Invertebrate Anatomy OnLine
Pomacea paludosa ©
Florida Apple Snail
3jul2006
Copyright 2003 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises can be accessed by clicking on the links to the left. A glossary and chapters on supplies and laboratory techniques are also available. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Mollusca P, Eumollusca, Conchifera, Ganglionura, Rhacopoda, Gastropoda C, Prosobranchia sC, Caenogastropoda O, Mesogastropoda sO, Ampullarioidea SF, Ampullariidae F (Fig 12-125)
Mollusca P
Mollusca, the second largest metazoan taxon, consists of Aplacophora, Polyplacophora, Monoplacophora, Gastropoda, Cephalopoda, Bivalvia, and Scaphopoda. The typical mollusc has a calcareous shell, muscular foot, head with mouth and sense organs, and a visceral mass containing most of the gut, the heart, gonads, and kidney. Dorsally the body wall is the mantle and a fold of this body wall forms and encloses that all important molluscan chamber, the mantle cavity. The mantle cavity is filled with water or air and in it are located the gill(s), anus, nephridiopore(s) and gonopore(s). The coelom is reduced to small spaces including the pericardial cavity containing the heart and the gonocoel containing the gonad.
The well-developed hemal system consists of the heart and vessels leading to a spacious hemocoel in which most of the viscera are located. The kidneys are large metanephridia. The central nervous system is cephalized and tetraneurous. There is a tendency to concentrate ganglia in the circumenteric nerve ring from which arise four major longitudinal nerve cords.
Molluscs may be either gonochoric or hermaphroditic. Spiral cleavage produces a veliger larva in many taxa unless it is suppressed in favor of direct development or another larva. Molluscs arose in the sea and most remain there but molluscs have also colonized freshwater and terrestrial habitats.
Eumollusca
Eumollusca, the sister taxon of Aplacophora, includes all molluscs other than aplacophorans. The eumolluscan gut has digestive ceca which are lacking in aplacophorans, the gut is coiled, and a complex radular musculature is present.
Conchifera
Conchifera, the sister taxon of Polyplacophora, includes all Recent molluscs other than aplacophorans and chitons. The conchiferan shell consists of an outer proteinaceous periostracum underlain by calcareous layers and is a single piece (although in some it may appear to be divided into two valves). The mantle margins are divided into three folds.
Ganglioneura
Most Recent molluscs are ganglioneurans, only the small taxa Aplacophora, Polyplacophora, and Monoplacophora are excluded. Neuron cell bodies are localized in ganglia.
Rhacopoda
The mantle cavity is posterior in the ancestor although it may be secondarily moved to an anterior position by torsion. This taxon includes gastropods and cephalopods.
Gastropoda C
Gastropoda is the largest molluscan taxon and is the sister group of Cephalopoda. Gastropods are united by descent from a torted ancestor although many exhibit various degrees of detorsion. Many are coiled and asymmetrical but the ancestor was probably symmetrical. Gastropods are relatively unspecialized molluscs known collectively as snails. The univalve shell, present in the ancestral gastropod and in the majority of Recent species, is reduced or lost in many representatives. The flat creeping foot was inherited from their eumolluscan ancestors but gastropods have developed a distinct head with an abundance of sophisticated sense organs. The originally posterior mantle cavity has become anterior as a consequence of torsion, although detorsion has reversed this condition in many. Gastropods were originally gonochoric and most remain so but many derived taxa are hermaphroditic. Most are marine but many taxa have invaded freshwater and the only terrestrial molluscs are gastropods. Most have a single gill, atrium, and nephridium but the most primitive representatives have two of each. Only one gonad, the right, is present. The ancestor probably had an operculum. The nervous system is streptoneurous (twisted by torsion).
Prosobranchia sC
Prosobranchia was once one of three great gastropod subclasses but it is no longer considered to be a monophyletic taxon, although the concept continues to be used as a pedagogical convenience. Prosobranchs are the gastropods most like the ancestral snails. They are torted and most have a shell and are coiled and asymmetrical. The mantle cavity is anterior. Most are gonochoric and most have an operculum. Most have only one gill in the mantle cavity but some primitive taxa have two. The right atrium is lost in most. Prosobranchs are specialized for life in marine benthic habitats although representatives are also found in freshwater and on land.
Caenogastropoda O
Caenogastropoda includes the two large and successful groups, mesogastropods and Neogastropoda. One gill, one nephridium, and one atrium are present. The gill is monopectinate, with filaments on only one side of the central axis. This new gill is less prone to fouling with sediment and silt and is probably largely responsible for the success of these snails as it allowed invasion of soft-bottom habitats.
Mesogastropoda sO
Although Mesogastropoda is no longer thought to be monophyletic it remains a useful pedagogic device and the concept is still widely employed. The mesogastropod osphradium is a simple ridge and the radula is taenioglossate, with seven teeth in each transverse row. Mesogastropods occur in marine, freshwater, and terrestrial habitats but most are marine.
Laboratory Specimens
Apple snails belonging to the genus Pomacea are sold by pet shops and are popular in home aquaria. They are large, readily available alive, easily dissected snails useful in the laboratory as exemplars of gastropod anatomy. They belong to the prosobranch taxon Ampullariidae which consists of tropical and subtropical, freshwater, amphibious prosobranchs. The world's largest freshwater snails belong to this taxon and one species, Pomacea paludosa, occurs naturally in the extreme southeastern United States. Apple snails of various species have been deliberately introduced as a possible human protein source in several tropical and subtropical localities. Unfortunately, as is often the case with introductions, they have become serious pests in rice fields and as intermediate hosts of parasites, resulting in restrictions on transport and sale. The sale of some ampullariids by the pet trade is now restricted although regulations vary from state to state. Pet and aquarium shops still offer a variety of living snails in several genera which can be purchased relatively inexpensively for laboratory study. Pomacea bridgesii, is still legally marketed and may appear in pet stores in various color morphs (banded, “blue”, white, and yellow). These are known by a variety of names such as blue snail, blue mystery snail, blue apple snail, ivory snail, and golden mystery snail. The banded snail has natural pigmentation whereas the others are the result of selective breeding by the pet trade. The ramshorn snail, Marisa, is also an ampulariid and is usually available. Any ampulariid can be used for this exercise although it was originally written specifically for the Florida apple snail, Pomacea paludosa. It works well with P. bridgesi.
Pet stores may also supply mystery snails in the same superfamily (Ampullarioidea) but the family Viviparidae. Pet shops and biological supply companies cannot be relied upon to distinguish between apple snails and mystery snails. Ampulariids and viviparids differ anatomically and biologically. The characteristics in Table 1 can be used to distinguish between the two families. The umbilicus, proboscis, and ocular peduncles are the easiest way to identify the snails without dissection. Either species is suitable for an introduction to gastropod anatomy but this exercise is written for an ampullariid.
