Invertebrate Anatomy OnLine
Bugula neritina ©
Cheilostome Bryozoan
28may2007
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises, a glossary, and chapters on supplies and laboratory techniques are also available at this site. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Lophophorata SP, Bryozoa P, Gymnolaemata C, Eurystomata SO, Cheilostomata O, Anasca sO, Bugulidae F (Fig 25-35, 9-26)
Lophophorata SP
Lophophorata includes three taxa, Phoronida, Bryozoa, and Brachiopoda, sharing several morphological characteristics. Some zoologists include Kamptozoa in this group. The first three taxa possess a funnel-shaped anterior ring of ciliated tentacles known as a lophophore (Fig 25-2, 25-25A). The lophophore surrounds the mouth and is an upstream collecting system for suspension feeding. Its tentacles are hollow, with extensions of a coelomic space thought to be a mesocoel. The gut is U-shaped with the anterior mouth at the center of the lophophore. The anus is also anterior, but is dorsal to the mouth, outside the ring of the lophophore (Fig 25-2A).
The lophophoral tentacles bear two types of cilia. Frontal cilia on the inside face of the tentacles extend into the interior of the lophophore, whereas lateral cilia on the sides of the tentacles extend into the gap between adjacent tentacles. The mouth is at the bottom (apex) of the lophophore funnel and is encircled by the ring of tentacles. The feeding current is generated by the lateral cilia. In lophophorates and many filter feeding deuterostomes (but not Kamptozoa) water enters the open end of the lophophore, moves toward the mouth, and then exits laterally between the tentacles (Fig 25-25A). Food particles are captured on the upstream side of the tentacles and transported to the mouth by the frontal cilia. In cross section the lophophore may be horseshoe-shaped or circular (Fig 25-4).
Lophophorates inhabit a secreted enclosure, tube, shell, or zooecium, that may be organic or mineral. The body is divided into two parts, the mesosome and metasome, each with a coelomic space. The small mesosome is the region of the lophophore and the much larger metasome is the trunk and accounts for most of the body. The tiny anterior epistome is sometimes considered to be a third body region homologous to the protosome of early deuterostomes. Lophophorates are suspension feeders and most are marine but some occur in freshwater.
Bryozoa P
Bryozoa (moss animals) are derived, almost always colonial, lophophorates. Colonies are composed of individuals, or zooids, which are usually less than 0.5 mm in length. Each zooid inhabits a secreted box, the zooecium, into which is can retract. Because of their small size, hemal, excretory, and respiratory systems are absent.
Colonies are usually attached to firm substrata and may be encrusting layers a single zooid thick, or foliose and leaflike, or fruticose (bushy) and branching (Fig 25-15). Some, such asZoobotryon, are stoloniferous and consist of tangles of long slender stolons (which are zooids). Bryozoans often resemble seaweeds (or mosses) with which they are frequently confused by the public. Alone among lophophorates, Bryozoa includes freshwater representatives.
The lophophore may be circular or horseshoe shaped in cross section. A small epistome may be present anterior to the mouth. The zooecium can be composed of chitin, calcium carbonate, or gelatinous organic compounds. Many bryozoans are polymorphic with zooids specialized for different functions.
Gymnolaemata C
Gymnolaemate bryozoans are almost entirely marine and have circular lophophores. There is no epistome and no body wall musculature. These are considered to be derived characteristics. Colonies are polymorphic with specialization of zooids.
Cheilostomata O
Cheliostomata is the largest extant bryozoan taxon. Cheilostomes have box-like calcareous zooecia with an operculate orifice (Bugula has lost the operculum). They may be polymorphic with ovicells, vibracula, kenozooids, and/or avicularia in addition to autozooids. Eggs are usually brooded (in the ovicells).
Anasca sO
The frontal wall of the anascan zooecium is membranous and flexible. The lophophore is protruded when parietal muscles pull this wall inward and compress the zooid (Fig 25-24D,E).
