Invertebrate Anatomy OnLine
Laboratory Techniques
©
30may2007
Copyright 2001 by
Richard Fox
Lander
University
Preface
This is a chapter from
Invertebrate Anatomy OnLine
, an Internet
laboratory manual for courses in Invertebrate Zoology.
Additional exercises can be accessed by clicking on the links to the
left. Terminology and phylogeny used in these
exercises correspond to usage in the Invertebrate Zoology textbook by
Ruppert, Fox, and Barnes (2004).
1.
Removing Microorganisms from Culture Jars
Use a
plastic or glass Pasteur pipet with a bulb.
Squeeze the air from the bulb BEFORE immersing the tip of the pipet into
the culture jar. Position the tip appropriately.
For example, if you are looking for bottom
dwellers such as Amoeba, place the tip at the bottom.
If you seek swimmers, such as
Euglena or Paramecium, then the tip should be
above the bottom. Release the pressure on the
bulb slightly so a few drops (only) of liquid are
drawn into the tip of the pipet. Transfer these
drops to a microscope slide.
2.
Preparation of a Wetmount
Place one or
two drops of liquid in the center of a clean glass microscope slide.
Add stain or other materials as appropriate and
mix thoroughly with a teasing needle. Do not add
more than a total of 2-3 drops of liquid.
Carefully lower a clean coverslip onto
the liquid in such a way that no air bubbles are trapped beneath the
coverslip. This is best accomplished by placing
one edge of the coverslip on the slide touching the liquid.
Support the opposite edge of the coverslip with
a teasing needle or forceps and slowly lower it to the surface of the
slide.
Use # 1½ or # 2 square 22 mm
coverslips for ordinary microscopic examination.
Use # 0 coverslips if you plan to use oil immersion.
These are thinner and much easier to break so you must be more careful
with them.
It will probably be necessary to
adjust the amount of liquid on the slide. There
should be no liquid outside the coverslip but there should be enough
under the coverslip to support it so it does not stick tenaciously to
the slide and crush or distort organisms. If you
have too much liquid, use a tissue to absorb the excess.
If you do not have enough, use a pipet to place a drop or two
on the slide next to the edge of the coverslip.
The liquid will run beneath the coverslip if it is needed.
If it is not needed, you will have to remove it
with tissue.
Excess liquid on the slide will run
off the slide onto the stage where it will form a thin film between the
slide and stage. This film acts like glue and
its presence makes it impossible to move the slide smoothly across the
stage and almost impossible to move it at all.
It may also damage the microscope, especially if it is a salt solution.
3.
Simple Squeeze Preparations
It is
sometimes desirable to use the weight of the coverslip to squeeze an
organism slightly. This may be done to
immobilize the specimen or to make it a little thinner so its internal
parts are easier to see.
The simplest way to accomplish this is
by removing some of the liquid from under the coverslip.
While watching the organism through the eyepiece, touch a piece
of absorbent tissue paper to one edge of the coverslip.
Capillarity will wick the liquid from beneath the coverslip into the
paper. Remove the paper when you have withdrawn
as much liquid as desired.
More complex supports are described below.
4. Supported and
Adjustable Squeeze Preparations
A more elaborate method of
making squeeze preparations employs wax feet on the corners of the
coverslip and gives you control over the pressure applied to the
organism. Dig each corner of a coverslip into
the surface of a piece of beeswax so that a tiny piece of wax adheres to
the glass. This is easier if the beeswax is
softened somewhat. Hold the piece of wax in your
hand to warm it or, better still, simply store it in a shirt pocket next
to your body when not in use. Remember to remove
it before you leave the lab. The four pieces of wax are referred to as
"feet".
Place the coverslip, with its feet down, over the
liquid on the slide and lower it as usual. When
the coverslip is in place, use the handle of a teasing needle or
applicator stick to push gently, directly downward, on the glass above
each foot. This will flatten the foot and move
the coverslip closer to the slide where it will trap and squeeze
organisms.
The degree of squeezing depends on the pressure
applied. Once squeezed, the process cannot be
reversed.
5. Support of
Coverslip with Sand
It is sometimes necessary to
support the coverslip so it does not crush or distort delicate organisms
beneath it. The wax feet described earlier can
be used for this purpose and have the advantage of being adjustable (in
one direction) but they are cumbersome to use and time-consuming to
prepare. Instead, many microscopists use 5-10
grains of fine beach sand scattered in the fluid on the slide before the
coverslip is applied. The sand grains support
the coverslip and protect the organisms.