Table 1. External identification characters separating Ampulariidae and Viviparidae.
|
Ampullariidae (apple snails) |
Viviparidae (mystery snails) |
|
amphibious |
aquatic |
|
respire in water or in air at water surface |
respire in water only |
|
mantle cavity with lung and gill |
mantle cavity with gill only |
|
with short eye-bearing ocular peduncles beside the cephalic tentacles |
no ocular peduncles |
|
no proboscis |
mouth is at the tip of a tubular proboscis |
|
lay eggs in calcareous shells above water surface |
brood young in uterus, do not lay eggs |
Natural History
Ampullariids lay large eggs in masses above the surface of the water and each egg is enclosed in a fragile, calcareous shell. Ampullariids are herbivores. They are typical prosobranchs except that they have a lung, in addition to the typical prosobranch left gill, and are capable of both aerial and aquatic respiration. Do not confuse apple snails with pulmonate snails. Pulmonates also have lungs but they do not have gills and the similarities between them are the result of convergence. The lungs of the two are analogous, not homologous.
Anesthetization and/or Relaxation
Conduct the study with a relaxed or recently killed specimen. Anesthetization and relaxation of freshwater snails is difficult and no method is entirely satisfactory.
Freezing, which kills and partially relaxes the specimens, is probably the best technique. Snails should be placed in a small bucket (small enough to fit in the freezer) with as much water as possible. The large volume slows the freezing process and increases the likelihood the snails will be relaxed. Allow them to freeze slowly without any disturbance. Most individuals will succumb in a partially relaxed condition that is entirely satisfactory for dissection. Thaw the ice well before the laboratory meeting. Dissect the snails immersed in tapwater.
One anesthetization method is to crack the shell into several pieces using a C-clamp or pliers and place the partially exposed animal in 5% non-denatured ethanol for about 6 hours prior to the dissection. A similar technique is to wedge the operculum of an intact specimen open with a piece of applicator stick and then place the snail in a dish or dissecting pan of 5% non-denatured ethanol or chloroform-saturated tapwater and let it relax.
According to the literature, snails can also be killed in a partially relaxed condition by chilling them thoroughly and then plunging them briefly into boiling water for about 30 seconds but I have not tried it.
External Anatomy
Most of the anatomy of an apple snail can be seen with no dissection other than removal of the shell and opening of the mantle cavity. Complete study of the nervous, reproductive, and digestive systems however requires that the hemocoel be opened.
Shell
Examine the shell of a living snail or an empty shell. The thin, translucent, calcareous shell is composed of a series of coils, or whorls (Fig 1, 12-27A,B), spiraled around a central axis, or columella. The columella is not visible in an intact shell. The shell of Pomacea is globular (resembling an apple), consisting of one enormous body whorl, which contains most of the soft parts of the animal, and a few much smaller whorls forming low spire above it. The body whorl opens to the exterior via the large aperture on one side of the central axis. A deep pit, the umbilicus, extends from the exterior into the core of the columella.
Figure 1a. The shell and operculum of Pomacea. Gastrop138L.gif
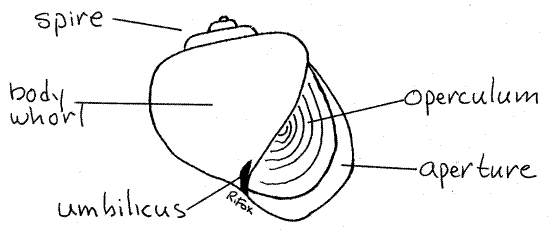
>1a. If available, examine an empty shell in which a window has been cut to reveal the columella. Observe the relationship between the umbilicus and the columella. <
>1b. If a computer with Internet connection is available in the laboratory visit www.applesnail.net/content/species/pomacea_bridgesi.htm (Ghesquiere, 2001) and click on Interactive 3D model. Point to the shell, click and hold, and drag the curser slowly to rotate the shell. View the shell from several angles and look into the umbilicus and aperture as far as you can see.
Hold the shell so the aperture is facing you and the spire points up as in Figure 1. Determine if the aperture is on the right or the left of the columella. If it is on the right, the snail is dextral, or right handed. If on the left, it is sinistral, or left-handed. Is your mud snail sinistral or dextral? ________ Most gastropods, including ampulariids, are dextral.
The calcareous layers of the shell are covered by a thin, glossy, proteinaceous periostracum. The operculum is a thin teardrop-shaped disk of flexible protein (conchiolin) that forms a door for the aperture. It is attached to the foot of the snail and is moved into position as the head and foot are retracted into the body whorl.
Extraction
" A snail is attached to its shell by the columellar muscle which must be detached in order to extract the animal from the shell. This muscle extends from the posterior foot to its origin on the columella deep inside the aperture. It is not accessible from the aperture and the shell must be broken to gain access to it.
Figure 1b. The shell of Viviparus. A window has been cut in the body whorl to reveal the columella. Viviparus has no umbilicus. Gastrop175L.gif
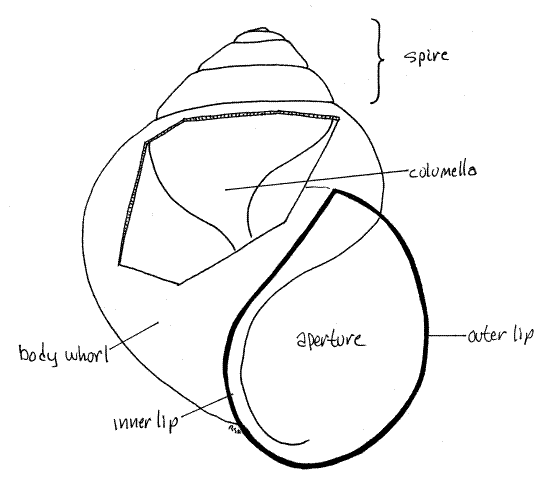
Pomacea shells are often thin and can usually be removed in pieces with a pair of heavy forceps but if it is too strong for this, use a small C-clamp, pliers, or vise to crack it. Follow these directions carefully.
Begin at the outer lip of the aperture and use the forceps to break away pieces of the shell, being very careful that you do not damage any soft tissues. If the shell is too strong for the forceps, use a C-clamp to crack it. If using a C-clamp never apply pressure to parts of the shell that are already broken and loose. Place the snail in the clamp and apply pressure around the aperture until the shell cracks. You can hear and feel it crack. Remove the shell from the clamp and see if any pieces can be removed without damage to the soft anatomy. If so, remove and discard them. Put the shell back in the clamp and apply pressure to an uncracked portion of the shell until it cracks. Remove any loose pieces of shell as before. Never close the clamp on soft parts or apply additional pressure after the shell cracks. Pressure should be applied only to sound, uncracked parts of the shell.
After you remove some of the body whorl around the aperture, look for the white columella in the core of the shell (Fig 1b, 12-27A). Continue around the body whorl, removing bits of shell, until you find the place where the columellar muscle inserts on the columella. Use a scalpel to scrape (rather than cut) the columellar muscle from the columella.
Once all parts of the muscle are severed, you can unwind the animal from the remains of the shell. When you think you have detached all the muscle, exert a steady but gentle pull on the operculum and/or foot. The visceral mass should uncoil from the whorls of the shell and slide smoothly out of the aperture but do not force it. If it does not come out easily, look for intact parts of the muscle you overlooked, detach or sever them, and try again. Never use force to remove the animal as the visceral mass is delicate and will tear apart. Should this happen your dissection will be made more difficult.
Place the snail in a small dissecting pan of tapwater (if dead) or 5% ethanol (if living).
Soft Anatomy
The major regions of the mollusc body are the head, foot, and visceral mass (Fig 2). The foot is ventral, the head is anterior to it and the visceral mass dorsal to the foot and posterior to the head. Much of the visceral mass is coiled and its posterior end tapers and spirals to fit in the upper whorls of the shell.
