacles.
The defining cnidarian feature is, of course, their possession of stinging cells, or cnidocytes (Fig 7-8). Characteristic of the epidermis, they are also sometimes found in the gastrodermis. Cnidocytes contain an explosive organelle, the cnida, which, upon proper stimulation, inverts and ejects a slender, often barbed and toxic thread in the direction of prey or predator (Fig 7-9). Three types of cnidae are found in cnidarians (Fig 7-10). Nematocysts (in nematocytes), spirocysts (in spirocytes), and ptychocysts (in ptychocytes). All toxic cnidae are nematocysts whereas spirocysts are sticky, and the everted tubules of ptychocysts are used for constructing feltlike tubes. Most cnidae are nematocysts and these are present in all three higher cnidarian taxa. Spirocysts and ptychocysts are found only in Anthozoa.
The basic body plan described above can be manifest as a swimming medusa or attached polyp. In some taxa only one generation is present whereas in others both are found. A life cycle featuring alternation of sexual, swimming medusae with benthic asexual polyps is typical of many cnidarians.
All cnidarians are carnivores feeding on live prey which they usually capture using tentacles armed with cnidocytes. Digestion occurs in the coelenteron which is typically equipped with ciliated canals for distribution of partly digested food. Cnidarians are ammonotelic and diffusion across the body and tentacle surface eliminated the ammonia from the body. Gas exchange is across the general body surface. The nervous system is a plexus of basiepithelial neurons serving sensory and motor systems (Fig 7-6). Most cnidarians are gonochoric. The life cycle typically includes a planula larva. Cnidarians are chiefly marine but the well-known Hydra is an exception.
Medusozoa
Medusozoa comprises those cnidarians whose life cycle includes a medusa generation that alternates with a polyp generation (Fig 7-75B). Symmetry is radial and tetramerous. Nematocysts are the only type of cnidocyte present. Included taxa are Scyphozoa (jellyfishes) and Hydrozoa (hydroids, Hydra, Portuguese men of war, etc).
Scyphozoa C
Scyphozoans, the jellyfishes, are cnidarians in which the medusoid generation is large and noticeable and the polypoid is small and inconspicuous. The medusae, known as scyphomedusae, tend to be large, mobile, pelagic, drifting, solitary carnivores and all are marine. The mesoglea of the medusa is thick, gelatinous, contains cells, and accounts for most of the mass of the organism, although it is mostly water. Cnidocytes are present, concentrated on tentacles, and are used for prey capture and defense. Locomotion is by muscular contraction antagonized by elastic recoil of the mesoglea. The velum characteristic of hydromedusae is absent. Sense organs are arrayed around the periphery of the organism. The sexes are separate. Polyps are small benthic scyphistomae. Scyphozoa includes about 200 species in five taxa; Semaeostomae, Rhizostomeae, Cubomedusae, Coronatae, and Stauromedusae.
Rhizostomeae O
The “root-mouth” jellyfishes (Fig 7-50) are characterized by the absence of a single central mouth. Tentacles are absent and the four oral arms are fused together at the center of the oral surface. Numerous small secondary mouths open along the oral arms of the manubrium. Canals extend from the secondary mouths through the oral arms to the stomach. Rhizostomes are suspension feeders that use the secondary mouths to ingest zooplankton.
Laboratory Exercise
Cassiopeia xamachana is known as the "upside down jellyfish" because it lies on the bottom with the aboral surface down, against the sand, and the oral surface up, exposed to the overlying water(Fig 7-50B). It inhabits shallow, quiet, sunlit, tropical, marine waters, usually on soft bottoms. It harbors zooxanthellae in its tissues and can exist, and even grow, utilizing photosynthate as its sole source of nutrition.