With this technique there is no possibility of
squeezing the organism should that become desirable.
Supported wholemounts may be too thick for use with the high dry
objective and will certainly be too thick for oil immersion.
6. Compound Microscope Light Adjustment
When using the compound microscope, the light
intensity must be carefully adjusted. There is a
tendency among beginning students to use too much light.
Transparent objects, such as Amoeba, will be invisible
if the light intensity is too high.
In general, light intensity should be adjusted using
the iris diaphragm, not the electrical rheostat control.
Set the rheostat control at about 75% of maximum and leave it
there. Use the iris diaphragm to adjust the
light intensity. Sometimes it will be necessary
to use the rheostat, as for example, when using the scanning lens.
7. Initial Focus
Finding the correct level at
which to focus is difficult when most of the material under the
coverslip is water. In the absence of a
conspicuous object, there is nothing on which to focus.
Use the edge of the coverslip as an easily-found "object" at the correct
level and focus on it first, then look for the small objects you are
really interested in, secure in the knowledge that you are focused at
about the right level.
8. Systematic Scan
Finding small and/or scarce
organisms on a slide is most efficient if done systematically.
First, be sure the light is adjusted properly
and the microscope is focused at the correct height (i.e. between the
coverslip and the slide). Organisms are
difficult or impossible to see if the light is too strong.
Using the scanning lens, find one corner of the
coverslip, say the upper left. Move the slide so
you follow the upper edge of the coverslip to the upper right corner.
Move down a distance equivalent to a little less
than the diameter of one field and then move back to the left side of
the coverslip. Your new field should just
overlap the already scanned strip next to the edge of the coverslip.
Be sure there is no unexamined space between the
two strips. Continue moving back and forth
across the slide, neglecting no part of it, until you find what you are
looking for or you reach the lower right corner.
9. Alteration of
Wetmount Without Removing Coverslip
It is frequently desirable to
change the liquid below the coverslip of a wetmount without removing the
coverslip. This is easily accomplished and can
(should) be done while you observe the organism.
To accomplish such a change, place a drop of the new
fluid on the slide next to and touching one edge of the coverslip.
Touch the edge of a piece of absorbent tissue
paper to the opposite edge of the coverslip so it is
in contact with the old liquid beneath the coverslip.
The old fluid will be absorbed by the paper and removed from the
wetmount. The new fluid will move under the
coverslip to replace the old as it is removed.
10. Optical
Sections
When observing
organisms with the compound microscope, keep the fine focus in nearly
continual motion, focusing up and down through the object.
Each position of the fine focus gives you a
crisp focused image of one level of the object, much as if you had
sliced, or sectioned, the object at that level.
The resulting view is known as an optical section.
Other levels are out of focus and cannot be seen clearly.
To get an accurate impression of the entire organism you must
take optical sections at all levels by continual refocusing up and down.
11.
Immobilization of Active Organisms
Rotifers and many protozoans, especially ciliates,
swim so rapidly that they cannot be observed alive unless immobilized.
This is accomplished by using a solution
containing long polymers that entangle and impede the organisms.
Traditionally a 10% solution of methyl cellulose
is used for this but other products are now available.
Your laboratory may be supplied with vials of homemade methyl cellulose
solution or with commercially prepared products, such as Detain or
Protoslo. Prepare the wetmount as usual but use
a mixture of culture and polymer. The more
polymer, the slower the organisms.
Squeezing the preparation slightly by removing a
little fluid may also immobilize swimming cells or organisms. Another
method is to place a whisp of cotton on the slide and add a drop of
culture and then the coverslip. The crossed
cotton fibers create small chambers or cells in which organisms may be
trapped thus limiting their motion.
12. Oil Immersion
To use the oil immersion
lens, focus on the object of interest with the high dry lens and be sure
the object is centered in the middle of the field.
Rotate the nosepiece halfway to the oil immersion
(100X) objective. Place a drop of immersion oil
on the slide directly atop the center of the hole in
the stage. Slowly move the nosepiece around
until the oil immersion lens clicks into position.
Be sure you rotate the nosepiece directly to the oil
immersion lens and not to the high dry. If you
do the latter, you will get oil on the high dry lens.
Should this happen, clean it immediately with lens paper (only) and no
harm will be done but do not allow oil to remain on this lens.
Look through the eyepiece and focus on the object
very carefully using fine adjustment only. When
finished with the oil immersion lens clean it carefully with lens paper.