Figure 2. Dorsal view of an undissected female Pomacea paludosa with the shell removed. Gastrop139La.gif
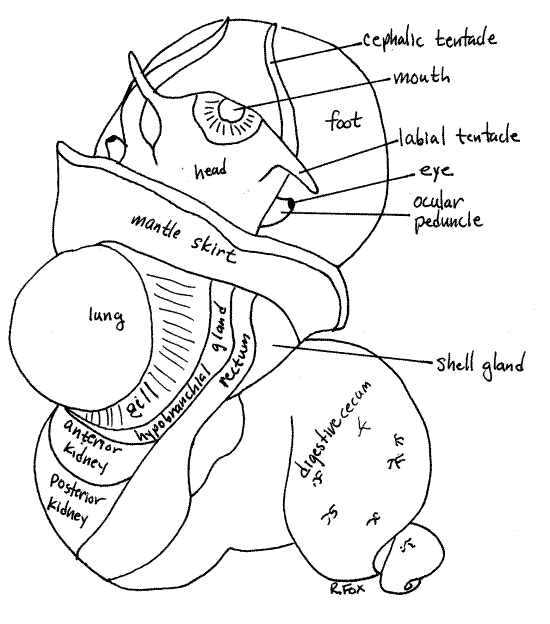
Foot
The broad, flat, foot is ventral and the rest of the body sits on it. The foot is a large muscular mass covering the ventral surface of the snail (Fig 2, 12-14A). Its smooth ventral surface is the sole. When retracted, it folds transversely on itself to fit into the aperture. In partly relaxed specimens the foot is usually folded in this manner. If it is folded, extend it. The horny operculum is attached to the dorsal surface of the posterior half of the foot.
Head
The head, dorsal to the anterior foot, is connected to the visceral mass by a slightly narrower neck. The anterior end of the head is blunt and bears the mouth. A labial tentacle is present on either side of the mouth (Fig 2) and a long cephalic tentacle extends on each side of the dorsal head. A short ocular peduncle arises from the base of each cephalic tentacle and bears an eye at its distal end.
Figure 3. View of the left side of an undissected female Pomacea. The shell has been removed. Gastrop140La.gif
Lateral to the tentacles, the two sides of the neck each bear a thin sheet of tissue that can be rolled into a tube, or siphon (Fig 4). That of the left is the inhalant siphon to bring the respiratory medium (water or air) to the respiratory organs (gill or lung) in the mantle cavity. The siphon on the right is the exhalant siphon through which the respiratory current exits the mantle cavity. In contracted specimens the siphons may be small and scarcely distinguishable but in active living animals the inhalant siphon can be extended, like a snorkel, several centimeters to reach the water surface while the animal remains submerged.
Figure 4. Dorsal view of an undissected male Pomacea. The shell has been removed. Gastrop141La.gif
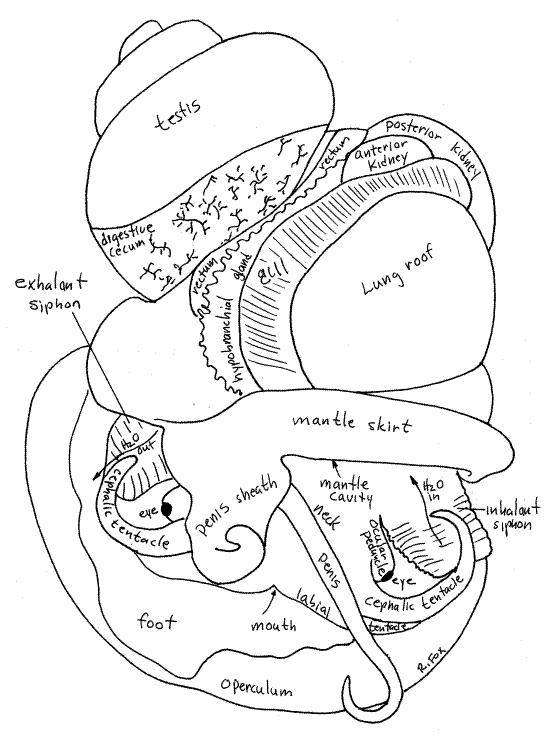
Mantle Cavity
Exterior
The mantle is the dorsal body wall. Anteriorly a fold of the mantle arches over the anterior visceral mass and forms a deep recessed pocket, the mantle cavity. Its free anterior edge forms a collar, the mantle skirt, which completely encircles the neck (Figs 2, 3, 4). The mantle cavity opens above the head to the exterior water. The mantle skirt fits against the edge of the aperture of the shell and secretes its growing edge.
The roof of the mantle cavity is the aforementioned fold of mantle and its floor is the dorsal body wall of the anterior visceral mass. The floor of the mantle cavity is also the roof of the hemocoel, or body cavity. Many organs are associated with the roof and the floor of the mantle cavity and can be seen there with little or no dissection (Figs 2, 3, 4).
The roof of the mantle cavity may be covered with a layer of black pigment. If so, it should be rubbed loose with a blunt probe and then squirted away with jets of water from a plastic Pasteur pipet.
The mantle cavity of fresh specimens will invariably be filled with copious mucus from the hypobranchial gland. Use jets of water from the pipet to clear it away. When the water in your dissecting pan becomes fouled with mucus and particulates, replace it with fresh.
Figure 5. Dorsal view of a female Pomacea with the mantle cavity opened. Gastrop142La.gif
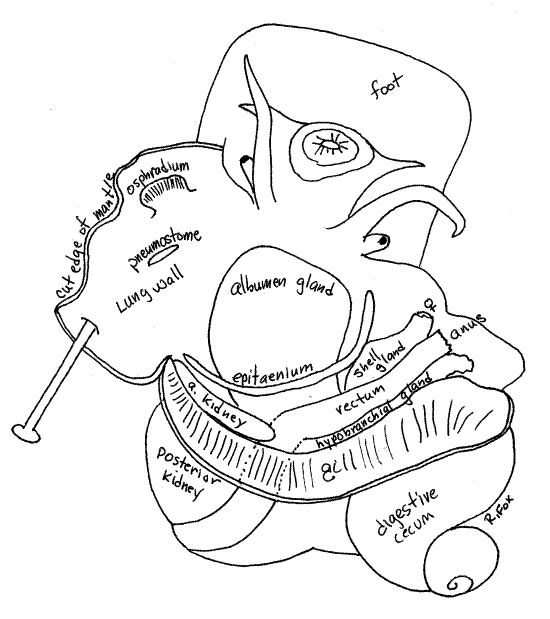
The ampullariid mantle cavity, unlike that of most mesogastropods, is divided into a lung on the left side and a larger branchial chamber containing the gill on the right. The branchial chamber opens anteriorly to the exterior. Ampulariids are amphibious and can respire in air or water.
In males, the right margin of the mantle skirt is elaborated to form a penis sheath through which the penis can be extended during copulation (Fig 4). The penis can be extended through the lumen of the sheath to emerge at the tip when needed. In some individuals, the sheath may be folded inside the mantle, under the skirt, where it is not immediately apparent.
Place the snail in the center of the dissecting pan and force two strong (#4) insect pins through the operculum to hold it flat against the wax of the pan so the mantle is uppermost, facing you. Gently slip a blunt probe into the mantle cavity to explore its depth. From the outside, study both sides of the roof of the mantle cavity. If the roof is heavily pigmented, you may not see all its organs at this time but you will get another chance later.