Casssiopeia xamachana, one of many species its genus, is common in south Florida where it reaches a diameter of about 15 cm. Individuals 5 cm or smaller are convenient for dissection because they fit into a culture dish small enough to be placed on the stage of the dissecting microscope. Furthermore, living specimens less than 5 cm are translucent and their internal anatomy can be studied using transmitted light and, although still desirable, dissection is not essential. The exercise is written for living (preferred) or preserved specimens. Much of the study should be performed on the stage of a dissecting microscope if the size of your specimen permits.
External Anatomy
Orientation
Place a small jelly in a glass fingerbowl of seawater (tapwater if using preserved material) and make an initial examination without magnification. The body of a jellyfish is known as thebell (Fig 1) because it is usually shaped more or less like a bell. That of Cassiopeia, however, is flat. The aboral surface (= exumbrella) of the bell is smooth and slightly concave (Fig 1). The oral surface (= subumbrella), which will be studied later, bears a complex of frilly arms and appendages and the two surfaces are easily distinguished from each other. The oral-aboral axis is an imaginary line passing through the centers of these two surfaces. It is the axis of radial symmetry. There are no marginal tentacles.
Figure 1. Cassiopeia in side view. Redrawn and modified from Hyman (1940). Scyphozoa11L.gif
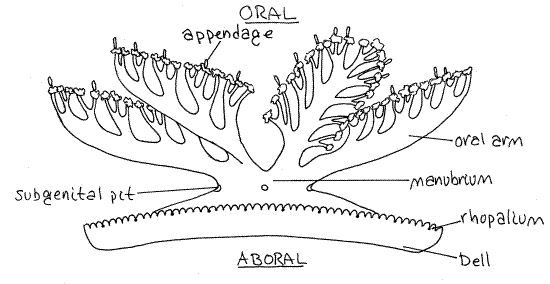
Scyphozoans are tetramerously radially symmetrical and have multiple planes of symmetry. Locate some of these planes. Each plane must include the axis of symmetry and divide the animal into mirror images. In reference to radially symmetrical animals the term peripheral is used in reference to the margins of the bell remote from the axis of symmetry whereas centraldenotes locations at or towards the axis.
Gently turn the animal so the aboral surface is uppermost, if it is not already so. The concavity of the aboral surface resembles a large sucker and, in fact, assists the animal in maintaining contact with the substratum via suction. With it the jelly can remain in place on smooth vertical surfaces. In nature this is the surface that faces downward, hence the name "upside down jelly".
Manubrium
Gently turn the animal over so the frilly oral surface faces up. This is the animal's life position and its orientation when feeding. The oral surface is covered by the feeding apparatus consisting of the manubrium and the oral arms.
The manubrium is a short central column emerging from the center of the oral surface of the bell (Fig 1). Hold the animal so you can view it from the side and relocate the large disk-like aboral surface. The short manubrium hangs below it.
Oral Arms
Eight frilly oral arms are attached to the free end of the manubrium and radiate outward from it (Fig 1).
Figure 2. Oral surface of Cassiopeia from Big Pine Key, Florida. Only one of the eight oral arms is shown in its entirety. The oral collars have been removed from all but two of the secondary mouths. Scyphozoa12L.gif
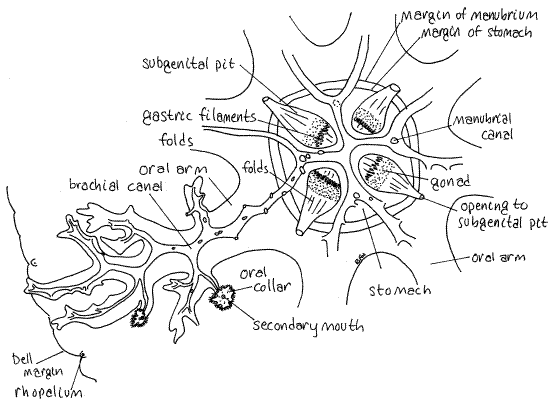
In most scyphozoans the oral apparatus consists of four oral arms surrounding the mouth in the center of the oral surface. The mouth opens into the stomach above (aboral to) it in the center of the bell.