If you are using a prepared slide, clean it
also. Coverslips contaminated with oil are
usually not reused and should be discarded. Oil
immersion is usually used without a coverslip or with a # 0 coverslip.
13. Cleaning
Lenses
You should keep the lenses of
your microscopes clean. Make it a point to clean
the ocular lenses (eyepieces) of your compound and dissecting
microscopes at the start of each laboratory session.
The objectives, however, rarely need cleaning unless they have been
accidentally smeared with the liquid from a wetmount.
Use nothing but fresh, unused lens paper to clean lenses and discard it
after use.
If you notice specks or smears in your field of view,
it means at least one of the many lenses in the system needs cleaning.
You can identify the offending lens using a
simple protocol.
Look through the eyepiece so you can see the spots.
Move the slide. If the
spots move, they are on the slide, which should then be cleaned.
If the spots don't move with the slide, try rotating
the eyepieces one at a time. If the spots
rotate, they are on one of the lenses of that particular eyepiece.
In this case they are almost always on the outer
surface of the upper lens because it is the one exposed to oily
eyelashes, dust, and the occasional fingerprint.
If the dirt did not rotate, it must be somewhere
else. Raise or lower the condenser.
If the spots go out of focus they could be
either on the top of the substage lamp or the condenser lens.
Clean both.
If the contamination manifests itself as a blurred
image that cannot be corrected by focusing, the objective is probably
fouled. Moisten a piece of lens paper with water
and clean the outer surface of its lens.
14. Cleaning
Permanent Slides
Clean prepared slides by
wiping lightly with a piece of absorbent paper or tissue.
The lower surface of the slide can be rubbed vigorously but the
coverslip must be treated gently. Do not apply
pressure to the coverslip that would tend to move it (the coverslip)
with respect to the slide. The undetectable
microscopic movements resulting from such handling can damage the
object. This is especially true of sections,
which can be destroyed by enthusiastic rubbing.
15. Sealing
Temporary Slides
Mineral oil can be used to
seal the edges of wetmounts to prevent evaporation and increase the life
of the slide. This is especially useful for
wetmounts made with seawater or magnesium chloride since evaporation of
water increases the salinity under the coverslip, making conditions
intolerable for living organisms.
After preparing a wetmount as usual, place a
tiny drop of mineral oil beside the coverslip.
The oil will flow over the surface of the
exposed liquid at the edge of the coverslip and will retard evaporation.
Temporary slides can also be sealed with petroleum
jelly. Smear a thin film of jelly on the fleshy
part of the heel of your hand and scrape all four edges of a coverslip
across the jelly leaving a low ridge of jelly on each edge.
Use this coverslip to make a wetmount in the
usual fashion. The jelly ridges will form a seal
between the coverslip and the slide.
16. Visualization of Ciliary Currents
One of the important advantages of
using living, relaxed specimens is that cilia and flagella continue
to beat, making it possible to see the currents they generate.
Vizualization of ciliary currents usually requires the
addition of fine particles or soluble dyes to the area of
interest. The particles or dye should be suspended or dissolved in
liquid of the same density (and temperature) as that surrounding the
animal or else density currents will interfere with your study of
ciliary currents. The suspensions should be made using either
seawater or pondwater as appropriate and stored in small bottles for
use when needed. The density of the suspension can be adjusted with
salt (non-iodized) or Instant Ocean.
Ciliary fields and tracts are usually very specific with
regard to the particles they can transport. Particle size and
particle density are strongly related to their transportability by a
particular ciliary field. It is useful to have a collection of
suspensions of particles of different sizes and weights.
Carmine suspended in water (seawater or pondwater as
appropriate) is probably the best general-purpose tracer.
Chalk dust from the eraser trays of the classroom
chalkboard (becoming harder and harder to find as more and more
classrooms are equipped with whiteboards) can be suspended in water
and used to advantage. (Chalk dust deliberately scraped from a stick
of chalk is coarser, less homogenous, and less satisfactory than
that found in the chalk tray.)
Water-based latex house paint, which consists of tiny
latex beads in an aqueous carrier can also be used when very fine
particles are needed.
India ink contains colloidal carbon and has long been a
popular source of tiny particles. Its density must be adjusted for
use in seawater.
Homogenized milk, with its density adjusted (with salt),
can also be used, especially when fine particles are needed. As an
alternative, powdered milk can be reconstituted using seawater of
the appropriate salinity (density).
If available, fine volcanic ash suspended in water is a
useful tracer.
