Invertebrate
Anatomy OnLine
Procambarus
©
Crayfish
27jun2006
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many
exercises available from
Invertebrate Anatomy OnLine
,
an Internet laboratory manual for
courses in Invertebrate Zoology. Additional exercises can be
accessed by clicking on the links to the left. A glossary and
chapters on supplies and laboratory techniques are also
available. Terminology and phylogeny used in these exercises
correspond to usage in the Invertebrate Zoology textbook by Ruppert,
Fox, and Barnes (2004). Hyphenated figure callouts refer to figures
in the textbook. Callouts that are not hyphenated refer to figures
embedded in the exercise. The glossary includes terms from this
textbook as well as the laboratory exercises.
Systematics
Arthropoda P, Mandibulata, Crustacea sP,
Eucrustacea, Thoracopoda, Phyllopodomorpha, Ostraca, Malacostraca
C, Eumalacostraca, Caridoida, Decapoda O,
Dendrobranchiata sO, Astacidea iO, Astacoidea
SF, Cambaridae F, (Fig 16-15, 19-67, 19-90)
Arthropoda P
Arthropoda, by far the largest and most diverse animal
taxon, includes chelicerates, insects, myriapods, and crustaceans as
well as many extinct taxa such as Trilobitomorpha. The segmented
body primitively bears a pair of jointed appendages on each
segment. The epidermis secretes a complex cuticular exoskeleton
which must be molted to permit increase in size. Extant arthropods
exhibit regional specialization in the structure and function of
segments and appendages but the ancestor probably had similar
appendages on all segments. The body is typically divided into a
head and trunk, of which the trunk is often further divided into
thorax and abdomen.
The gut consists of foregut, midgut, and hindgut and
extends the length of the body from anterior mouth to posterior
anus. Foregut and hindgut are epidermal invaginations, being derived
from the embryonic stomodeum and proctodeum respectively, and are
lined by cuticle, as are all epidermal surfaces of arthropods. The
midgut is endodermal and is responsible for most enzyme secretion,
hydrolysis, and absorption.
The coelom is reduced to small spaces associated with
the gonads and kidney. The functional body cavity is a spacious
hemocoel divided by a horizontal diaphragm into a dorsal pericardial
sinus and a much larger perivisceral sinus. Sometimes there is a
small ventral perineural sinus surrounding the ventral nerve cord.
The hemal system includes a dorsal, contractile,
tubular, ostiate heart that pumps blood to the hemocoel. Excretory
organs vary with taxon and include Malpighian tubules, saccate
nephridia, and nephrocytes. Respiratory organs also vary with taxon
and include many types of gills, book lungs, and tracheae.
The nervous system consists of a dorsal, anterior brain
of two or three pairs of ganglia, circumenteric connectives, and a
paired ventral nerve cord with segmental ganglia and segmental
peripheral nerves. Various degrees of condensation and cephalization
are found in different taxa.
Development is derived with centrolecithal eggs and
superficial cleavage. There is frequently a larva although
development is direct in many. Juveniles pass through a series of
instars separated by molts until reaching the adult size and
reproductive condition. At this time molting and growth may cease or
continue, depending on taxon.
Mandibulata
Mandibulata is the sister taxon of Chelicerata and
in contrast has antennae on the first head segment, mandibles on the
third, and maxillae on the fourth. The brain is a syncerebrum with
three pairs of ganglia rather than the two of chelicerates. The
ancestral mandibulate probably had biramous appendages and a
J-shaped gut, posterior-facing mouth, and a ventral food groove. The
two highest level mandibulate taxa are Crustacea and Tracheata.
Crustacea sP
Crustacea is the sister taxon of Tracheata and is
different in having antennae on the second head segment resulting in
a total of 2 pairs, which is unique. The original crustacean
appendages were biramous but uniramous limbs are common in derived
taxa. The original tagmata were head but this has been replaced by
head, thorax, and abdomen or cephalothorax and abdomen in many taxa.
Excretion is via one, sometimes two, pairs of saccate nephridia and
respiration is accomplished by a wide variety of gills, sometimes by
the body surface. The nauplius is the earliest hatching stage and
the naupliar eye consists of three or four median ocelli.
Eucrustacea
Eucrustacea includes all Recent crustaceans except
the remipedes. The taxon is characterized by a primary tagmosis
consisting of heat, thorax, and abdomen although the derived
condition of cephalothorax and abdomen is more common. Eight is the
maximum number of thoracic segments.
Thoracopoda
In the ancestral thoracopod the thoracic
appendages were turgor appendages used for suspension feeding in
conjunction with a ventral food groove. Such appendages and feeding
persist in several Recent taxa but have been modified in many
others.
Phyllopodomorpha
The compound eyes are stalked primitively although
derived sessile eyes occur in many taxa.
Malacostraca C
Malacostraca includes most of the large and
familiar crustaceans such as crabs, shrimps, lobsters, crayfish,
isopods, and amphipods. Primitively the trunk consists of 15
segments, eight in the thorax and seven in the abdomen but in most
Recent species the abdomen has only six segments (Fig 19-19). The
female gonopore is on the eighth thoracic segment and the male on
the sixth.
