Invertebrate Anatomy OnLine
Philodina ©
Rotifer
5jul2006
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises, a glossary, and chapters on supplies and laboratory techniques are also available at this site. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Gnathifera SP, Syndermata, Rotifera P, Bdelloidea C, Philodinidae F (23-26, 9-26)
Gnathifera SP
Gnathiferans have a complex pharynx with a cuticular jaw apparatus operated by an elaborate pharyngeal musculature. Most are small, less than 1 mm. The taxon includes Rotifera, Gnathostomulida, Micrognathozoa, Acanthocephala, and Seisonida.
Syndermata
Rotifers, seisonidans, and acanthocephalans share a syncytial epidermis with an cytoplasmic skeleton known as the intrasyncytial lamina (Fig 23-12).
Rotifera P
Rotifera includes some of the smallest metazoans and most of the 2000 described species are less than 0.5 mm in length. Most are microscopic, freshwater, and free swimming. These tiny animals superficially resemble ciliate protozoans in size and behavior but are multicellular, with about 1000 cells each.
Rotifers are common animals that can be found in almost every conceivable freshwater habitat including such obvious locations as lakes and rivers but also in places that are moist only periodically, such as moss, soil, and leaf litter. They are the most abundant metazoans in freshwater benthic habitats and are one of the three dominant metazoan taxa in the freshwater zooplankton. Rotifers occur at all except polar latitudes. Most inhabit freshwater habitats although about 50 species are marine. Two genera are exclusively marine.
Rotifers have a fluid filled body cavity containing a loose syncytium of amoeboid cells. The body cavity is a hemocoel, or pseudocoel, derived from the blastocoel of the embryo. There is no mesothelium. These animals are eutelic and individuals of each species have a characteristic and constant number of cells. Tissues are often syncytial. The characteristic number of cells originates in the embryo and remains for life with growth occurring through increase in cell size, rather than through addition of new cells.
Rotifers have characteristic ciliary ring, or corona, near the anterior end to generate the feeding and locomotory current. The body wall consists of a syncytial epidermis, containing an intrasyncytial lamina, known as the lorica, and underlain by individual circular and longitudinal muscles. The lorica may be thin and flexible or thick and rigid. The gut is usually, but not always, complete and the muscular pharynx includes an elaborate, cuticular mastax with up to seven hard jaws, or trophi. Osmoregulation is via protonephridia. No special respiratory or hemal systems are present. Gas exchange is accomplished over the unelaborated body surface and fluid transport by diffusion and movement of the blood in the hemocoel.
The nervous system includes a dorsal cerebral ganglion, two pairs of longitudinal nerve cords, a mastax ganglion, and a caudal ganglion. The sense organs include eyes, bristles, pits, and antennae. Rotifers are gonochoric but, depending on taxon, males are rare, unknown, or produced only occasionally.
Reproduction is usually parthenogenetic but is sometimes sexual in those monogonont species with both sexes. Males, when present, are smaller than females, short-lived, aberrant or degenerate, and usually have no functional gut and do not feed (Fig 23-20A). Females have a gonad consisting of a combined germarium (ovary) and vitellarium (yolk gland). Descriptions of rotifer anatomy and biology, including this one, apply to females unless stated otherwise. Most rotifers are oviparous but a few planktonic species are viviparous.
Bdelloidea
Bdelloids inhabit freshwater and most are found in intermittently moist terrestrial mosses and soil but some are benthic in aquatic habitats. Those that inhabit terrestrial habitats are tolerant of desiccation. Male bdelloids have never been observed and are presumed to be nonexistant. Reproduction is always parthenogenetic. Females have two germovitellaria as opposed to the solitary one of Monogononta. The contractile, wormlike body is elongate and composed of 16 telescoping rings. The head is retractable and has a well developed corona with two trochal discs. The mastax is adapted for grinding.
Laboratory Specimens
Philodina is a more or less typical rotifer although its mastax does not exhibit the ancestral plan. It is exceedingly abundant and has been called the world's most common metazoan (Hyman, 1951).
This exercise is written for living specimens. Cultures of living Philodina are available inexpensively from biological supply companies. The study should be conducted on wetmounts of living female specimens. Males are not known for any bdelloid rotifer, including Philodina.
External Anatomy
Place a drop of Protoslo on a slide, add a tiny bit of debris from the bottom of a culture jar of Philodina, and mix. Apply a coverslip equipped with wax feet (see Techniques chapter) on two adjacent corners and press it into the liquid so the animals are provided with a thin film of water but are not squeezed. Use the scanning lens of the compound microscope to find a clump of debris with rotifers attached. Alternate between 100 and 400X for the rest of your observations. Most specimens will move occasionally, giving you views from several angles but you will need to study several specimens to see everything.