When tracing ciliary currents on surfaces, be sure the
surface is horizontal. If you do not then gravity currents may be
mistaken for ciliary currents.
Common household food coloring, being non-toxic and
inexpensive, is ideal for freshwater work but its density must be
adjusted for use in seawater or else it will simply float on the
surface of the water.
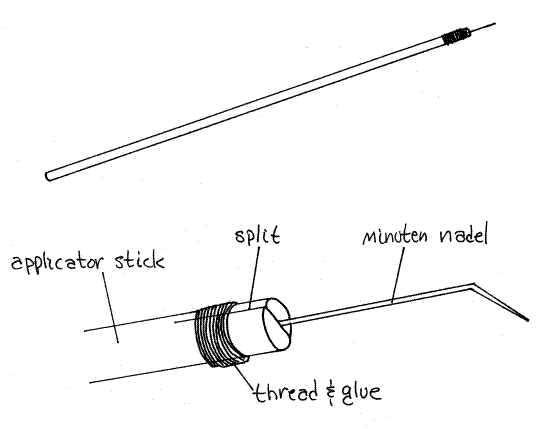
17. Micro Teasing Needles
Very fine needles, known by the German minuten
nadeln, or the English microneedles, mounted at the ends of
wooden applicator sticks (Fig 1) are essential for manipulation and
dissection of small animals and are easily prepared. They are not
indestructible and must be replaced periodically.
Split, with the grain if possible, 2-3 mm of the tip of
an applicator stick with a scalpel. Do this safely by placing the
stick on the surface of the table and pushing the blade of your
scalpel into the side of the end of the stick (not the end
of the end). Do not hold the stick in your hand while splitting it
and do not cut toward yourself.
Insert about 2 mm of the blunt end of a 0.2 mm
microneedle into the cleft, applying a drop of water-insoluble glue,
such as Duco Cement, to the end of the stick around the cleft and
base of the needle, and wrapping it tightly with sewing
thread. Twirl the end of the stick between your thumb and forefinger
to spread the glue over the thread, end of the stick, base of the
needle and into the cleft.
Make at least two miocroneedles, one with a straight tip
and the other bent at a 45 ° angle (Fig 1). Use two
different colors of thread, such as yellow for the straight needle
and blue (B for bent) for the bent tip, to color code your two
microneedles. This makes it easy to select the desired needlewithout
having to look closely at the tip.
Figure 1. A micro teasing needle made from a minuten
nadel and an applicator stick. lab5.gif
Stainless steel minuten nadeln (without
sticks, listed as minutien pins) are available from Fine
Science Tools (www.finescience.com). Be sure to order 0.2 mm needles
and not 0.1 mm. Be sure to get stainless rather than black
anodized. Applicator sticks are available from VWR Scientific
Products.
Be careful when working with these tiny
needles. Microneedles are tiny and are easily lost at the
workbench. Lost needles have a habit of ending up later sticking
into fingers or elbows.
The delicate needles are easily bent but are almost as
easily restored to their original condition. If the tip gets turned
over into a tiny hook, it can be straightened by pulling it between
the nail of your forefinger pinched tightly against the ball of the
thumb, or between the points of your forceps.
18. Modification of Microdissection
Forceps
Watchmaker's or microdissecting forceps are essential
for most of the dissections in this collection. They are available
from Fine Science Tools, Carolina Biological, and Ward's. They are
delicate tools that require careful handling and preparation.
The tips of new microdissecting forceps (especially
inexpensive ones) often require modification before they are
suitable for use. This is accomplished by grinding excess metal from
the tips with a sharpening stone such as a Ouachita or Arkansas
stone (available from Fine Science Tools or a hardware store).
Hold the blades of the forceps together lightly
so the opposing tips touch each other but do not press firmly
against each other. Look at them with the dissecting microscope. Do
they meet each other in perfect alignment or do they pass each other
or attempt to do so? Are the two tips the same length? Are the tips
as sharp as you would like? Perhaps they are too sharp?
You can correct the alignment and produce the shape you
want with the stone. Place the stone on the stage of the microscope
and focus on its upper surface. Place a few drops of light machine
oil on the stone. Hold the tips of the forceps lightly together and
slide the point back and forth over the surface of the stone against
the metal you want to remove. These stones will remove metal rapidly
so check the tips frequently to be sure you are not removing too
much. If the tips meet each other at a spot behind the tip, it will
help to bend the tips a little. Never try to remove metal from the
inside of the tips.