In the rhizostomes, however, some changes have been made in this ancestral. The four oral arms are present but each is bifurcated so there are eight functional arms radiating from the manubrium in the center of the oral surface. The original four arms are perradial (Fig 2) as they are in other scyphozoans. Since each of the original arms is divided into two, the resulting eight arms are adradial. The two arms resulting from the division of one original arm flank a perradial axis. The interradial axes separate adjacent pairs of arms (Fig 3).
The oral arms are highly branched and contractile. In other scyphozoans the oral surface of the each oral arm bears a ciliated brachial groove that runs from the tip of the arm to the mouth. In rhizostomes the edges of the ciliated groove have grown together to form a closed tunnel, or brachial canal (Fig 2). There are eight of these canals and their branches correspond to the branches of the arms. The edges of the mouth have also grown together and it is permanently closed (in most species). Consequently most rhizostomes have no central, i.e. primary, mouth. Instead, the many branches of the brachial canals open to the sea via numerous tiny secondary mouths scattered about the branches of the arms (Fig 2). Secondary mouths are openings in the tissue that encloses the brachial canal. Each secondary mouth is surrounded by a frilly oral collar. Centrally the brachial canals empty into the stomach.
Figure 3. Diagrammatic representation of the brachial canals and axes of Cassiopeia. Scyphozoa13L.gif
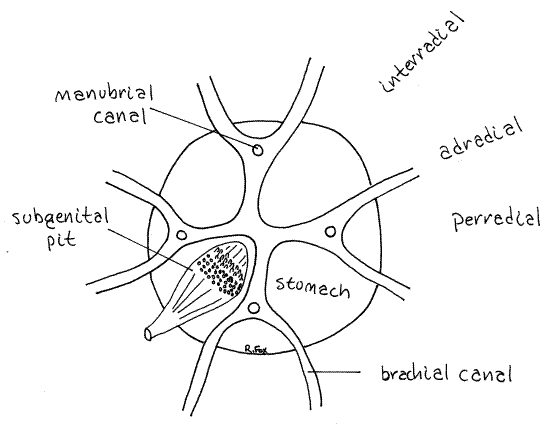
Feeding
Cassiopeia rests on its "back" (aboral surface) on the bottom and pulsates the margins of the bell to move fresh water over the surface of the arms. This brings small crustaceans (and oxygen) to the oral (upper) surface of the arms. The food organisms are stung by cnidocytes, entangled in mucus, and moved into the secondary mouths.
Food items enter the brachial canals via the ciliated secondary mouths and are moved through the canal by a ciliary current to the stomach. The arrangement of these mouths on the highly branched arms is the basis for the name rhizostome, or "root mouth".
1a. >If you have a living animal, watch the margins of the bell pulsate. Add some dye/seawater to visualize the currents generated by the pulsations. Do the pulsations move water over the oral arms as stated ? <
1b. >As you examine the oral surface, look for the nudibranch (sea slug), Dondice parguerensis, which feeds on the oral arms of Cassiopeia in Puerto Rico but may eventually be found elsewhere in the Caribbean. Another nudibranch, Austraeolis catina, is sometimes found on Cassiopeia in south Florida. The cyclopoid copepod, Sewellochiron fideus, has also been reported from the oral surface of Cassiopeia in Puerto Rico. <
1c. >If your specimen is living, replace the water in its dish with 33% magnesium chloride and let it relax. It will relax rapidly. <
Place the dish with the relaxed, or preserved, specimen on the stage of the dissecting microscope and examine the arms on the oral surface with low power. Count them and verify that there are eight.
Secondary Mouths
Look for the small secondary mouths on the oral surfaces of the arms. Each is surrounded by a ruffled lacelike oral collar of tissue. The oral surface of each arm has a frilly appearance due to the presence of these hundreds of collars.