Laboratory Specimens
This exercise is written for any species of the common and widespread marine genus Bugula. At least one species of this genus should be available on any North American coast. Living and preserved materials are available from biological supply companies. A purple species, Bugula neritina is abundant on the southeastern coast, Bugula fulva, which is yellowish, is common in the northeast and Bugula pacifica is widespread on the Pacific coast. There are several other species as well. Bugula colonies are erect, branching, and bushy (fruticose).
The study of bryozoans should be conducted using freshly collected living material whenever possible. Commercially prepared slides or preserved material can be used if necessary. Living material should be in seawater in a small dish on the stage of a dissecting microscope.
External Anatomy
Colony Morphology
Place a small piece of a colony of Bugula in a finger bowl of seawater (if your specimen is alive) or tap water (if preserved). Place the dish or prepared slide (if that is what you are using) on the stage of the dissecting microscope and begin your observations with low power.
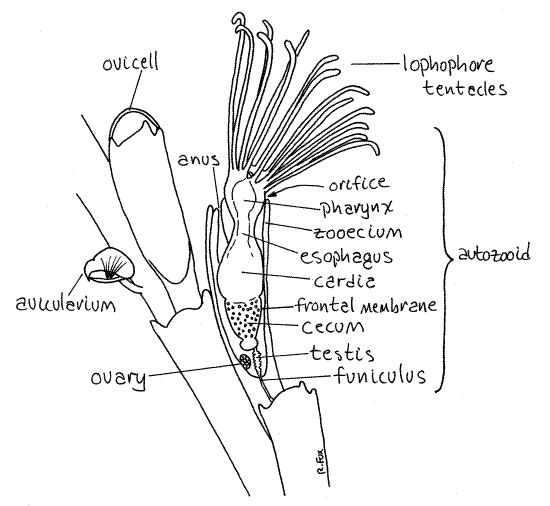
The colony is composed of branching stems (Fig 25-16A). The central stem of the bush is attached firmly to some hard substratum (in life). The colony is made of large numbers of typical feeding zooids called autozooids, as well as a variety of specialized zooids, known as heterozooids, modified for functions other than feeding. Three types of heterozooids occur inBugula colonies. The stems, and hence the framework, of the colony consists of double rows of autozooids. The heterozooids are much less conspicuous and are associated with some of the autozooids (Fig 1, 25-21A, 25-27D)
Note the arrangement of the autozooids in the colony. The colony is composed entirely of zooids arranged in double branching rows in a bush-like pattern. Find some of the elongate autozooids in a branch and be sure you can recognize them (Fig 1, 25-16A, 25-21A). They look like joints or segments arranged side by side and end to end in two staggered rows. Increase the magnification so you can see that each stem or branch is composed of two rows of zooids. The zooids are aligned end to end with other zooids in their row and staggered side by side with zooids in the adjacent row.
Zooecium
Each bryozoan zooid secretes and inhabits a nonliving zooecium (zoo = animal, oikos = house, Fig 1). The zooecium is secreted by the epidermis of the zooid’s body wall and In Bugulaconsists of lightly calcified chitin. They are elongate and transparent. A zooid consists of its body wall (zooecium, epidermis and mesothelium) and known as the cystid, and its more internal parts (lophophore, muscles, gonads and gut), known as the polypide.
One wall of the zooecium of anascan autozooids is thin and uncalcified. This wall is the frontal membrane (Fig 25-21A, 25-24D,E). The transparency of the frontal membrane renders the internal structures visible. The frontal membrane of Bugula has the shape of a deep "V" or "U" and covers one side of the zooecium (Fig 1). Look carefully at the surface of some of the autozooids near the tips of the branches and find the long U-shaped frontal membrane extending almost the entire length of the zooecium (Fig 1). You may have to turn the branch over to see the membrane. The frontal membranes of all the zooids of a given branch will be on the same side of the branch.
Movement of the frontal membrane inward puts the coelomic fluid under pressure and everts the feeding tentacles, or lophophore. The lophophore is retracted by special retractor muscles (Fig 25-24D,E). The zooid inhabits the interior of the zooecium but the feeding tentacles can be extended from its open end. The opening through which the lophophore is extended is the orifice. The orifice is at the distal end of the zooecium and in Bugula lacks an operculum.