Decapoda O
The largest and most familiar crustaceans belong
to Decapoda. The 10,000 species of crabs, shrimps, crayfishes,
lobsters, and their relatives are decapods. The first three segments
of the decapod thorax are fused with the head to form a
cephalothorax and their appendages are maxillipeds. The remaining
five pairs of thoracic appendages bear simple or chelate walking
legs. The resulting ten legs accounts for the name “decapod”. A
large carapace extends posteriorly from the head and is fused
dorsally with all eight thoracic segments. Laterally the overhang of
the carapace encloses the branchial chamber with the gills. The most
primitive decapods (shrimps, lobsters, and crayfishes) have well
developed abdomens whereas the most derived taxa (true crabs in
Brachyura) have reduced, almost vestigial, abdomens (Fig 19-24).
Laboratory Specimens
Astacidea iO includes the clawed lobsters and
the freshwater crayfishes. This exercise can be used with any
crayfish or clawed lobster. Preserved crayfishes are provided by
biological supply companies without indication of identity but most
North American species belong to Procambarus (115 taxa),
Cambarus (82 taxa), or Orconectes (76 taxa).
Pacifastacus has eight species, restricted to Pacific coast
watersheds and Missouri River headwaters. The study should be
conducted on the stage of a dissecting microscope. Living specimens
should be anesthetized with chloroform-saturated water.
External Anatomy
Place a living or preserved crayfish in a dissecting pan
of appropriate size and take it to your bench for study. Crayfish
have bodies similar to that of the presumed ancestral
crustacean. Such a body is essentially shrimplike in that it is
elongate and nearly cylindrical in cross section. The abdomen is
well developed, its segmentation is readily apparent, and appendages
are present on every segment. Note the bilateral symmetry
of the animal and find anterior, posterior, dorsal, ventral, right,
and left.
Exoskeleton
The body is covered by a hard, jointed, nonliving
cuticle, or exoskeleton secreted by the underlying
epidermis. In general, the body wall consists of little more than
the exoskeleton and the epidermis beneath it because an animal with
a rigid exoskeleton has little need for any additional body
wall. The original body wall muscles have become specialized
individual muscles and no longer form continuous layers in the body
wall as they do in the wormlike ancestors of the
arthropods. Continuous layers of circular and longitudinal muscles
would be inefficient under a solid and immovable exoskeleton. The
exoskeleton provides strength, structural support, and protection so
connective tissue is not needed. The body cavity is a hemocoel, not
a coelom, so there is no peritoneum. Only the epidermis is essential
and must be retained because it secretes the exoskeleton.
The exoskeleton is molted periodically to allow the
animal to increase in size. A complicated musculature, derived from
the circular and longitudinal muscles of the ancestors, originates
and inserts on the inner surfaces of the exoskeleton and moves its
many parts. The exoskeleton between adjacent regions is thin and
flexible to permit motion. A complicated "endoskeleton" composed of
internal processes, called apodemes, extends internally from the
inner surface of the exoskeleton. The gills and the anterior and
posterior regions of the gut are covered with epidermis and a thin
exoskeleton.
Many parts of the exoskeleton bear small, articulated,
movable bristles called setae. These can be seen
over most of the body. Thicker articulated processes from the
exoskeleton are spines. Simple outgrowths of the
exoskeleton that are not articulated are usually called
teeth.
Tagmata
The body is composed of a linear series
of segments, or somites. The malacostracan body
consists of 19 segments but you cannot see or count them all. Each
one, however, bears a pair of jointed appendages, which are
visible and countable. In the ancestral crustacean all segments were
identical, or nearly so (homonomous), as were their appendages. In
derived crustaceans the segments and their appendages are
specialized for various purposes and for the most part no longer
resemble each other closely (heteronomous).
Groups of adjacent segments and their appendages tend to
have similar functions and together accomplish certain specialized
tasks. This results in a regionalization of the body into
tagmata. In crustaceans there were originally three
tagmata; the head, thorax, and abdomen. The crustacean head is
always composed of five segments but the thorax and abdomen are
variable. Within Malacostraca there is no variability in segment
number and there are always eight segments in the thorax and
(almost) always six in the abdomen.
Cephalothorax
It is common in crustaceans for the head to fuse with
some anterior thoracic segments to form a new tagma, the
cephalothorax. The appendages of these thoracic segments are
modified to serve as mouthparts and are known as maxillipeds. The
remaining unaffected thoracic segments form another new tagma, the
pereon. Such secondary tagmosis occurs in Decapoda where the
cephalothorax consists of the five head segments and the first three
thoracic segments and bears five pairs of head appendages and three
pairs of maxillipeds. The remaining five thoracic segments are the
pereon and their appendages are pereopods. Most authors use
different rules for decapods and refer to the head and entire thorax
as the cephalothorax even though only three appendages are fused
with the head. That custom is not followed here.