If one tip is longer than the other the forceps will be
unusable until corrected. Hold the tips together and rub the point
of the combined tips together on the stone. Check the lengths
frequently and stop grinding when the two are equal.
For storage push the tips into the end of a small cork
and keep them there when not being used.
19. Dissecting Pans
Standard dissecting pans available from biological
supply companies are too large to be used conveniently with most
invertebrates. Most dissections will be conducted on the stage of a
dissecting microscope and standard pans are too large to fit. An
assortment of small dissecting pans can be made from a variety of
metal containers.
Dissecting pan wax can be purchased from Wards Natural
Science Establishment. The wax is melted in a saucepan dedicated to
that purpose (it will be ruined for other uses) and the molten wax
poured to an appropriate depth into the chosen container.
The most useful sizes are those made from empty 56 g
anchovy tins, 105 g sardine or smoked oyster tins, and 100 g
kippered herring tins. The old-fashioned tins that require a can
opener are best but those with pull tops can also be used. Their
disadvantage is that they have a lip around the opening. Tuna fish
tins can also be used and the now-obsolete aluminum refrigerator ice
trays make excellent pans for long worms. They can still be found at
yard sales and flea markets.
The wax should be poured so there is ample freeboard
above the wax to more than accommodate the thickness of the specimen
and the fluid in which it will be immersed. Because it does not
rust, aluminum pans are preferable to steel
20. Incident and Transmitted Light
Dissecting microscopes are usually equipped with two
light sources. The incident light shines down on
the object from above the stage whereas transmitted
light illuminates the object from below and passes through it. The
two lights have different effects and different uses and are not
interchangable. In general, incident light should be used for opaque
objects and transmitted light for transparent objects. Avoid using
transmitted light on opaque objects as the contrast between the
bright light and the dark object makes it difficult to see the
features of the object.
21. Anesthetization
Most marine invertebrates can be anesthetized and
relaxed with isotonic magnesium chloride. The sensitivity of animals
to magnesium compounds is variable. Some species succumb rapidly,
almost immediately, whereas others resist its effects for long
periods. In general those species that succumb rapidly will recover
rapidly when returned to seawater but those that require long
periods to relax often will not recover. There is a tendency for
species with thick, impermeable cuticles or exoskeletons to resist
relaxation by magnesium chloride whereas those with thin, permeable
integuments are susceptible.
Magnesium chloride is usually used at a
concentration having the same tonicity as that of the animal's body
fluids. For marine invertebrates this is usually the same as that of
the surrounding seawater. This is referred to as isotonic magnesium
chloride throughout these exercises. For animals from undiluted
seawater this is about 33 ‰ (parts per thousand). Recipes are given
in the Supplies chapter.
A weak solution of non-denatured ethyl alcohol
(5-10%) in seawater (or tapwater for terrestrial or freshwater
species) rapidly relaxes some species that can resist magnesium
chloride for hours.
Magnesium chloride cannot be used to anesthetize
freshwater animals because concentrations high enough to be
effective are strongly hyperosmotic to the animal and cause its
cells to plasmolyze. Other anesthetics must be used instead but it
is sometimes very difficult, especially with freshwater or
terrestrial molluscs, to find an anesthetic that will relax the
organism in an expanded condition. Ethanol (5%), chloretone (0.2%),
carbonated water, chilling, and chloroform-saturated water are the
most widely used anesthetics for freshwater animals. For soft-bodied
animals the anesthetic should be made up in 0.75% saline solution to
avoid osmotic damage.
Most terrestrial arthropods can be anesthetized
with fumes of chloroform, ether. Carbon dioxide, either as a gas for
terrestrial animals or in solution as carbonated water (Club Soda,
Perrier) for freshwater animals is an excellent anesthetic.
22. Gradual Relaxation
Some marine animals such as the sea
hare, Aplysia, and most snails cannot be placed directly
into anesthetic. Aplysia will contract irregularly into an
unusable and incomprehensible lump of tissues and snails simply
withdraw into shell, close the operculum, and wait for things to get
better. The problem can sometimes be circumvented by the slow
addition of anesthetic to the water containing the animal.
Use a strong solution of anesthetic (e.g. 70 ‰)
magnesium chloride, 95% ethanol, or 5% chloretone) administered
dropwise via a siphon with a carefully adjusted screw
clamp. Alternatively, a strip of absorbent cloth or a string (known
as a drip string) can be used as a wick to drip anesthetic slowly
from the anesthetic container to the animal's into the container.