Rotifers are active animals and are difficult to observe when swimming. Some of your specimens will probably be attached to debris or the glass and these will be the easier to observe. Find a quiet, attached specimen and observe it. If it swims away during the observation be prepared to follow it or find another. This will probably happen several times.
Lorica
The body is elongate, widest in the middle, and strongly tapered posteriorly. It is composed of a short anterior head, long, thick, middle trunk, and short, narrow, posterior foot (Fig 1, 23-17).
The body is enclosed in a well-developed intraepidermal lorica, produced by and contained within the cytoplasm of the syncytial epidermis. The many longitudinal and circular muscles in the body wall are not arranged in distinct layers.
The body cavity is a large, spacious hemocoel (= pseudocoel) filled with fluid and cells. It is derived from the blastocoel and lacks a mesothelium. Neither the muscles nor hemocoel are visible in wholemounts.
The body is encircled by about 16 circumferential rings, or annuli, and consequently appears to be segmented but the divisions are confined to the lorica (which is intraepidermal) and are superficial (Fig 1, 23-17). The annuli are of different diameters and allow the body to shorten by telescoping. If you are observing a living specimen, you will see this occur.
The smaller annuli of the head and foot telescope into the larger ones of the trunk so that the head and foot can be completely retracted into the trunk. Telescoping is accomplished by contraction longitudinal muscles, which have their origins and insertions on the inner surface of the epidermis (lorica) (Fig 23-13).
Head
The head consists of the anterior six annuli but they are difficult to count (Fig 1). The head narrows posteriorly to become a short neck which joins the wider trunk.
At its apparent anterior end the head bears a conspicuous and characteristic array of cilia known as the corona. The corona is completely retractable into the trunk and is not visible when retracted. Find an animal with its corona expanded and study it. Swimming specimens always have the corona expanded but they are difficult to observe.
The pattern formed by the cilia of the corona varies with taxon but that of Philodina is composed of two ciliary rings around the anterior body (Fig 1, 23-17). The anterior ring, known as the trochus, is divided into right and left circles of cilia, each of which is known as a trochal disk. When beating, the disks resemble rotating wheels.
The second ciliary ring of the corona is the cingulum, which encircles the body just posterior to the trochus and mouth. The trochus and cingulum may be difficult to distinguish from each other. The trochus is composed of large cilia, often organized into membranelles, whereas the cilia of the cingulum are much smaller.
Figure 1. Ventral view of a female Philodina from a commercial culture. Rotifer69La.gif
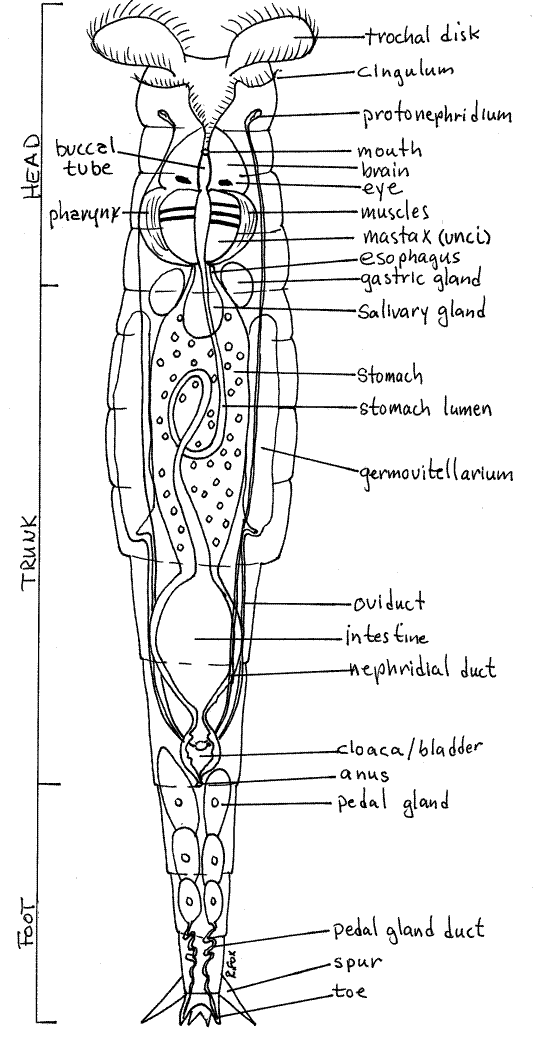
The corona generates the feeding and locomotory current. The trochal disks are the source of the older common name "wheel animacules" and the scientific name "Rotifera". Watch the trochal disks and note the synchronized waves of ciliary motion which move in circles around each wheel. When the animal is attached and stationary, the cilia draw a feeding current of water and suspended particles to the mouth, which is located between the two trochal disks. When the rotifer releases its hold on the substratum, the beating of the cilia pull it rapidly through the water.