The lung is a large sac on the far left of the roof of the mantle cavity. When filled with air, as it often is, it bulges conspicuously (Figs 2, 3, 4).
The gill is on the roof of the mantle cavity to the right of the lung. It may be visible through the roof or you may need to lift the mantle skirt and look into the cavity to see it. Its anterior end lies just about on the dorsal midline. The gill is in the branchial chamber which is the right side of the mantle cavity. Although located on the right, the gill is homologous to the left gill of the ancestral gastropods.
Figure 6. Dorsal view of a male Pomacea with the mantle opened and deflected. Gastrop143La.gif
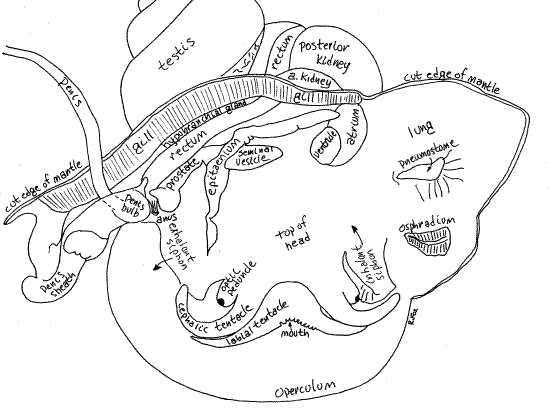
Mantle Cavity Interior
" Look on the outside of the mantle cavity and locate the oblique line between the lung and gill. With fine scissors, make a cut beginning at the free edge of the mantle skirt and extending along this oblique line all the way to the posterior end of the mantle cavity. The incision will be oblique and should not cut into the lung on its left or the gill on its right. Deflect the right and left sides of the roof and observe that the mantle cavity is divided into two parts; a lung and branchial chamber (Figs 5, 6).
A symbiotic mite in the genus Atax lives in the mantle cavity of Pomacea. Watch for it and tell the instructor if you find one.
The lung is entirely contained within the left side of the roof and is physically separate from the branchial chamber by a partition. A small but distinct opening, the pneumostome, penetrates the partition between lung and branchial cavity and connects the two (Figs 5, 6). Note the proximity of the pneumostome to the inhalant siphon.
The osphradium is a sense organ attached to the roof of the mantle cavity on the left side near the base of the left siphon (Figs 5, 6). It resembles a miniature gill and monitors the chemistry and silt load of the inhalant water current.
The gill consists of a longitudinal central axis from which numerous slender, pointed gill filaments extend along one side. The axis is connected to the roof and the filaments hang into the cavity. The gill extends obliquely to the posterior end of the mantle cavity.
The long, narrow, white hypobranchial gland lies in the roof of the mantle cavity, along the right margin of the gill (Figs 5, 6). It may be obscure and difficult to distinguish if the epithelium is pigmented. It secretes mucus into the mantle cavity.
The rectum is a large tube immediately to the right of the hypobranchial gland (Figs 2, 5, 6). It emerges from the left side of the visceral mass and runs across the right side of the roof of the mantle cavity and can be seen on the outside. When it contains feces, it is dark and easily recognized but the empty rectum of a starved animal is less conspicuous.
The end of the rectum forms a short anal papilla with the anus at its tip (Fig 4, 5, 6). The anus is inside the mantle cavity on the right of the gill where it empties into the exhalant current.
Examine the floor of the mantle cavity and find a low ridge, the epitaenium, extending obliquely across it (Fig 5, 6). The epitaenium begins at the posterior end of the cavity and extends anteriorly to the exhalant siphon on the right.
During aerial respiration, the epitaenium is elevated to form a partition reaching from the floor to the roof of the mantle cavity. When so raised, it isolates the inhalant flow of air on the left side from the branchial chamber on the right. It channels air into the pneumostome and lung and prevents the flow of water into the lung. Muscular activity generates a tidal flow of air into and out of the lung. When breathing air, the snail extends the inhalant siphon above the surface of the water and uses it as a snorkel.
During aquatic respiration, with the epitaenium lowered, water enters the left siphon and passes over the osphradium. It then flows posteriorly in the left side of the mantle cavity and moves across the gill filaments to the right side of the mantle cavity. From here it passes anteriorly to exit the right siphon after passing the anus and nephridiopore.
" Open the roof of the lung with a longitudinal incision made with fine scissors. The roof is muscular and heavily vascularized. Find the opening of the pneumostome inside the lung and insert a blunt probe through it. The probe should emerge in the branchial chamber.
Reproductive System
Most of the reproductive system is visible without dissection and much of it is located in the mantle cavity making it convenient to study at this time. Like most prosobranchs, Pomacea paludosa is gonochoric and fertilization is internal. Members of this family are oviparous and females lay masses of large, yolky, shelled eggs above the waterline. The reproductive system consists of the gonad and a regionally specialized and differentiated gonoduct which connects it with the exterior (Fig 12-56). The gonad is high in the coils of the visceral mass but most of the rest of the reproductive system is visible on the floor of the mantle cavity and no further dissection is needed to reveal it. The size and appearance of many reproductive organs vary considerably depending on maturity. In immature or reproductively inactive individuals some structures, including the ovary, may be difficult or impossible to find.
Female
The ovary is on the inner curve of the upper whorls of the visceral mass where it is surrounded by the digestive cecum (Fig 2). It can be difficult to find. It consists of numerous, superficial, branched tubes draining into the inconspicuous oviduct which connects with the albumen gland on the floor of the mantle cavity. You probably will not see the ovary or oviduct.
The female gonoduct exits the vicinity of the digestive cecum and crosses the floor of the mantle cavity where it is differentiated into an albumen gland and a shell gland, both of which are easy to find in mature specimens (Fig 5). The albumen gland of mature, reproductively active females is a large, pink, orange, or green organ lying on the floor of the mantle cavity and bulging into it. During oviposition the albumen gland applies a thick coat of pink albumen to each egg. Part of the gland extends into the visceral mass where it is lost from view.
The shell gland exits the right side of the albumen gland, passes under the epitaenium, and extends on the right side of the mantle cavity to the base of the exhalant siphon. It is a large diameter tube resembling the rectum, which it lies beside and parallels. This gland secretes the calcareous shell around the egg.
The external opening of the female gonoduct is the female gonopore which, in mature specimens, is visible in the mantle cavity on the end of a short papilla on the shell gland, just to the right of the anus. In immature individuals it cannot be demonstrated.
Male
The pale, cream-colored testis is located in the upper whorls of the visceral mass and, in mature reproductively active animals, is large (Fig 4). Do not confuse it with the much darker digestive ceca which also occupy the upper whorls. The inconspicuous vas deferens exits the testis and passes along the inside surface of the whorls of the visceral mass to the mantle cavity. The distal portions of the male gonoduct, like those of the female, are visible, without dissection, in the mantle cavity (Fig 6).
The vas deferens becomes the seminal vesicle which enters the posterior left side of the mantle cavity and passes to the right under the epitaenium (Fig 6). Spermatozoa are stored in the seminal vesicle.
To the right of the epitaenium the male duct becomes the prostate gland. This is a low long mound on the far right side of the floor of the mantle cavity and is the distal end of the male gonoduct. The male gonopore opens atop a papilla at the end of the prostate gland.