The edge of each collar bears tiny papillae. With your nadel (see techniques chapter) push the rim of one of the collars aside and find the secondary mouth near its center. Put thenadel in the mouth to demonstrate that it is, in fact, an opening and that it leads to a canal within the arm. The collar is ciliated and generates the current that moves food into the mouth.
1d. >The secondary mouths open when food is present. You can demonstrate this with amino acid solutions (eg. glutathion, proline, or Artemia extract. <
1e. >Feed living Artemia nauplii (see Artemia chapter for culturing methods) to a small transparent Cassiopeia in a culture dish on the stage of your dissecting microscope. Watch the capture of the shrimps and their transport through the brachial canals to the stomach. <
Brachial Canals
A brachial canal is visible just under the oral surface of each arm. Find the canal on one of the arms and follow it to the center of the oral surface. The canal is very close to the oral surface. Its position near the surface is consistent with its being formed by closing the brachial groove by an overgrowing tissue layer. Note that its branching pattern reflects that of the arm itself. The secondary mouths open into it the branches. Note that there is no primary mouth at the center of the animal at the point where all the brachial canals converge.
" You can improve your view of the oral arm and its brachial canal by removing the oral collars from the secondary mouths along the length of one arm and on the bases of two adjacent arms. This is easily done with a pair of fine scissors. Try to remove only the collars and avoid cutting into the brachial canals just below the surface. After removing the collars, place the animal in a glass-bottomed dish and view it with transmitted light. Look again at the brachial canals and the secondary mouths. The branching pattern of the canals, and arms too for that matter, is much easier to see. The junctions of the brachial canals clearly reveal which pairs of arms belong together and are derived by branching from a single ancestral oral arm.
1f. >Inject toluidine blue/seawater solution into a brachial canal. It will spread throughout the arm and demonstrate dramatically the branching pattern of the brachial canal. Some of it may leak from the secondary mouths along the canal and some may enter the stomach. This injection is best accomplished as follows.
Use fine scissors to remove the collar from one of the secondary mouths close to the center of the animal. Load a hypodermic syringe with toluidine blue/ seawater solution (see Supplies chapter). Insert the needle into the exposed secondary mouth, at a low angle, so it enters the underlying brachial canal and moves well into it. The needle should point toward the peripheral tip of the arm. Do not push the needle into the surrounding mesoglea.
Depress the plunger of the syringe to inject the dye into the brachial canal. Inject enough dye so that it reaches the tip of the arm. Some of the dye will escape through the secondary mouths. You can blow this superfluous dye away with a few squirts of water from a pipet. Remove the needle from the mouth and reinsert it so it points centrally. Inject more dye. This time the dye will tend to move centrally and will reveal the connections of the brachial canals with each other and with the stomach. Some of the dye will enter the manubrial canals and the stomach.<
Vesicular Appendages
In addition to the mouths, there are numerous flattened, club-shaped vesicular appendages that also open into the brachial canals. The number, size, and color of these appendages varies with sex and age. Their function is not well understood. Each is a hollow, blind sac whose lumen is continuous with that of the brachial canal to which it is attached. The vesicular appendages are flat, elongate, and of many sizes. Some are distally bifurcate. The interior of each large vesicle may be purple, sometimes colorless, bluish, or bluegreen. The inner epithelium of the appendages is gastrodermis and bears numerous small, nearly spherical nematocytes and zooxanthellae that are easily seen in a wet mount.
The vesicles are used in trapping small crustaceans such as copepods and amphipods which are then stung to death. They also provide surface area for the deployment of zooxanthellae in the sunlight. They may also help camouflage the jelly in grass beds. Small clusters of nematocysts on their outer surface break easily from the vesicle and presumably discourage predators. Disturbed jellies fill the nearby water with these nematocysts which may then sting nearby predators or even swimmers.