When the corona is deployed, it appears to be the extreme anterior end of the body but in fact it is on the third annulus of the head and there are two annuli anterior to it. These two anterior annuli form the rostrum which is much narrower than the rest of the head and can be seen protruding anteriorly when the corona is retracted (Fig 23-13). It bears sensory bristles and cilia.
Posterior to the corona the dorsal midline of the head bears a slender, sensory dorsal antenna (Fig 23-14C). It is visible in side view but undetectable in ventral and dorsal views. The antenna is always visible if the animal presents its side view and is much easier to see than the rostrum. It is longer and thinner than the rostrum. The large retrocerebral apparatus (Fig 23-9) in the head open dorsally. Its probably secretes mucus but is difficult to see.
Trunk
The trunk is the wide, long, middle region of the body plus the first two annuli of the tapered posterior body (Fig 1). It begins with the first wide annulus posterior to the narrower head and extends posteriorly to the anus. The trunk consists of six annuli with the anus is on the sixth. Most of the viscera are contained in the hemocoel of the trunk. The annuli of the head and foot telescope into the trunk to shorten the animal.
Foot
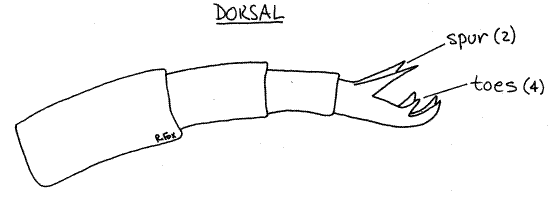
The foot extends posteriorly from the trunk and is the region of the body posterior to the anus. It tapers posteriorly and consists of four annuli. The posterior annulus of the foot bears a pair of dorsal spurs and four small, sticky toes (Fig 1, 2, 23-8A). The spurs are always evident and conspicuous but the toes are usually hard to find. The spurs are dorsolateral and the toes posterior to them. Pedal glands in the foot secrete adhesive, which is released from the tips of the toes and used to stick temporarily to the substratum.
Most of the time the toes are either retracted, or attached to the glass of the slide or coverslip, or oriented in such a way that you cannot see them clearly. They are best glimpsed during the brief intervals when the animal repositions its foot (Fig 2). In doing this the rotifer releases the toes, extends them fully, reattaches them, and then partly retracts them. Watch for them during the brief moment when they are reextended.
The animal uses its toes to attach temporarily to the substratum. With low power watch a moving rotifer use its toes to attach to the glass or debris.
Internal Anatomy
Find an animal that is quiet and relatively easy to observe and study its interior using 400X and Figure 1.
Digestive System
The gut is complete and straight with anterior, ventral mouth and posterior dorsal anus. There are muscles in the gut wall.
Figure 2. The foot of Philodina in side view. Rotifer70L.gif
The mouth opens into a short, narrow, buccal tube (Fig 1, 23-8B, 23-9) with cuticularized ciliated walls. In immobile animals the cuticular walls look like a pair of closely spaced parentheses () whose concave surfaces are ciliated.
Posteriorly the buccal cavity joins the muscular pharynx (Fig 1, 23-8A,B, 23-9, 23-15A,C). This large conspicuous organ is elaborated to form a complicated cuticular mastax, or jaw apparatus, embedded in a powerful muscular bulb (Fig 23-15B). The pharyngeal lumen in its interior is narrow and lined with cuticle. The rotifer mastax is composed of seven cuticular elements, known as trophi (Fig 23-15B). The structure of the trophi varies markedly with species and is correlated with feeding mode. Those of Philodina are adapted for grinding and are dominated by a pair of flat, ridged plates, the unci. They are examples of the ramate condition. The unci rub against each other to grind food particles.
In living specimens the mastax is easily seen in the interior of the posterior head on the anterior edge of the trunk. It is continuously active and its two flat unci can be seen moving conspicuously. The mastax is unique to rotifers although seisonidans, gnathostomulids, and micrognathozoans have similar structures. It is used to process food and in other species may be specialized for grinding, grasping, sucking, or cutting.
A few secretory cells, known as salivary glands, are associated with the pharynx (Fig 1, 23-8A) . Their function is unknown but they are suspected of secreting hydrolytic enzymes.