In Malacostraca the cephalothorax and most of the pereon
is covered dorsally and laterally, but not ventrally, by a double
sheet of exoskeleton called the carapace (Fig 1,
19-19). Although you cannot tell it by looking at the animal, the
carapace is an outgrowth of the exoskeleton of the last head
segment. It grows posteriorly to cover and protect the thoracic
segments. It is a fold of the body wall and as such consists of two
complete layers of the wall (Fig 8). The outer wall of the carapace
is sclerotized and covered by a thick exoskeleton and is hard and
strong but the inner wall has only a thin exoskeleton and is
transparent and flexible.
The segments of the cephalothorax are fused together and
cannot be told apart but those of the pereon remain independent and
are not fused together, even though dorsally and laterally they
appear to be. The carapace that covers them is not segmented but it,
remember, is not part of the pereon. Under the carapace, the pereon
is segmented. The segmentation of the thorax is apparent ventrally
where it is not covered by carapace. No such segmentation can be
seen in any view of the cephalothorax. A conspicuous transverse
dorsal groove divides the carapace into an anterior 1/3 and
posterior 2/3. This is the cervical groove and it
marks the boundary between head and thorax.
Figure 1. A dorsal view of an American lobster,
Homarus americanus. Adapted from Herrick, 1909.
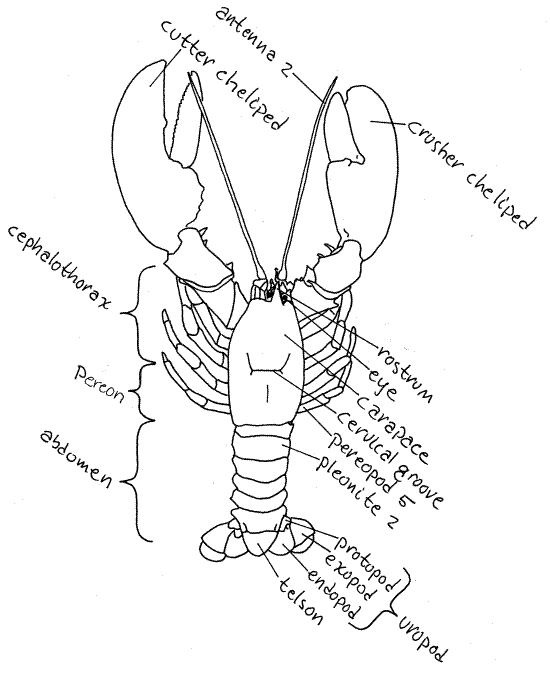
Anteriorly the carapace bears a
conspicuous anterior, median, pointed process called the
rostrum. The orbits are a pair of
semicircular notches, or sinuses, in the carapace lateral to the
base of the rostrum. Each orbit contains an eyestalk
with a compound eye at its distal end. The black,
multifaceted cornea of the eye covers almost of the
entire circumference of the stalk.
Pereon
The thorax is composed of eight segments, called
thoracomeres, and as we have seen, all eight are hidden
beneath the carapace. Each thoracic segment bears a pair of
appendages, or thoracopods. The anterior thorax,
consisting of three segments, is fused with the head to form the
cephalothorax. The posterior five segments remain independent of
each other and of the head. The posterior thorax, composed of these
five segments, is the pereon and its segments are
pereomeres. The pereon is not part of the
cephalothorax even though it is covered by the carapace. Do not
confuse the carapace with the cephalothorax. They are not the same
thing. The carapace is a fold of the body wall which, in decapods,
covers the cephalothorax and pereon.
Abdomen
The abdomen of primitive decapods is
well developed with clearly visible segments and powerful
longitudinal muscles. It is this abdominal musculature that is
primarily responsible for the culinary popularity of shrimp,
lobsters, and crayfish. The abdomen is also known as the
pleon and its segments are pleomeres.
Count the abdominal segments. There are six of them, all are
clearly visible, with none fused with another or with the
thorax. The posterior end of the body is not a segment. It is the
telson. If you counted it as a segment, you came up
with the wrong number. The anus is located on the
ventral side of the telson.
The exoskeleton of the abdominal segments of the
crayfish approximates the typical ancestral condition. Primitively,
each body segment is enclosed in four articulated exoskeletal
plates, or sclerites, that form a complete ring
around the segment. Dorsally is the tergite,
ventrally the sternite, and laterally there are two
pleurites, one on each side.
In astacideans (lobsters and crayfish) the tergite and
pleurites are fused together to form a hard arch of exoskeleton
covering the dorsal and lateral aspects of the segment. On segments
2-6 the pleurites extend ventrally past the body as side plates, or
epimera, which together form a shallow ventral
channel below the body of the abdomen.
The sternites cover most of the ventral surface of the
abdomen but the pleurites cover its lateral parts. For the most part
sternites are thinner and more flexible than tergites and
pleurites. They are transparent and, in living specimens, the
abdominal musculature and nerve cord can be seen through them. The
posterior margin of each sternite, however, is thick and heavy and
forms a reinforcing arch across the venter from one pleurite to the
other. The appendages articulate with the pleurites at the ends of
this sternal arch.
Appendages
Decapod appendages are easiest to study by beginning at
the posterior end and working forward. As you do this, keep in mind
that they are numbered in the opposite direction, from anterior to
posterior.