Zooids
Lophophore
The anterior end of the zooid can be everted from the orifice and you should be able to find some individuals in which this has happened. The everted portion of the zooid is theintrovert, consisting of the lophophore and anterior end of the metasome. The lophophore consists of a circular ridge of tissue around the mouth and a ring of ciliated tentacles arising from the ridge. When extended, the tentacles of the lophophore are graceful and conspicuous (Fig 1, 25-17). They are arranged in a cone-shaped funnel with the mouth at its apex (narrow end). The lophophore is a filter feeding device.
Look for a zooid whose lophophore is extended. The inner surfaces and sides of the tentacles are ciliated but the outer surface is not. The cilia on the two sides of each tentacle are thelateral cilia. They generate the current that moves water and food particles in the open end of the lophophore and then out between the tentacles. The cilia on the inner edge of the tentacle are the frontal cilia. Their responsibility is to move captured food toward the mouth Food particles are retained inside the cone and then moved to the mouth by the frontal cilia.
If your colony is alive, find an extended lophophore and watch it with high power of the dissecting microscope. Watch suspended particles in the water near the lophophore and note how they move with respect to the lophophore. Do the particles enter by passing between the tentacles or do they come in through the open base of the funnel and exit between the tentacles? Watch for motion of the tentacles, especially the characteristic "flicking" of bryozoan tentacles. What effect does the flicking have on particles?
Heterozooids
Look for heterozooids with high power of the dissecting microscope. Many autozooids bear hemispherical ovicells, or ooecia, at their distal, or free, ends (Fig 1, 25-27D). Ooecia are highly modified heterozooids which serve as brood chambers for the adjacent autozooid. A single egg is brooded in each ooecium. Look for embryos in the ooecia.
Most Bugula species have avicularia (but Bugula neritina does not). Find some on your specimen (unless you have B. neritina). These are defensive heterozooids shaped like the head of a diminutive raptorial bird (Fig 1, 25-21A,B). Avicularia are specialized for discouraging predation and settling by the larvae of fouling organisms. They are attached to the sides of the autozooids and have a pair of mandibles to pinch predators or larvae. Note the large bulbous "cranium" with fixed upper and movable lower mandibles. The cranium is the zooecium of the heterozooid whereas the movable lower mandible is its operculum. Try to find some open and some closed avicularia.
The youngest and most transparent zooids are at and near the free tips of the branches. The stems and bases of the branches consist largely of another type of heterozooid known askenozooids. In Bugula the kenozooids are simply the empty zooecia of dead autozooids but, although dead, they continue to be important structurally in anchoring the colony to the substratum. In some bryozoans, such as Zoobotryon, kenozooids are living but lack most of the organs found in the autozooids and may appear to be empty zooecia.
Figure 1. A small piece of a Bugula pacifica colony from Coos Bay, Oregon. Bugula65La.gif
Wetmount
Colony Morphology and Zooecia
With your fine forceps tear off two or three small branches of the colony and place them in a drop of isotonic magnesium chloride (if alive) or tapwater (if preserved) on a glass slide. Make sure the frontal membrane is facing up, toward you. Carefully apply a coverslip with wax feet and observe the preparation with 100X of the compound microscope. (Alternatively, you may make do with a commercially prepared slide if necessary).
Look again at the stems and branches and review their makeup. Find the two side by side rows of autozooids making up each branch. The zooids are probably fouled with a variety of organisms often including filamentous algae, vorticellid ciliates, and sessile diatoms. The older zooids at the base of the branches are the most heavily fouled whereas those at the tips, being younger, have had less time for colonists to settle and are less fouled.
Focus your attention on some young, unfouled autozooids near the tip of a branch. Do not pick zooids at the very end of the branches as these very young individuals may not be completely formed yet.
Locate the proximal and distal ends of a zooid (Fig 1). The proximal end is the apex of the V-shaped zooecium and is narrower. The orifice, through which the lophophore is extended, is at the distal end. Most zooecia have spines at the distal corners, beside the orifice. The orifice, remember, is at the wide end of the V-shaped zooecium. Find the frontal membrane and look for avicularia. Look for ooecia (oo = egg, oikos = house).