1g. >Remove a pigmented vesicle from the specimen, open it and make a wetmount of it. Examine it with the compound microscope and look for spherical zooxanthellae and the small, nearly spherical nematocysts of the nematocytes. If your specimen is alive, a little weak acetic acid added under the coverslip will result in discharge of the nematocysts. What color are the zooxanthellae in the living material? <
Manubrium
The eight arms are joined at their bases to the thick, but short, vertical manubrium that connects with the bell (Fig 1). Remember that the arms are in pairs. The two brachial canals from the two arms of a pair join each other upon reaching the manubrium and form a larger manubrial canal that runs vertically in the manubrium, perpendicular to the brachial canals, to enter the stomach (Fig 2).
The stomach is a central space located at the aboral end of the manubrium in the bell. There are four columnar canals, one for each of the original four oral arms. Use your minuten nadel to show the existence of a manubrial canal for each pair of arms and the continuity of the brachial canals and manubrial canals.
Note that a short vestigial canal runs to the center of the oral surface from the point of confluence of the brachial canals (Fig 2). There are four of these little canals and they join at the point where the central mouth would be if there were one. In other scyphozoans the primary mouth would be here. Demonstrate for yourself that there is no mouth in this position in Cassiopeia.
Rhopalia
The margin of the bell is downturned (turned toward the oral side) and is slightly scalloped. Evenly spaced around the margin are 16 (usually) sense organs, or rhopalia (Figs 1, 2, 7-48). Each of these contains a tiny, but easily seen, spherical statocyst, flanked by a pair of small, flaplike rhopalial lappets.
Each rhopalium consists of the two lappets, a deep sensory niche, and an elongate, fingerlike lithostyle. The latter is hollow and contains an extension of the nearby radial canal. The extension is the rhopaliar canal. The statocyst is located at the distal end of the lithostyle and contains a calcareous otolith. There is a yellowish brown pigment spot on the aboral side of the rhopalium aborad the otolith.
Internal Anatomy of Transparent Specimens
Stomach
The large stomach can be seen clearly with the transmitted light of your dissecting microscope (Fig 2). It has four convex sides and looks like a rounded square or flattened circle in oral view. It is located in the center of the bell at the level where the manubrium joins the oral surface of the bell.
Radial Canals
Thirty-two radial canals radiate from the periphery of the stomach to the margin of the bell. There is no ring canal. Sixteen of the radial canals end at sensory rhopalia and are rhopaliar canals. Another 16 lie between these and are adrhopaliar. All 32 radial canals can be seen crossing the oral surface of the bell but are better seen from the aboral surface where their junctions with the stomach are most evident.
Turn the jelly over so you can see the canals through the aboral surface. Viewed aborally, the adrhopaliar canals are darker (in life) than the rhopaliar and can be seen without a lens. Adjacent canals are connected with each other by an extensive system of branches and anastomoses. Bidirectional ciliary currents move material through the radial canals. An outbound current moves along the roof of the canal while an inbound current moves along its floor.
Subgenital Pits
Turn the jelly back over so its oral surface is facing you. Between you and the stomach are four ectodermal pouches, the subgenital pits (Fig 2, 7-47, = septal funnels). These are flask-shaped invaginations into the mesoglea of the manubrium. They lie on the interradial axes in the axils between adjacent oral arms (Fig 7-46C). Each opens to the sea on the side of the manubrium by a pore on an interradial axis (Fig 1).
Use a teasing needle (or, preferably, a blunt probe if the pore is large enough to accept it) to find one of these pores and demonstrate its continuity with the pit. Carefully probe the interior of the pit and determine its extent. Convince yourself that the pits are NOT continuous with the stomach above them.
In larger individuals the central ends of the four pits coalesce with each other to form a single continuous space called the porticus in the interior of the manubrium, oral to the stomach. Use the teasing needle to determine if your specimen has four separate subgenital pits or a single porticus with four openings to the exterior.