Before beginning your study of crayfish appendages it
might be a good idea to review the morphology of a typical
crustacean appendage. Arthropod appendages are paired, with one
pair per segment. Each appendage is a linear series of articles
joined by flexible articulations. Appendages may be biramous with
two branches, or uniramous, with only one branch.
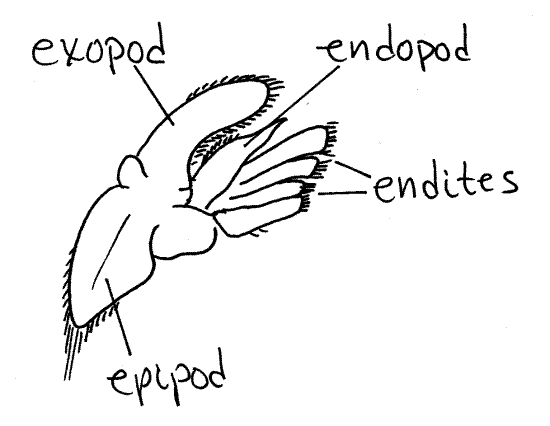
A biramous appendage has a basal
article attached by its proximal end to the body. From its distal
end arise two rami, or branches (Fig 2, 19-3B,
19-4A). The basal article is the protopod. Often
the protopod is divided into two articles, the coxa and basis. The
two rami are an outer, or lateral, exopod and an
inner, or medial, endopod. The two rami may be
composed of any number of articles depending on what they are
specialized to accomplish. They may be similar to each other or
different. If only one ramus is present and the appendage is said to
be uniramous (Fig 3). Sometimes additional branches
of the protopod or rami are present. Any additional branch on the
lateral side of the appendage is an exite and any
extra medial branch is an endite. Finally, an exite
on the base of the appendage is given the special name of
epipod.
Abdominal Appendages
Uropods
Study the abdominal appendages of your specimen but do
not remove them. Each of the six abdominal segments bears a pair of
appendages. Most of these are biramous. The last
(posteriormost) pair of abdominal appendages, located on abdominal
segment 6, are uropods (Fig 3, 19.2B).
Figure 2. A lobster (Homarus americanus)
uropod. Redrawn from Herrick (1909).
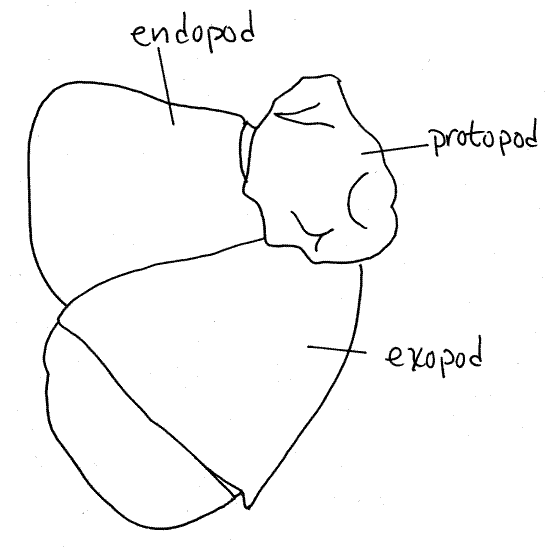
The uropods have a relatively small protopod and two
large, flat rami. The exopod is biarticulate (has
two articles). The distal border of each ramus bears a fringe of
setae. Spread the rami of the two uropods apart and array them
beside the telson (the telson is neither an
appendage nor a segment). The four rami plus the telson make up the
tail fan, which functions as a large paddle. With
the fan deployed, flexure of the powerful abdominal muscles moves
the fan rapidly forward under the body and results in the generation
of a forward jet of water that propels the animal backwards.
Pleopods
The remaining five pairs of abdominal appendages are
pleopods 1-5 (counting from anterior to
posterior). Pairs 2-5 are biramous and are similar to each other.
They are narrow and whiplike, although not very long (Fig 19-12B).
The pleopods of females are better developed than those of males and
are used to carry the eggs, which are glued to the fringe of setae
around the rami.
The first pleopods of males are uniramous and are
modified to serve as intromittent organs to transfer spermatozoa to
the female. In males the first pleopods are uniramous and are
referred to as gonopods. Adult male crayfish can
be either first form or second form. The gonopods of first form
males are sclerotized and hard and suitable for intromission. Those
of second form males are unsclerotized and soft and cannot transfer
sperm to the female. The first form gonopods have a species specific
shape that fits like a key into a seminal receptacle, the annulus
ventralis, with a corresponding shape, like a lock, on the female
venter. If you have a male determine if it is first form or second
form.
Using your own specimen if you have a first form male or
a borrowed one from another student, identify the specimen to genus
using the sculpturing of the 1 st pleopod. In the genus
Procambarus the male first pleopod usually terminates in
more than two processes. In the genus Cambarus, the first
form male first pleopod has two or less terminal processes and they
are bent at right angles to the shaft of the pleopod. In the genus
Orconectes the first form male first pleopod has two or
fewer processes and they are not bent. They arise at the end of the
shaft or from its posterior side and are parallel to the shaft or
slightly curved.