The unusual penis and penis sheath characteristic of ampullariids develop from the mantle skirt (Fig 6). These are not present in this form in other prosobranchs. The penis sheath is an extension of the right side of the mantle skirt. It is a glandular tube which houses and guides the penis.
The penis is long and wormlike arising from a swollen bulb at the base of the sheath but if it is retracted it may not be apparent. Look for a white thread extending from the penis sheath and use forceps to pull its full length out of the sheath. The rectum and anal papilla lie beside the penis bulb.
Excretory System
The single kidney, or nephridium, is divided into anterior and posterior lobes in ampullariids. Both parts are derived from the left metanephridium of the ancestral prosobranchs.
The anterior kidney lobe is in the visceral mass at the posterior end of the mantle cavity. In life it is pink or pale brown and is visible from within the mantle cavity and also on the outside of the visceral mass (Figs 2, 4, 5, 6). The large, slit-shaped nephridiopore on the surface of the anterior kidney opens into the posterior mantle cavity. It is hidden in a deep posterior recess.
The posterior kidney lobe is beige or brown and much larger than the anterior lobe. It is visible externally on the left side of the visceral mass, posterior to the anterior kidney (Figs 2, 4, 5, 6).
Hemal System
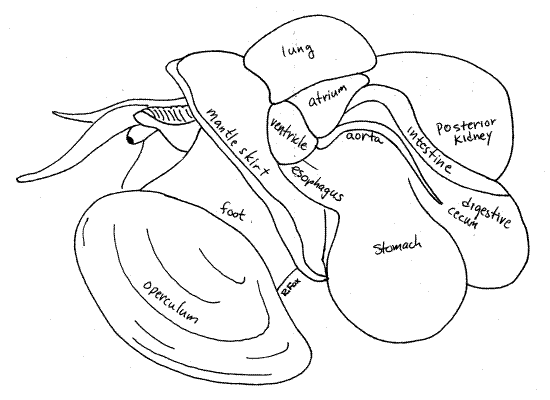
The pericardial cavity is on the left surface of the visceral mass posterior to the mantle cavity. The two-chambered heart is contained within it and may be visible if the body wall is not heavily pigmented (Fig 3, 6). The pericardial cavity is a coelomic space and as such is enclosed by a membranous mesothelium, the pericardium.
" With your fine scissors open the pericardium to reveal the heart. The pink or beige atrium is adjacent to the kidney and the bright white ventricle is ventral to the atrium (Fig 3, 6). The atrium is attached to the anterodorsal wall of the pericardial cavity by the three vessels which deliver blood to the atrium. These vessels, which drain the gill, lung, and kidney, resemble the walls of the atrium and are not immediately recognizable as vessels. Blood from the gill, lungs, and kidney drains into the atrium from these vessels and passes from there into the ventricle.
The large transparent common aorta exits the posteroventral corner of the ventricle and is the only exit from the heart. It divides immediately into the cephalic aorta to the hemocoels of the head and foot and the visceral aorta to the visceral mass. The latter is the white vessel on the left side of the visceral mass. Blood from the aortae empties into the sinuses of the hemocoel and then returns to the heart passing over the kidney and a respiratory organ. The aorta and arteries are usually bright white due to deposits of calcium carbonate in their walls. A large renopericardial pore opens from the dorsal wall of the pericardial cavity into the posterior lobe of the kidney. Primary urine formed by ultrafiltration of the blood into the pericardial cavity flows through the canal into the kidney. As the urine flows through the posterior and then anterior kidney lobes its composition is modified until it is released, as final urine, from the nephridiopore in the mantle cavity.
Digestive System
The small upper whorls of the visceral mass are occupied by the two glossy, black (sometimes yellow or brown, their color depends on diet) digestive ceca and the gonad (Figs 2, 3, 4). These organs are best seen on the right side.
The large muscular stomach is visible on the surface of the posterior left side of the visceral mass (Fig 3). The stomach is immediately posterior to the pericardial cavity and ventral to the posterior lobe of the kidney. Most of it is embedded in the digestive cecum and it is pink, beige, or white.
The rectum can sometimes be seen through the body wall crossing the visceral mass along the right side of the posterior kidney. Its color and visibility depend on its contents, if any.
Internal Anatomy
Digestive System
" With fine forceps lift the thin body wall on the midline of the top of the head and cut through it with fine scissors. Insert one blade of the scissors into the resulting opening and make a very shallow, longitudinal, median incision through the thin dorsal body wall of the head.
Avoid cutting the nerve ring immediately inside the body wall. The exposed, and thus vulnerable, portion of the ring is the white, straplike cerebral commissure running transversely exactly between the bases of the two ocular peduncles.
Extend the middorsal incision from the anterior end of the head posteriorly, along the floor of the mantle cavity, almost to the posterior end of the mantle cavity. The large space thus opened is the cephalic hemocoel, one of the major blood sinuses of the hemal system.
The bulbous muscular buccal mass occupies much of the space in the anterior end of the cephalic hemocoel (Fig 7). In life the muscles of the mass are pink due to the respiratory muscle pigment myoglobin. These muscles operate the radula and odontophore. The anterior gut passes longitudinally through the interior of the buccal mass. The esophagus exits the buccal mass about halfway along the length of the mass and lies on its dorsal midline (Fig 7).
" Insert the point of a pair of fine scissors into the mouth and make a longitudinal middorsal incision posteriorly for the length of the buccal mass (but avoid cutting the cerebral commissure). This will reveal and open the anteriormost region of the gut, the buccal cavity, within the mass. The mouth opens into the buccal cavity.
The radula is visible in a pouch, the radular sac, on the floor of the buccal cavity. It is a filelike ribbon of renewable chitinous teeth supported by a heavy connective tissue odontophore. The radula and the odontophore are operated by the numerous muscles of the buccal mass.
A pair of white and yellow jaws flank the anterior end of the buccal cavity but they may have been damaged when the hemocoel was opened. The lateral walls of the buccal cavity, beside the radula, are the strongly folded buccal glands of unknown function.
Figure 7. Dorsal view of cephalic hemocoel with the nervous system and anterior gut. B = buccal, C = cerebral, c = commissure, g = ganglion, Pe = pedal, Pl = pleural, Sb = (left) subesophageal, Sp = (right) supraesophageal, V = visceral, Z = zygoneury. Gastrop144L.gif
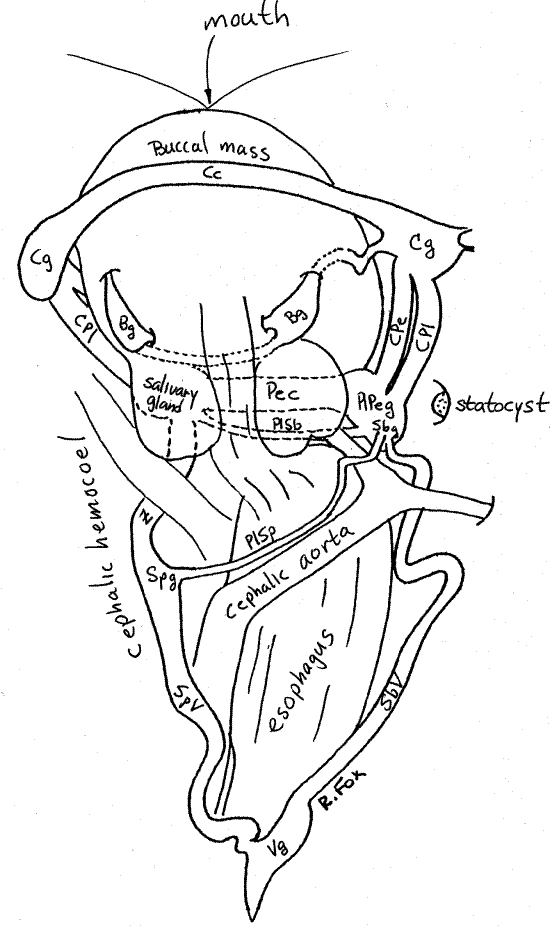
Carefully remove the mucus and debris from the vicinity of the radula. Make sure the water in the pan is clean and examine the radula with 30X of the dissecting microscope. Use fine forceps to move the radula while you watch its teeth. Count the teeth in each transverse row. _______ Mesogastropods, such as Pomacea, have taenioglossate radulae (Fig 12-40C) with seven teeth in each row.