A short, narrow, ciliated esophagus connects the pharynx with the enormous stomach, which occupies much of the interior of the trunk (Fig 1, 23-15A, 23-8). The stomach of Philodinaand its relatives is a thick, syncytial mass with a narrow lumen. The lumen loops through the syncytium and its walls are ciliated. Digestion and absorption occur in the stomach, whose walls typically contain colored inclusions, granules, and lipid droplets. A pair of secretory gastric glands on the anterior edge of the stomach discharge hydrolytic enzymes into the stomach (Fig 1, 23-8A, 23-15A).
Following the stomach is a short but wide intestine in the narrow, posterior end of the trunk (Fig 1, 23-8A, 23-15A). It opens to a small cloaca which, in turn, opens to the exterior via the middorsal anus at the anterior end of the foot. Undigested material from the stomach is moved to the exterior through the intestine, cloaca, and anus.
>1a. Make a wet mount using a tiny speck of yeast/Congo red (see Supplies chapter) with a coverslip but without Protoslo. Search the preparation with 100X of the compound microscope to find a specimen with corona deployed and beating, then switch to 400X. Be prepared to locate new specimens if the one originally selected stops feeding or swims away.
The red yeast particles make the feeding current easy to observe. Note the strength of the current and its path. The position of the mouth can now be determined precisely. What happens to the yeast cells in the feeding current? Are all ingested? Does there appear to be any mechanism for particle rejection?
Watch yeast cells as they pass the mastax and accumulate in the stomach. As the colored cells accumulate, the gut lumen becomes clearly visible and you can ivsualize its looping path through the stomach. It will take about 15-30 minutes for the stomach and intestinal lumina to fill with red cells. You may want to go on with the exercise, using either of your two wetmounts, and return occasionally to note the progress of the cells moving through the gut. Don’t let the wetmounts dry out while you are waiting. <
Respiratory and Fluid Transport Systems
No special respiratory surface exists in these tiny animals nor is there a hemal system. Gas exchange occurs across the general body surface. Transport of oxygen and nutrients is accomplished by diffusion and movement of fluid in the hemocoel. For animals such as these, less than 1mm in length, diffusion is adequate to transport materials through the body but movement of the fluid in the hemocoel may assist.
Excretory/Osmoregulatory System
As freshwater animals with a permeable integument, rotifers have severe osmoregulatory problems with the influx of water from their hyposmotic enviromnent and must have some method of eliminating the excess water that floods the body. Lacking a pressurized hemal system, they are denied metanephridia. Instead, a pair of flame bulb protonephridia in the hemocoel eliminates excess water from the body. Each consists of numerous terminal cells and the system is syncytial.
Look for the flickering cilia of the terminal cells in the sides of the head (Fig 1). You may find it helpful lower the coverslip a little so the specimen is squeezed slightly. The terminal cells drain into a pair of lateral longitudinal ducts which empty into the bladder ventral to the cloaca. Periodic contractions of the cloaca force the urine out the anus. Large volumes of water move through this system in freshwater rotifers to counteract the osmotic influx across the body surface. Excretion of nitrogenous wastes, as ammonia, probably takes place across the permeable body surface but the protonephridia may also play a role.
Find an immobile specimen for closer observation. Locate the bladder in the anterior end of the foot and watch it for a few minutes (Fig 1, 23-8A,B). You should be able to see it expand as it fills with water and then suddenly contract as it empties. This cycle of filling and voiding may occur as often as once every 15 seconds. Rotifers pump out a volume of dilute urine equal to their own volume every 10 minutes.
Nervous System
The nervous system consists of a large dorsal brain (Fig 1, 23-18) and a pair of ventral longitudinal nerve cords. The brain can be seen with good light and careful focusing just anterior to the mastax.
Two reddish-orange eyes are present, but not always visible, dorsal to the brain. The dorsal antenna, which you saw earlier, is a tactile sense organ as are the bristles and cilia on the rostrum. The antenna can be recognized only in side view.
Locomotion
Philodina swims using its corona and does so frequently but it more often creeps along a hard substratum with its corona retracted. It uses its toes and rostrum, at opposite ends of its body, as anchors for temporary attachment. It creeps like a leech, alternately attaching and releasing the anterior and posterior ends (Fig 23-14C). This leech-like locomotion is the inspiration for the name bdelloid (bdell = leech). Scan your slide for an individual engaged in creeping and observe it. If you have not already done so, watch a swimming individual also.