Pereon and Pereopods
Each thoracic segment bears a pair of appendages but
just as there are two distinctly different regions of the thorax,
there are two distinctly different types of thoracic appendages. The
appendages of the anterior three thoracic segments are
maxillipeds, are part of the cephalothorax, and function as
auxiliary mouthparts. The appendages of the posterior five thoracic
segments (pereomeres) are pereopods and function as
walking legs or pincers. They are part of the pereon.
The five segments of the pereon bear a total of 10
appendages which accounts for the name Decapoda (= 10 feet). All
decapods have five free thoracic segments and five pairs of
pereopods. The ten appendages are usually referred to loosely as
"walking legs" whether or not they are used for walking. All
pereopods lack the exopod and thus are uniramous. The
endopod is long and narrow (Fig 3). This shape of ramus is
referred to as stenopodous in contrast to a broad, flat, leaflike
phyllopod such as the uropod.
" Lift the ventral edge of
the carapace and note that it is attached dorsally to the thorax but
is free laterally. With strong scissors cut away the unattached
lateral portion of the left side of the carapace
without cutting into the attached portion. Be careful that you do
not cut into the body and do not damage the numerous structures in
the space below the carapace. This space is the branchial
chamber and it contains the gills. The
gills, which are outgrowths (epipodites) of the thoracic appendages,
will be studied later. They are feathery, white, filamentous
processes. Keep them moist so they do not dry out. Removal of the
carapace exposes the entire length of the pereopods and makes it
easier to study them. The lateral wings of the carapace are the
branchiostegites (Fig 8). The branchiostegites
enclose the branchial chamber. You just removed the left
branchiostegite.
Look at the middle walking leg. It is pereopod 3 (Fig 3,
19-20). The typical malacostracan thoracopod is composed of seven
articles. The two proximal articles are the subdivided protopod and
the distal five are the five articles of the endopod. The seven
articles are, in order from proximal to distal; coxa, basis,
ischium, merus, carpus, propodus, and dactyl. Find the seven
articles of pereopod 5. The proximal article is the coxa. It
is wide and short and articulates with the sternite of the third
pereomere. Distally it articulates with a short, narrow
basis. The basis joins with the ischium
along an oblique articulation.
Notice that the ischium appears to be composed of two
articles in that it has an oblique groove encircling it near its
articulation with the basis. This groove marks the location of the
fracture plane where the crayfish can deliberately
autotomize (auto = self, tome = cut) its limb. This plane is
specialized for this function and the animal can loose its limb, at
this plane only, without trauma or significant blood loss. It
usually replaces the limb with subsequent molts.
The ischium articulates with a long narrow merus. Next
there is a short carpus followed by a long
propodus. The final article is a sharp, pointed
dactyl, or nail.
Figure 3. Pereopod 3 of the lobster, Homarus americanus.
Adapted from Herrick, 1909.
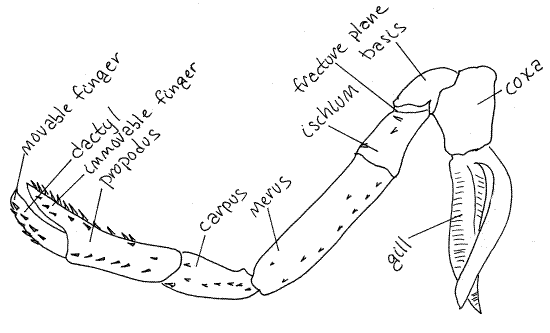
Pereopods 1-3 resemble each other in that the propodus
and dactyl form a prehensile, or grasping, pincer. The propodus
bears a long, fingerlike, distal process against which the dactyl
opens and closes. The dactyl is a movable finger
and the propodal process is an immovable finger. Such
a pincer is known as a chela and appendages bearing
one is said to be chelate. Pereopods 4 and five do
not have chelae and are " simple". The small chelae
of pereopods 2 and 3 are used to transfer food to the mouth.
Gills are associated with all thoracopods except
maxilliped 1. Most appendages have more than one gill and they may
be attached to the pleurite, coxa, or the articulating membrane
between the pleurite and coxa. The gills will be considered later.
Pereopods 4 and 5 are almost identical to each other and
differ from 1-3 in being simple, rather than chelate. The coxa of
pereopod 4 has a large membranous, leaflike epipod that is absent
from 5. This epipod extends vertically between the gills. Similar
epipods are present on the pereopods 1-3 and maxilliped 3. The first
pereopods are much larger than any other appendage. They are chelate
and, because of the striking size of their chelae, are referred to
as chelipeds and rarely as walking legs. The usual
seven articles are present and the chelae, as expected, are formed
of the propodus and dactyl.
Notice the variety of articulations in the joints of the
chelipeds. Flex and extend each joint to see what kinds of motion
its articulation allows. Each joint has an axis on which its two
articles rotate with respect to each other. Determine the axis of
rotation for each of the six joints of the cheliped.