>1c. Carefully remove the radula and make a wetmount for examination with the compound microscope. Observe the radular teeth. <
About halfway through length of the buccal mass, the buccal cavity narrows abruptly to become the thin-walled esophagus. The esophagus exits the buccal mass, widens, and continues posteriorly through the hemocoel (Fig 7).
A pair of yellowish salivary glands lies beside the anterior esophagus and connect, via ducts, with the buccal cavity (Fig 7). (The size and color of the salivary glands vary.) They presumably produce a lubricating mucus secretion.
The esophageal pouches are paired glandular pouches, of unknown function, opening laterally from the anterior end of the esophagus at the junction of the esophagus with the buccal cavity (Fig 12-39A). They lie ventral to the salivary glands and are hidden by them.
Follow the esophagus posteriorly from its beginning on the dorsal midline of the buccal mass. It passes posteriorly on the surface of the buccal mass, turns sharply ventrally, and then adopts a more or less horizontal orientation as it runs posteriorly through the cephalic hemocoel. It veers to the left of center as it passes posteriorly through the hemocoel.
Shortly after leaving the buccal mass the esophagus is crossed by the large cephalic aorta (Fig 7). The aorta has granular white walls and is the blood supply to the hemocoels of the head and foot. A slender, inconspicuous, white nerve cord (pedal-right esophageal connective) crosses the esophagus beside the aorta.
At the posterior end of the cephalic hemocoel the esophagus penetrates the wall of the hemocoel, enters the visceral mass, passes ventral to the heart, and opens into the stomach.
" Use fine scissors to open the esophagus and follow it to the beige or pinkish stomach. (This will destroy the visceral ganglion of the nervous system in the posterior hemocoel.)
" Open the stomach by cutting through its left wall, which is visible on the surface of the visceral mass. Extend this incision to follow the lumen of the stomach. It is U-shaped (a consequence of torsion) with both esophagus and intestine attached to its anterior ends. The "U" is oriented with its open end pointed anteriorly. The esophagus, ventral to the heart, is the lower arm of the "U" and the intestine the upper (Fig 3).
Wash the interior of the stomach with a few squirts of water from a pipet and examine its walls. They are exceedingly complex with elaborate sorting fields of ciliated ridges and grooves for sorting organic food from mineral particles. Ducts from the digestive ceca open into the right wall. Squeeze the digestive cecum in the vicinity of the duct to cause a cloud of fine black or brown particles to flow out of the duct into the stomach lumen. A tiny gastric cecum extends from the stomach. There is no style.
Find the intestine where it exits the stomach but do not attempt to trace it. After leaving the stomach the intestine makes a loop, turning sharply posteriorly to pass along the left border of the posterior kidney between the kidney and the stomach. This region is on the surface of the visceral mass and can be seen through the body wall. The intestine then turns to the right, still visible on the surface, and passes between the kidney and the digestive cecum. When it reaches the right side of the visceral mass it passes deep beneath the kidney and is lost to view.
While under the kidney the intestine makes several coils and then, as the rectum, exits the anterior visceral mass and extends diagonally across the roof of the mantle cavity to the anus on the right (Figs 5, 6).
" Open the anus and the rectum with a longitudinal incision made with fine scissors. Follow the lumen of the rectum posteriorly to the point where it emerges from the visceral mass. Note the large bilobed typhlosole, a longitudinal ridge on the inner wall of the rectum.
Nervous System
Ampulariids are good subjects for the study of the gastropod nervous system. They show most of the features of a typical bilaterally symmetrical molluscan nervous system but also illustrate well the streptoneurous, or twisted, asymmetrical nervous system that results from torsion. That most of the system is already exposed in the cephalic hemocoel, thus requiring no further dissection, makes the study relatively easy. Even so, careful examination of the gastropod nervous system may require more time than is available and this exercise may be omitted if desired by the instructor. The following is an account of the nervous system of the Florida apple snail, Pomacea paludosa. That of Viviparus is similar.
The molluscan central nervous system consists of several pairs of ganglia joined by cords, either commissures or connectives (Fig 7, 12-53, 12-54). The major ganglia of the molluscan nervous system are the paired cerebral, buccal, pedal, pleural, esophageal (=intestinal or parietal), and the unpaired visceral ganglia. Commissures are nerve cords between the ganglia of any pair. They are transverse and cross the midline. Connectives, on the other hand, are cords between ganglia of different pairs and are usually longitudinal. Commissures and connectives are named for the gangila they link; e.g. the buccal commissure connects the two buccal ganglia and the cerebrobuccal connective joins the cerebral and buccal ganglia. In some species the ganglia are red or orange, making them easy to see and recognize. This color, which is present only in living or fresh specimens, is due to the respiratory pigment, neuroglobin.
Cephalization is the tendency, displayed by molluscs, arthropods, vertebrates, and other animals, to concentrate ganglia anteriorly by shortening the connectives and commissures. The result is a brainlike accumulation of many ganglia often arranged in, or associated with, a nerve ring around the anterior gut. Such a centralized integration center has been exploited to great advantage by the cephalopods and to a lesser extent by the higher gastropods. The trend is exhibited to a small degree by Pomacea, a mesogastropod, in which the pleural and pedal ganglia are coalesced but for the most part the connectives and commissures remain relatively long and the ganglia widely separated. The tendency to coalesce the central nervous system into a brain is better expressed in neogastropods than in mesogastropods.
Direct your attention to the cephalic hemocoel and buccal mass in the head (Fig 7). You studied the mass earlier, at which time you were careful to avoid damaging the nerve ring. The ring encircles the anterior buccal mass and consists of the paired cerebral and pedal ganglia joined to each other by commissures and connectives. The two large white (sometimes red with neuroglobin) cerebral ganglia (Cg) lie on either side of the anterior buccal mass, between the bases of the cephalic tentacles. The cerebral ganglia are connected to each other by the broad, flat cerebral commissure (Cc), which passes dorsally over the buccal mass at the level of the cephalic tentacles. The intact cerebral commissure is conspicuous but it is easily cut when opening the hemocoel as it is close to the dorsal body wall.