1h. >If you wish, inject dye (of a color different than that used for the brachial canals) into one of the subgenital pits. The spread of the dye will reveal the extent of the pit or the porticus, as the case may be. <
1i. >Inject dye, via a manubrial canal, into the stomach and watch it move into the radial canals. If the animal is alive, the flow of dye will be assisted by ciliary currents. If you have a preserved specimen, distribution of the dye will depend entirely on the pressure you apply with the syringe. <
The anatomy of the partitions between the subgenital pits and the stomach is complex. Keep in mind that the pits are oral to the stomach and are interradial. Where the stomach overlies the four pits its floor (oral wall) is thin and folded with radial ribs. As a consequence, the roof of the subgenital pit is folded also.
Gonads
The four gonads lie in the gastrodermis of the four areas of folded stomach floor (Fig 2, 3, 7-46C). They are presumably supplied with oxygen by water flowing through the subgenital pits. Each gonad forms a band across the widest part of the folded area in the floor of the stomach. Although the gonads appear to be part of the subgenital pits, they are actually part of the oral wall of the stomach.
Gastric Filaments
The central ends of the stomach folds bear tiny gastric filaments that protrude into the lumen of the stomach (Fig 2,3, 7-46C). There are four bands of gastric filaments, one central to each gonad on the folded area. The epithelium of the filaments is gastrodermis and bears cnidocytes, cilia, and secretory cells. The secretory cells synthesize and release digestive enzymes into the stomach.
Internal Anatomy of Opaque Specimens
If you have a larger specimen or a preserved specimen, it will be opaque and you will not be able to see the internal structures without making a simple dissection. Even if you have a transparent specimen, you may want to make this dissection to reveal more detail and to help you understand the spatial relationships between the internal structures.
" To begin, look at the animal from the side so you can clearly see the place where the manubrium joins the bell. With scissors or a sharp scalpel make a transverse section across the aboral end of the manubrium as close to the bell as possible. Try to avoid cutting the tissues in the interior of the manubrium. Separate the bell from the manubrium and oral arms. Keep both in fluid. Examine the center of the oral surface of the bell. "
Stomach
The section should pass through the stomach. The circle you see is the roof of the stomach (Fig 2, 3, 7-46C). Set the bell aside (but keep it wet) and study the aboral surface of the manubrium. Here you will see the floor (oral wall) of the stomach, which is also circular.
Subgenital Pits
Lying on the four interradii are the four pouchlike subgenital pits (septal funnels, Fig 2,3, 7-46C). They are invaginations of the surface epidermis and each connects with the outside by a short canal and axillary pore located on the interradius in the axil between two oral arms. The canals may (probably) have been sectioned when you made the transverse cut across the manubrium.
Insert a probe or teasing needle into one of the axillary pores and demonstrate its continuity with a subgenital pit. The space between and above the four pits is the stomach. Each pit has a thin folded (plaited) roof that is also the floor of the stomach above (aboral to) it (Fig 3). The folds are oriented radially.
Gonads
A band of white tissue extends across the widest part of each of the four folded areas. These are the four gonads, either ovaries or testes (Fig 2, 3, 7-46C). The long axis of the gonad is perpendicular to that of the folds.
Gastric Filaments
Immediately central to the gonad there is a narrow band of tiny papillae that extend aborally into the lumen of the stomach. These are the gastric filaments.
Find the four openings of the manubrial canals into the floor of the stomach (Fig 2, 3, 7-46C). These are located on the perradii between adjacent subgenital pits near the periphery of the stomach.
References
Bigelow, R. P. 1900. The anatomy and development of Cassiopeia xamachana. Boston Soc. Nat. Hist. Mem. 5:191-236, pls 31-38.
Hyman, L. H. 1940. The Invertebrates: Protozoa through Ctenophora. McGraw Hill, New York. 726 p.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Supplies
I dissecting microscope
1% acetic acid
1/s living or preserved Cassiopeia, preferably small.
1 % toluidine blue / seawater solution
hypodermic syringe
1 glass culture dishes