The female gonopores are the external openings of the
oviducts. They are located on the medial side of the coxa of
pereopod 3 (= thoracomere 6). The male gonopores are the external
openings of the vasa differentia from the testes and are found at
the tip of the two short genital papillae (= penes) on the medial
surface of the coxa of pereopod 5 (= thoracomere 8). The position of
the male and female gonopores on these segments is constant
throughout Malacostraca.
A conspicuous oval annulus ventralis (=
seminal receptacle) is located on the ventral surface of the female
pereon between the coxae of pereopods 4 and 5. It bears a deep
median cleft, known as achink. The male gonopod is
inserted into the chink where it deposits sperm into the recess.
If your specimen is a male, find a female crayfish and look at the
annulus.
In the genus Procambarus the annulus ventralis is
surrounded by flexible cuticle and is freely movable (but may be
partly hidden by an overhang of the preceding sternite). The annulus
ventralis of Orconectes is inflexibly fused to the sternum
immediately anterior to it. In Cambarus the annulus
ventralis is not uniformly sclerotized and, even though fused with
the sternite, is capable of slight hingelike motion between the
anterior and more heavily sclerotized posterior portion. Study a
female crayfish and see if you can identify it to genus using these
features of the annulus ventralis.
During copulation the male turns the female over so
their ventral surfaces face each other. The male uses his chelipeds
to hold the female in position. The male gonopods (pleopod 1) are
held together and inserted into the annulus ventralis of the
female. The male genital papillae deliver sperm to the base of the
gonopods. The sperm travel along grooves in the gonopods to the
female annulus where they are stored, sometimes for weeks or months
before being used for fertilization. Immediately before shedding
eggs the female secretes a glue-like glair onto the ventral surface
of the abdomen and its pleopods. She then releases sperm from the
annulus onto the glair. Next she releases eggs from her gonopores on
thoracomere 6 onto the glair-covered pleopods. The eggs are
fertilized and stick to the pleopods where they are then brooded
until they hatch into miniature crayfish which remain associated
with the mother for a time.
Cephalothorax
Maxillipeds
The anterior three pairs of thoracic appendages are
maxillipeds. Unlike the pereopods, the maxillipeds are biramous. The
third maxillipeds are on the third thoracomere and
are immediately anterior to the chelipeds. Each is large and
intermediate in shape and size between the heavy, robust legs of the
pereon and the delicate mouthparts of the head. Each has a large,
stenopodous endopod and a small filamentous
exopod (Fig 4, Fig 19-2A). The protopod is divided into a
coxa and a basis, as it is in all thoracic appendages. The small
exopod arises from the distolateral corner of the basis. One
function of the third maxilliped is to protect the more delicate
appendages anterior to it.
Figure 4. Maxilliped 3 of the lobster Homarus. Redrawn
from Herrick (1909).
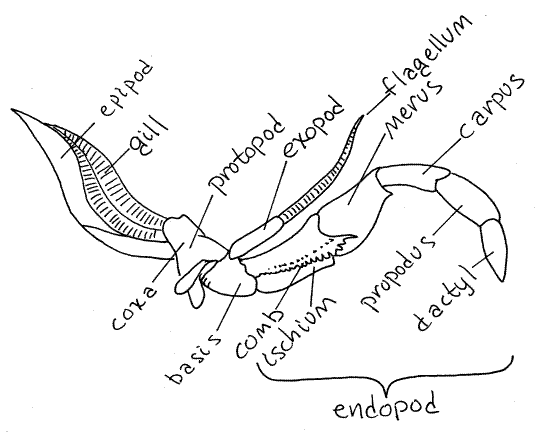
Hold the third maxillipeds aside and look at the next
appendage. It is the second maxilliped. It
too is biramous but is much smaller that the third. Its exopod is
longer than its endopod.
The first maxilliped is the appendage
of the first thoracomere. Its exopod resembles those of the other
maxillipeds and is long and narrow. Its endopod is short and
inconspicuous. There are two large wide, thin endites that cup over
the bulge of the mandible. The long posterior epipod extends
posteriorly into the branchial chamber.
The remaining five pairs of appendages are those of the
five head segments. The posterior three are mouthparts whereas the
anterior two are antennae with a sensory function.
Maxillae
The second maxilla is the appendage of
the fifth and posteriormost head segment and it lies immediately
anterior to the first maxilliped. It generates the water current
that pumps water out of the front of the branchial chamber. Its
basal portion bears four flat, narrow endites, a slender endopod,
and a long flat gill bailer, whose movements
generate the respiratory current through the branchial chamber. The
gill bailer, also known as the scaphognathite, is composed of the
exopod and epipod (Fig 5). The bailer lies beside the carapace and
extends anterior to and posterior to the basal part of the second
maxilla. The large thin trough-shaped epipod of the first maxilliped
extends back toward the branchial chamber. It functions in concert
with the gill bailer of the second maxilla. The bailer lies in and
beats in the trough formed by the epipod of the first maxilliped.
Figure 5. Maxilla 2 of the lobster, Homarus. After
Herrick (1909).
The first maxillae are small and more
delicate than the second. They are the smallest of the mouthparts
and lie curved tightly against the smooth, hard surface of the
mandible. Each has two broad endites and a narrow, larger,
endopod. There is no exopod.