Two triangular buccal ganglia (Bg) are located dorsal to the posterior buccal mass posterior to the cerebral ganglia. They are immediately anterior to the esophageal pouches and salivary glands. Each is joined to a cerebral ganglion by a cerebrobuccal connective. This connective exits the cerebral ganglion ventrally, passes into the muscle of the buccal mass and then emerges on the dorsal side of the mass just posterior to the cerebral commissure. The two buccal ganglia are connected to each other by the buccal commissure. This relatively short cord exits the buccal ganglion on its posterior, ventral, lateral corner, then curves under the esophageal pouches and esophagus to cross the midline to the other buccal ganglion. Note that it passes ventral to the gut.
The pleural (Pl) and pedal ganglia (Pe) are ventral to the buccal mass and lie to the side of it. They are a little posterior to the cerebral ganglia. The two ganglia of each side are fused and cannot be distinguished from each other. Together they are known as the pleuropedal ganglion (PlPeg) of which there is one on either side of the ventral buccal mass. The posterior portion of the pleuropedal ganglion is pleural, the anterior portion is pedal. The pleuropedal connectives between the pleural and pedal ganglia of each side are very short and embedded in the ganglionic mass where they cannot be seen.
Each pleural and pedal ganglion is connected to a cerebral ganglia by its own connective, either the cerebropleural (CPl) or cerebropedal (CPe), as the case may be, on each side. These are large connectives lying side by side lateral to the buccal mass. The cerebropleural connective is posterior to the cerebropedal. One of the muscles from the buccal mass to the lateral body wall passes between the two connectives.
Ventrally two transverse pedal commissures (Pec) connect the pedal ganglia. One is large and obvious, the other one small and inconspicuous) connect the right and left pedal ganglia to complete the nerve ring. The small one cannot be seen without additional dissection, which you should postpone. There is no pleural commissure.
The nerve ring consists of the cerebral ganglia, cerebral commissure, pedal ganglia, cerebropedal connectives, and pedal commissures (Fig 12-17B). The pleural ganglia and the cerebropleural connectives are closely associated with the ring but, strictly speaking, are not part of it.
A connective extends posteriorly from each pleuropedal ganglion to the single (unpaired) visceral ganglion (Vg) to form a closed circle, known as the visceral loop. This ganglion lies in the connective tissue forming the posterior wall of the cephalic hemocoel. It is not conspicuous and is best found by tracing the two arms of the visceral loop posteriorly to the point where they join. It is on the right side of the pericardial cavity and dorsal to the esophagus. It is immediately posterior to the point where the esophagus exits the hemocoel and turns sharply ventrally to enter the visceral mass. The visceral loop and visceral ganglia may have been destroyed by the earlier exploration of the digestive system. If so, the visceral loop cannot be studied on this specimen.
The parietal ganglia of the ancestral mollusc are given a variety of names in the gastropods including intestinal and esophageal ganglia by different authors. Since they are associated with the esophagus, we will use the term esophageal. The two inconspicuous, paired esophageal ganglia are located on the two connectives between the pleural and visceral ganglia in the visceral loop (Fig 7, 12-17B, 12-53). The locations of the two esophageal ganglia are asymmetrical due to torsion. The ancestral left esophageal ganglion is the subesophageal ganglion (SbV) of gastropods whereas the right esophageal becomes the supraesophageal ganglion (SpV). The supraesophageal ganglion is located halfway between the right pleural ganglion and the visceral ganglion. It is on the left of the esophagus. The subesophageal ganglion is fused with the right pleural ganglion.
The loop formed by the connectives linking the pleural, esophageal, and visceral ganglia is known as the visceral loop. Only the visceral loop is affected by torsion, the remainder of the nervous system being unaffected. On casual inspection the visceral loop of Pomacea will appear bilaterally symmetrical and will seem to be unmodified by torsion. This is due to secondary postorsional modifications that, in mesogastropods and neogastropods, restore an impression of bilateral symmetry. This will be discussed in more detail later.
Peripheral nerves to and from the body extend from the ganglia and connectives of the central nervous system. The larger ones are easily seen and, if desired, may be traced to the organs they serve. Tracing them is necessary to confirm their identity.
The many nerves of the cerebral ganglia serve the sensory structures of the head including the skin, labial tentacles, cephalic tentacles, statocysts, and eyes. The anterior buccal mass is also innervated by the cerebral ganglia. The tentacular nerve to the cephalic tentacle is the largest of the cerebral nerves. The optic nerve, which is smaller, arises beside it. The statocysts, which lie beside the pedal ganglia, are innervated by a nerve from the cerebral ganglion which passes ventrally with the cerebropleural connective.
Nerves from the buccal ganglia innervate the adjacent areas of the pharynx, esophagus, buccal mass, radular sheath, and buccal (salivary?) glands.
A complex of large nerves from the pedal ganglia supplies the musculature of the foot. These nerves leave the ventral border of the pedal (anterior) half of the pleuropedal ganglion and cannot be seen without further dissection (which should not be attempted at this time).
Nerves from the pleural ganglia extend to the body wall, mantle, siphons, osphradium, columellar muscle, penis, epitaenia, rectum, and distal gonoduct.
The esophageal ganglia innervate the gills, and osphradium.
The visceral ganglia innervate the heart, pericardium, kidney, most of the reproductive system, posterior gut, and digestive gland. Further study of the nervous system may be omitted if time is short.
The remaining consideration of the nervous system and the effects of torsion is more detailed than is usually attempted in introductory invertebrate zoology courses. Continue if specifically instructed to do so by your instructor, otherwise skip from here to the section on Sense Organs.
Effects of Torsion on the Nervous System
In gastropods, torsion twists the visceral mass 180° counterclockwise with respect to the head and foot, which are themselves unaffected by torsion (Fig 12-16).The nerve ring and pedal nervous system, located as they are in the head and foot, are not changed by torsion. The visceral loop, on the other hand, extends from the unaffected part of the body up into the twisted part and consequently is itself twisted by torsion (Fig12-17A,B). After torsion, the loop has the shape of a figure 8 and is asymmetrical (Fig 12-53). This condition is said to be streptoneurous (strep = twisted) whereas the original symmetrical condition is euthyneurous. Prior to torsion the entire visceral loop is ventral to the gut. After torsion part of it is dorsal to the gut and some ganglia that were formerly on the left are now on the right and vice versa.
Review the locations of the cerebral ganglia, cerebral commissure, buccal ganglia, cerebrobuccal connectives, buccal commissure, fused pleuropedal ganglia, cerebropedal connectives, and cerebropleural connectives. Locate the large, broad pedal commissure connecting the two pedal ganglia ventral to the gut. It is, in fact, only one of two pedal commissures but the second is much smaller and more difficult to find. The large one, known as the primary pedal commissure, obscures your view of the secondary pedal commissure and you may not find the latter unless you remove the buccal mass. The primary commissure is dorsal to the secondary. All the above structures, located as they are in the head and foot, are unaffected by torsion and are bilaterally symmetrical.
As already discussed, a pair of long, longitudinal connectives arises from the pleural ganglia and runs posteriorly, through the esophageal ganglia, to the median, unpaired visceral ganglion and form the visceral loop. The visceral loop, including the pleural ganglia, intestinal ganglia, visceral ganglia, and their connectives, is twisted into a figure 8 by torsion.