This process may take several hours, often overnight. Be very
careful that the animal is not disturbed until it is anesthetized
sufficiently that it cannot respond.
23. Preservative
Preserved material, if it must be used, should
always be ordered in one of the commercial holding fluids rather
than formalin. Prolonged exposure of human skin to formalin often
results in development of a dermal sensitivity that manifests itself
much like poison ivy with weeping pustules and intense
itching. Further, formaldehyde causes respiratory problems and is
now on the federal list of carcinogens.
The developed sensitivity to formalin was once an
occupational hazard of biologists, many of whom spent much of their
professional lives handling preserved animals. The sensitivity
develops as a result of prolonged exposure, with the amount of
exposure required varying with individuals. Once acquired, the
sensitivity remains for life and the response is triggered by
exposure to minute amounts of formalin and aggravated by other
chemicals. It is best to avoid all unnecessary exposure.
Fortunately it is now relatively easy to avoid
formalin. All supply companies offer preserved specimens in some
holding fluid other than formalin. This does not mean, however, that
no formalin is present. Formalin remains the best agent for the
initial fixation and hardening of tissues to be used for gross
dissection. Specimens are usually fixed in formalin first and then
transferred to some innocuous holding fluid that prevents
decomposition.
Some formalin inevitably remains in the
tissues. Its odor may be strong and unpleasant and there is more
than enough to initiate a reaction in sensitized individuals.
Formalin-sensitive people should wear rubber gloves when using
materials prepared in this manner. The teaching staff should remove
specimens from the holding fluid the day before the laboratory
session and place them in tap water to soak overnight. At the
beginning of the laboratory period the water should be drained and
replaced with fresh. Preserved specimens should be dissected
immersed in tapwater. Once opened, the body cavity of the specimen
should be rinsed gently with tapwater.
24.
Pinning
Most dissections of soft-bodied invertebrates require
that you pin the body wall aside to reveal the body cavity and its
viscera. In most instances # 1 insect pins are appropriate but for
very small or delicate animals # 000 may be preferable whereas for
larger specimens # 4 may be best. The pins should be stainless
steel, especially if they are used in seawater or magnesium
chloride. High quality stainless steel insect pins are available
from Fine Science Tools (See Supplies chapter).
Insert the pins at 45 ° angles through the
body wall and push them firmly into the wax of the dissecting
pan. Think ahead before you insert the first pin and arrange the
position of the specimen in the pan so it, the specimen, will be
conveniently located after all pins are in place. The animal should
be positioned so it will be visible with the dissecting microscope
and so none of it extends beyond the edges of the pan.
25. Care of Iridectomy Scissors
Your laboratory may provide you with iridectomy scissors
or you may be required to purchase a pair. In either case they
require careful attention to remain in working order. The blades of
these fine scissors are closed by the muscles of your fingers but
they are opened by the elastic recoil of their spring-like
handles. The blades are held together at an articulation by a pin,
as are the blades of any scissors. Whereas your fingers are capable
of supplying more than enough power to close the blades, the weaker
springs cannot open them again if there is any
resistance. Specifically, if rust accumulates in the articulation,
it will prevent the blades from opening and the scissors will be
useless.
After each session the scissors should be cleaned,
dried, and greased before storing. Wash the scissors with warm soapy
water. Dry with a soft cloth towel. Dip the scissors in acetone and
then dry again. Unfasten the handles and open the
articulation. Inspect the flat surfaces and scrape away any rust
deposits you find there. Apply a tiny gobbet of petroleum jelly to
the articulation and smear it over the surfaces. Close the scissors
and secure the handles again.
All your scissors would benefit from this treatment but
it is essential for iridectomy scissors.
26. Berlese Leaf Litter Extraction
The diverse and populous arthropod community of forest
floor leaf litter is easily sampled using a Berlese funnel. A few
handfuls of leaf litter are placed in a large funnel (about 20-25 cm
in diameter at the large end), oriented with its large opening
uppermost. A vial of water or alcohol (40% isopropanol or 80%
ethanol) is attached to the small, lower end of the funnel. A
75-watt light bulb is left burning for several days over the top of
the funnel. The heat from the lamp drives the animals downward in
the funnel until they drop into the vial from which they can be
recovered and studied.
The Berlese technique will yield a variety of
small soil arthropods, usually including many species of mites and
collembolans as well as some pseudoscorpions, spiders, millipedes,
centipedes, beetles, flies, ants, thrips, and others.
Berlese equipment can be purchased from Carolina Biological and Ward's
Natural Science (See Supplies chapter).