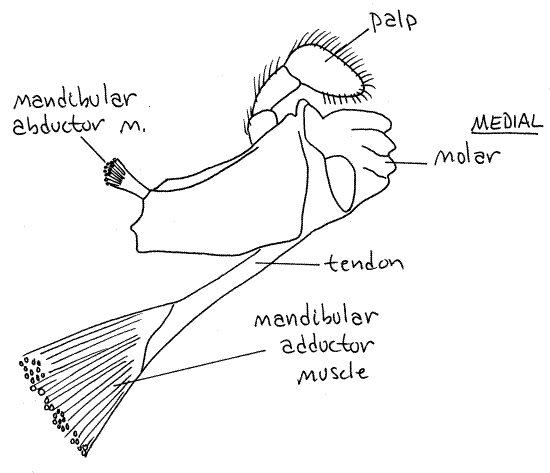
Mandibles
The mandibles are the most anterior of
the mouthparts. They are heavily calcified and equipped with
powerful muscles. There is a large basal portion which bears a
cutting edge on a medial lobe. A three articled
palp arches over the cutting edge. The mandible has
partial responsibility for shearing small pieces of food from larger
ones. It can rotate only slightly on its axis.
A single, large, fleshy labrum, or
upper lip, attaches to the anterior body wall just dorsal to the
mandibles and fills much of the space behind the cutting lobes. The
labrum is a fold of the body wall and is not an appendage.
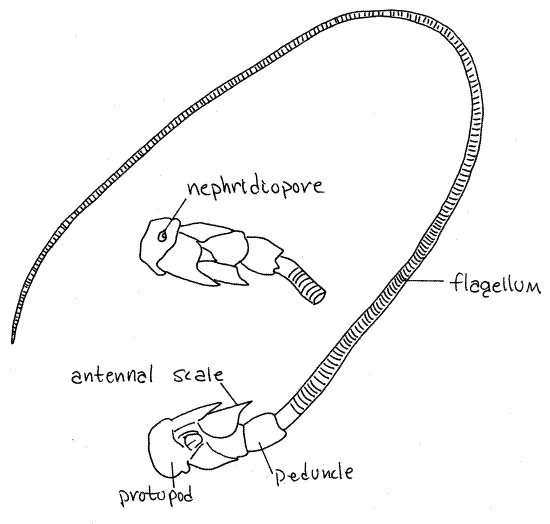
Antennae
The remaining two pairs of appendages are both sensory
antennae (Fig 1, 19-2A). The biramous second antennae
are by far the larger of the two pairs (Fig 7, 19-2B). Each arises
by a biarticulate protopod consisting of a proximal coxa and a
distal basis. The short, wide exopod is called the antennal
scale. It arises from the basis. The endopod, which also
arises from the basis, has a short thick basal peduncle
of three articles and a very long narrow, whiplike flagellum
of many articles. The lower surface of the coxa bears a small
circular tubercle with an opening in its center. This is the
nephridiopore, the external opening of the kidney.
Figure 6. Mandible of the lobster, Homarus. Redrawn
from Herrick (1909).
The first antennae (= antennules), are
situated below the eyestalks. They are much smaller than the second
antennae. Each has a triarticulate basal stalk from which arise two
slender multiarticulate flagella. There is a
statocyst in the basal article of each first antenna.
The two eyestalks are also on the
anterior head. Each bears a conspicuous compound eye
at its distal end.
Respiratory System
The respiratory apparatus of decapod crustaceans
consists of numerous gills, a branchial chamber to house them, and a
water pump to generate a respiratory current over them. The gill
bailer of the second maxilla is the pump. The number of gills in
crayfishes is 17-18 pairs and lobsters have 20 pairs. The gills are
epipods attached to the coxa or the adjacent articulating membrane
or pleurite of most thoracopods.
The pale, feathery gills are housed in
the branchial chamber between the lateral carapace
and the body (Fig 8, 19-3A). You opened the left branchial chamber
when you removed the left carapace and the gills on that side are
exposed to view. The right chamber should still be intact and
covered by the branchiostegites of the carapace. Note that the
branchial chamber is outside the body and is filled with water, even
though it is under the carapace and appears to be internal.
The gills extend vertically into the branchial chamber
from their attachments on or near the coxae of the thoracopods (Fig
19-38B). Look closely and see that the gills of successive
appendages are separated from each other by the long, membranous
epipods of those appendages. The epipods form the boundaries of
water channels that extend vertically from the free, unattached
ventral edge of the carapace upwards to the attached dorsal edge
(Fig 19-38B). Notice that the coxae of each pair of adjacent
pereopods are shaped so that together they form V-shaped
inhalant channels that lead into one of the vertical
channels in the branchial chamber. There are five such inhalant
channels.
Figure 7. Antenna 2 of the lobster, Homarus. The
ventral surface and nephridiopore are shown in the inset. Redrawn
from Herrick (1909).