Pedal glands fill the interior of the foot and connect via convoluted ducts with the tips of the toes (Fig 1). These glands secrete the glue that sticks the toes to the substratum. The toes can be released at will although the releasing mechanism is not known. There apparently are no duoglands in rotifer toes.
Reproduction
Rotifers are gonochoric but, like other bdelloids, Philodina has no males and reproduction is parthenogenetic. The females are oviparous. The gonads of bdelloids are two large, lateral germovitellaria located beside the stomach in the trunk (Fig 1, 23-8A,B). Each is composed of an inconspicuous germarium (or ovary), for the production of ova, and a vitellarium, for yolk production (Fig 23-19). The yolk is incorporated into the cytoplasm of the developing ovum.
In all rotifers the germovitellarium is a syncytium consisting of the nuclei and cytoplasm of cells which have lost the intervening cell membranes. It is a good example of a strongly eutelic organ with syncytial organization. An inconspicuous oviduct runs from each germovitellarium to the cloaca.
Commercial cultures usually include numerous ovoid eggs containing embryos in various stages of development. You can probably see a moving mastax in some of the more mature embryos.
Planktonic Rotifers
Rotifers, cladocerans, and copepods are the major components of the freshwater zooplankton. Whereas most rotifers are benthic, about 100 species of monogonont rotifers are free-swimming and planktonic. Most monogononts, however, are benthic. Philodina is a typical benthic bdelloid species.
Planktonic rotifers tend to be are modified for life suspended in the water remote from firm substrata. They often have a reduced lorica, frequently have no toes, and usually have some adaptations to keep their eggs from sinking to the bottom. Females usually hold the eggs attached to their own body, gestate them internally, or attach them to other rotifers. A plankton tow in almost any body of fresh water will yield several species of rotifers.
>1b. If fresh plankton samples are available, make a series of wetmounts for examination with the compound microscope. Identify the rotifers you find to genus using the references available in the laboratory. <
Moss Rotifers
Terrestrial bdelloids undergo cryptobiosis and withstand extended periods of desiccation and return to metabolic activity when wetted. This ability makes it possible for them to inhabit bryophytes (mosses) in terrestrial habitats that are only periodically wet. During wet conditions the rotifers inhabit the thin film of water on the surface of the moss. Here they feed and complete the parthenogenetic life cycle. The well developed foot and toes maintains contact with the moss substratum. They are elongate and vermiform to facilitate maneuvering in the tiny spaces among the moss "leaves".
>1c. Take a small sample of moss, perhaps collected from a nearby tree, and place it in an 8-cm culture dish and completely cover it with spring water. Deionized water or lake water is acceptable but do not use tap water. Examine the submerged moss with the highest power of the dissecting microscope for signs of activity. You probably will not see anything. Set the dish aside for 15-30 minutes and look once more at the moss with the dissecting microscope. This time you should see active bdelloid rotifers.
Using 40X find and watch a rotifer for a few minutes. Observe the way it uses the toes at the posterior end of the foot to attach to and move over the substratum (Fig 23-14C). The rotifer may move in a leech-like looping manner in which it alternately attaches and releases the posterior and anterior ends.
Dishes that have been standing for several days may have rotifers concentrated on surface tension which they use as a substratum. These rotifers are especially easy to observe as they move leech-like over the under side of the surface tension.
If desired, a wetmount may be prepared and the rotifers studied with the compound microscope. <
References
Clement P, Wurdak E . 1991. Rotifera, in Harrison FW, Ruppert EE (eds.) . Microscopic Anatomy of Invertebrates vol. 4 Aschelminthes . Wiley-Liss, New York. 424p.
Donner J. 1966. Rotifers. Warne, London. 80p. Translated by H.G.S. Wright.
Hyman LH. 1951. The Invertebrates: Acanthocephala, Aschelminthes, and Entoprocta. The coelomate Bilateria, vol. III. McGraw-Hill, New York. 572pp.
Pejler B. et al (eds.) 1983. Biology of Rotifers. Hydrobiologia 104:1-396.
Pennak RW . 1989. Fresh-water Invertebrates of the United States, 3 rd ed. Wiley, New York. 628p.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Ruttner-Kolisko A. 1974. Plankton rotifers, biology and taxonomy. Binnengewasser 26 supplement:1-146.
Wallace RL, Snell TW. 2001. Phylum Rotifera, pp 195-254 in Thorp JH, Covich AP (Eds), Ecology and classification of North American freshwater invertebrates, 2 nd ed. Academic Press, San Diego. 1056 pp.
Supplies
Compound microscope
Rotifer culture (Philodina)
Yeast/Congo red suspension
Moss
Dissecting microscope
Slides and coverslips