Study the visceral loop carefully, being watchful for the effect of torsion (and for subsequent modifications to reverse its effects). Torsion places the pretorsional right pleuro-visceral connective and the right esophageal ganglion dorsal to the gut where the ganglion becomes known as the supraesophageal ganglion. In Pomacea it is located about a third of the way along the left arm of the visceral loop. It is an inconspicuous swelling located at the junction of three connectives (which will be named later). At the same time the left pleurovisceral connective and the left esophageal ganglion move right and ventral to the gut and the ganglion becomes the subesophageal ganglion. In Pomacea it is fused with the right pleural ganglion. The posterior gut, which is bent into a U-shape, is also twisted around its long axis so its ventral side becomes dorsal and vice versa. In consequence, the visceral ganglion comes to lie dorsal to the gut rather than ventral to it.
Look for the short pleuro-subesophageal connective extending transversely from the left pleural to the subesophageal ganglion. It is posterior to the large dorsal pedal commissure and is easily seen. It passes ventral to the esophagus but you can see it by pushing tissues aside. It forms a shallow semicircular loop curving from one side to the other. It is the homolog of the left pleuroparietal connective, which in untorted molluscs runs posteriorly to the subesophageal ganglion (= left parietal ganglion). In untorted molluscs the subesophageal ganglion is on the left and the connective runs from the left pleural ganglion posteriorly to it. In Pomacea, however, the ganglion is moved to the right and is fused with the right pleuropedal ganglion. The subesophageal connective runs from the left pleural ganglion, ventral to the gut, to the subesophageal ganglion attached to the right pleuropedal ganglion. It still runs from the left pleural ganglion to the subesophageal ganglion but the position of the latter is changed so it is now on the right rather than the left. From the subesophageal ganglion (in the pleuropedal mass) there arises the large subesophageal-visceral connective which runs posteriorly along the right side of the hemocoel to the visceral ganglion. It follows a looping, sinuous path along the wall of the hemocoel, passing ventral to the cephalic artery just after it (the nerve) exits the ganglion. It is free in the hemocoel but is bound to its right wall by connective tissue which obscures your view of it. It does not penetrate the hemocoel wall. It is the homolog of the pretorsional left parietovisceral connective.
Also arising from the right pleural ganglion, but running obliquely across the hemocoel, dorsal to the esophagus, is the pleuro-supraesophageal connective (=supraesophageal nerve). It exits the pleural ganglion, then crosses dorsal to the cephalic aorta where the aorta lies on the right side of the esophagus. The nerve then lies beside and parallels the cephalic aorta as the aorta crosses the esophagus. (Torsion is the reason the aorta and connectives cross the midline.) It runs to the inconspicuous supraesophageal ganglion which, after torsion, lies on the left even though it is homologous to the right parietal ganglion. It is a small swelling on the visceral loop. The supraesophageal ganglion is connected to the visceral ganglion by the supraesophageal-visceral connective on the left side of the hemocoel. This connective is homologous to the right parietovisceral connective but torsion has moved it to the left. Its position more or less mirrors that of the subesophageal-visceral connective on the right side. Can you, with a little imagination, see the figure 8 of the torted gastropod visceral loop? (The extreme top transverse bar of the "8" is the pedal commissure, which is not actually part of the visceral loop.)
Torsion leaves an apparent gap in the visceral loop between the left pleural ganglion and the supraesophageal ganglion that destroys the appearance of symmetry. There is a tendency in the mesogastropods and neogastropods to reestablish the bilateral symmetry destroyed by torsion with a new connective, the left zygoneury (Z). This connective runs from the left pleural ganglion to the point where the pleural-supraesophageal connective joins the supraesophageal ganglion. It is a large connective larger than the subesophageal-visceral connective in the similar position on the right side. It is usually hidden from view by a thick layer of connective tissue. Such a connection was not present in the ancestral mollusc. The supraesophageal ganglion is located at the junction of the left zygoneury, right pleuro-supraesophageal connective, and supraesophageal-visceral connective. There is also a right zygoneury between the right pleural ganglion and the subesophageal ganglion but it is embedded in the right pleuropedal ganglionic mass and cannot be seen.
Sense Organs
The labial and cephalic tentacles are mechanoreceptors, sensitive to tactile stimuli, and are probably also chemoreceptive (Fig 2). The general body surface is also sensitive to chemical and mechanical stimuli.
The paired eyes are photoreceptors but are not capable of forming an image. They are borne distally on the ocular peduncles (Fig 2).
The osphradium is located in the inhalant respiratory water current where it monitors water on the way to the gill (Figs 5, 6). It is chemosensory but probably is also a mechanoreceptor capable of detecting silt.
The statocysts are organs for the detection of gravity. Each is a large, yellowish, spherical vesicle lateral to a pleuropedal ganglion in a deep pocket in the floor of the hemocoel (Fig 7). The vesicle contains a statolith composed of calcium carbonate granules.
Behavior
Pomacea is usually slow to emerge from its shell when in a small dish making behavioral observations difficult. It is better to watch undisturbed snails in an aquarium. Find a snail adhering to the glass so you can see its extended foot. Observe the head and watch the activity of the sensory equipment (labial and cephalic tentacles and eyes). Find the mouth and examine it with a 10X hand lens. You should see the radula rasping algae from the aquarium walls. Note the inhalant siphon on the left side as an intake for water or air. The snail may approach the water surface and extend the siphon above the surface. If this happens inform your instructor.
References
Andrews EB. 1964. The functional anatomy and histology of the reproductive system of some pilid gastropod molluscs. Proc. Malac. Soc. London 36:121-140.
Annandale N, Seymour Sewell RB . 1921. The banded pond-snail of India (Vivipara bengalensis). Rec. Indian Mus. 22:215-292, pls. 1-3.
Applesnail Web Page www.applesnail.net
Baker FC. 1928. The freshwater Mollusca of Wisconsin, Part I. Gastropoda. 507 p. Wisconsin. Aca. Sci. Arts Lett., Madison.
Cal, R . 1888. Gross anatomy of Campeloma. American Natur. 22:491-497, pl. 7.
Chang M. 1928. The anatomy of fresh-water viviparous snails. Peking Soc. Nat. Hist. Bull. 3:45-57, pls. 1-2.
Cook PM. 1949. A ciliary feeding mechanism in Viviparus (L.). Proc. Malacological Soc. London , 27:265-271.
Ghesquiere S. 2001. A rotatable 3-D model of Pomacea bridgesi.
www.applesnail.net/content/3d_models/pomacea_bridgesi.htm
Hyman LH . 1967. The Invertebrates, Mollusca, vol. VI. McGraw-Hill, New York. 792p.
Li F-C. 1935. Anatomie von Paludina. Chinese J. Zool. 1:1-18.
Mattox N. 1938. Morphology of Campeloma rufum. J. Morph. 62:243-258, pls. 1-2.
Pennak RW . 1989. Fresh-water Invertebrates of the United States, 3 rd ed. Wiley, New York. 628p.
Prashad B. 1925. Anatomy of the common Indian apple-snail, Pila globosa. Mem. Indian Mus. 8:91-149, pls. 16-18.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Vermeire, P. E. and G. W. Hinsch . 1984. Structure of the female reproductive tract of the apple snail II. Scanning electron microscopy. Tissue Cell 16:107-114.
Supplies
Dissecting microscope
Large apple snail
Small dissecting pan
# 4 stainless steel insect pins
Dissecting set with microdissecting tools
Plastic Pasteur pipet
Freezer