Dorsally the several vertical channels converge on an
oblique, exhalant channel that runs anteriorly
along the dorsal margin of the branchial chamber (Fig 19-38B). The
floor of the anterior half of this channel is made of the epipod of
maxilliped 1 which separates the channel from the gills. The roof
and walls are formed by the branchiostegite of the carapace and
body. The gill bailer of maxilla 2 lies in the anterior end of the
exhalent canal in the epipod of maxilliped 1. Undulations of the
bailer generate the negative pressure that draws water in the
inhalant canals, vertically over the gills, and then anteriorly, to
exit lateral to the mouthparts.
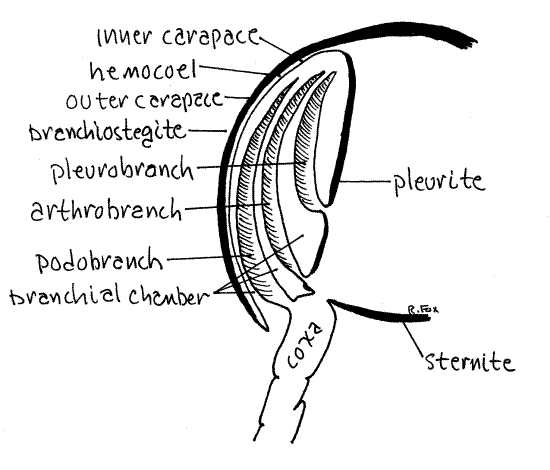
All decapod gills are associated with thoracopods but
differ in the exact location of their attachment. Podobranchs (podo
= foot, branch = gill) arise on the lateral surface of the coxa of
the thoracopod (Fig 8). Arthrobranchs (arthro = joint) arise on the
thin articulating membrane between the coxa and the pleurite of the
body wall. Pleurobranchs (pleuro = side) are attached to the
pleurite dorsal to the limb articulation but are usually absent in
crayfishes.
Look at pereopod 4. Like most crayfish thoracopods it
has one podobranch and two arthrobranchs (one anterior and one
posterior). Pereopods 1-4 and maxilliped 3 each have one podobranch
and two arthrobranchs. Pereopod 5 may or may not have a
pleurobranch, depending on taxon. Maxilliped 2 has one small
podobranch and an arthrobranch. Maxilliped 1 has no gills but has
the important epipod that encloses the exhalant water channel and
the gill bailer. Some crayfishes have a pleurobranch on pereopod 5
and some do not. Crayfishes thus have a total of 17-18 pairs of
gills whereas lobsters, with several pleurobranchs, have 20.
Figure 8. Cross section of the branchial chamber and gills
of a generalized decapod.
" Snip the end from one of
the gills, place it in a 6-cm culture dish of water and examine it
with the dissecting microscope.
Crayfishes and lobsters have filamentous (=
trichobranchiate) gills in which the respiratory surface consists of
numerous long filaments radiating from a central
axis, rather like a bottlebrush (Fig 19-37C,D).
Look at the cut surface of the gill axis. Here you will
see two blood channels, cut in cross-section, that extend the length
of the gill (Fig 19-37C). One is the afferent channel that takes
unoxygenated blood into the gill and the other is the efferent
vessel that drains oxygenated blood away from the gill. Similarly,
each filament is partitioned into two channels by a longitudinal
septum. One channel is afferent, the other efferent.
Additional Exercises
Watch the video "America's crayfish, Crawling in
troubled waters", produced by Virginia Tech and the U.S. Fish and
Wildlife Service.
Internal Anatomy
Pending completion of this section, the
description of the internal anatomy of Homarus americanus
(American lobster) in this series (
Invertebrate Anatomy OnLine
)can be substituted
.
References
Bullough WS.
1958. Practical Invertebrate Anatomy
2 nd ed. MacMillan, New York. 483p.
Herrick FH. 1909. Natural
History of the American Lobster. Bull. Bur. Fish. 26:150-408, pls
33-47.
Hobbs
HH. 1991. Decapoda, in Thorp JW, Covich AP
(eds). Ecology and classification of North American freshwater
invertebrates. Academic Press, San Diego.
Huxley TH. 1880. The
Crayfish, An Introduction to the Study of Zoology. Appleton, New
York. 371p. (Reprinted 1973, M.I.T. Press, Cambridge.)
Lochhead JH. 1950. Crayfishes
(and Homarus) in F. A. Brown (ed) Selected Invertebrate
Types. Wiley, New York. pp422-447.
Pennak, R.W. 1989. Freshwater
invertebrates of the United States, 3ed. Wiley.
Ruppert EE, Fox RS, Barnes
RB. 2004.
Invertebrate Zoology, A functional evolutionary approach, 7 th
ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Snodgrass RE.
1952. A Textbook of
Arthropod Anatomy. Cornell Univ. Press, Ithaca. 363 p. (reprinted
1971 by Hafer Publishing, New York) (crayfish pp 142-179).
Supplies
Dissecting pan
Living or preserved crayfish
Dissecting set
Chloroform-saturated water for living specimens.
Video projection equipment
The video, “Crawling in troubled waters” and a spectacular crayfish
poster are available from Earthwave Productions Inc,
www.earthwave.org,
817.443.0258 or Virginia Cooperative Extension,
monteh@vt.edu, 540.231.6192