Find a zooid with the lophophore extended. The lophophore is a cone, or funnel, of hollow, ciliated tentacles with the mouth at its apex. Find another zooid in which the lophophore is retracted. The distal end of this zooecium will be filled with tightly packed retracted tentacles.
Internal Anatomy
On your wetmount find several autozooids with extended lophophores for a study of internal anatomy. Internal organs are best studied using several zooids as you probably will not be able to see all structures in any single individual. The thin and much reduced body wall lies inside and against the zooecium. It will not be apparent in your preparations. The interior space of the zooid is the trunk coelom (= metacoel), in which lie most of the animal's organs are found. The anterior end of the zooid is the mesosome and bears the lophophore. It contains the lophophore coelom (= mesocoel).
Digestive System
The largest and most obvious organ system is the digestive system. It is U-shaped with both mouth and anus at the anterior end of the zooid. The mouth is at the apex of the lophophore and is at the center of the ring of lophophoral tentacles. The mouth opens into a large muscular pharynx (Fig 1, 25-17). If your specimen is alive, you may see the pharynx dilate occasionally.
The pharynx leads to a large, usually brown esophagus. The large three-part stomach follows the esophagus and occupies the bottom of the loop of the U-shaped gut. The stomach consists of the cardia on the descending limb of the “U”, the cecum at the bottom of the “U” and the pylorus at the beginning of the ascending limb of the “U” (Fig 25-17). The intestineextends anteriorly from the pylorus to the anus situated on the dorsal midline but outside the lophophore. The pharynx, esophagus, and cardia are the descending limb of the “U” whereas the pylorus and intestine are the ascending limb. The walls of the cecum usually contain numerous tiny brown inclusions which give it a dark brown color. These inclusions accumulate and increase as the zooid ages. They are thought to contain wastes and will eventually be incorporated into a brown body for disposal (see below).
Funicular System
A funiculus composed of short, transparent, cellular funicular cords extends from the proximal end of the cecum to the posterior end of the zooid (Fig 25-17, 25-23B). The cords are in contact, across pores in the zooecium, with similar cords in other zooids (Fig 2523-E). The funicular system is an interzooid transport system.
Reproductive System
The testis surrounds the funiculus and may contain visibly mobile sperm (Fig 1, 25-17). The spherical ovary is attached to the inner wall of the zooecium.
Brown Bodies
Some zooids may contain dark brown spheres, the brown bodies. Each brown body is the remains of an obsolescent zooid polypide that has been reabsorbed by the colony (via the funiculus) and replaced by a new polypide, which is the current inhabitant of the zooecium (Fig 25-23F). Some kenozooids may contain brown bodies remaining from the original zooid but, of course, they have no active zooid currently present. Look for brown bodies in the autozooids.
Excretory and Respiratory Systems
Bryozoans have no special respiratory or excretory organs, i.e. no gills or nephridia. Gas exchange and nitrogen excretion (as ammonia) are accomplished over the general body surface, especially epithelium of the lophophore.
Hemal System
As tiny animals individual zooids have no need for an intrazooidal fluid transport system. The distance from source to sink of both gasses and nutrient is less than 1mm and simple diffusion is adequate for transport over such short distances. There is no hemal system although the ciliated mesothelium may assist simple diffusion in transporting materials within the zooid.
In contrast, the entire colony can be much larger, with distances far too great to be traversed by diffusion alone. The funicular system is an interzooidal transport system responsible for transport over distances too great to be efficiently accomplished by diffusion (Fig 25-17).
Nervous System
The nervous system cannot be studied in these preparations.
References
Bullough WS . 1958. Practical Invertebrate Anatomy (2 nd ed). MacMillan, London. 483p.
Rogick MD. 1950. Bugula, pp. 267-269 in Brown FA. (ed) Selected Invertebrate Types. Wiley, New York. 597p.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Supplies
Dissecting microscope
Compound microscope
Living (or preserved) Bugula colony or prepared slide
Small culture dish
Seawater
Magnesium chloride solution
Slides, coverslips