Invertebrate Anatomy OnLine
« Peripatus » ©
Velvet worm
18Jun2006
Copyright 2005 by
Richard Fox
Lander University
Preface
This is an exercise from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises can be accessed by clicking on the links to the left. A glossary and chapters on supplies and laboratory techniques are also available through these links. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Panarthropoda SP, Onychophora P, Udeonychophora C, Euonychophora O, Peripatopsidae F
Panarthropoda SP
Panarthropoda includes Onychophora, Tardigrada, and Arthropoda. These taxa share segmentation, a hemocoel, saccate nephridia, ecdysis of a secreted chitinous but non-collagenous exoskeleton, loss of locomotory cilia, a tubular, dorsal, ostiate heart in a pericardial sinus, a coelom reduced to end sacs and gonocoel, and paired segmental legs.
Onychophora P
Onychophorans are also known as velvet worms or walking worms and sometimes the original generic name Peripatus is used as a common name, in which case it is not capitalized or italicized (Fig 15-1A). Velvet worms are wormlike, tropical or temperate southern hemisphere terrestrial panarthropods. Onychophorans are closely related to arthropods and have more distant connection with annelids and have characteristics of both. Their modern distribution indicates an earlier confinement to Gondwana.
The body surface is dry, hydrophobic, and velvety, hence the common name “velvet worm”. The three segmented head bears three pairs of appendages; antennae, mandibles, and oral papillae (Fig 15-1B). The trunk has 13-43 paired segmental legs. These are soft fleshy lobopods without articulations or joints. Because these legs resemble the fleshy, unjointed prolegs of caterpillars, onychophorans resemble these lepidopteran larvae, although they have far more legs than do caterpillars. The legs terminate in claws and are sometimes referred to as onychopods (onych = claw, phor = to carry, pod = foot). Tiny spiracles open into tufted tracheae which deliver oxygen to the tissues.
The exoskeleton (= cuticle) is arthropod-like, and is periodically molted, although thin and largely unsclerotized. The cuticle consists of an outer epicuticle and inner procuticle with the procuticle divisible into endocuticle and exocuticle. The cuticle contains a -chitin and protein but lacks collagen. These cuticular characteristics are apomorphies shared with Arthropoda P. The remainder of the body wall, however, is annelid-like. Sclerotization is minimal, sclerites and articulations (joints) are absent, and the cuticle is soft. The body wall contains layers of unspecialized circular and longitudinal muscles. The body cavity is a blood-filled hemocoel, like that of arthropods and unlike annelids. The hemocoel is divided into a dorsal pericardial sinus containing the long tubular heart and a ventral perivisceral hemocoel containing the viscera as is that of arthropods.
The two widely separated cords of the double ventral nerve cord are connected by multiple commissures in each segment. Segmentation is apparent in the arrangement of limbs, ostia, crural glands, coxal organs, and saccate nephridia. The ganglia, although paired and segmentally arranged, are not swollen and are not visible in gross examination.
Almost 200 species in 70 genera and two families have been described. Many more are thought to await discovery. Australia has the greatest diversity, currently with over 40 described species and counting. Onychophorans have been thought to be a link between annelids and arthropods and recent studies show them to be closer to arthropods.
Families
Onychophora comprises two extant families, Peripatopsidae and Peripatidae.
Peripatopsidae, with about 50 known genera, is restricted to temperate regions of the southern hemisphere including Chile, Australia, New Zealand, Tasmania, and South Africa. Peripatopsids are blue, grey, green, or sometimes brown. These colors persist in preservative. The trunk has 13-25 pairs of legs. The gonopore is between the last pair of legs.
Peripatidae, with about 10 known genera, has a circumtropical distribution including the Caribbean islands, Central America and southern Mexico, tropical South America, Malaysia, and the Congo Basin of Central Africa. Peripatids are usually reddish brown but the color is water-soluble and fades in preservative. The trunk has 22-43 pairs of legs. The gonopore is between the penultimate pair of legs (Fig 15-1C). The family includes Peripatus.
Natural History
Extant onychophorans are exclusively terrestrial and inhabit humid habitats (leaf litter, under stones, under bark, in rotten wood) on the floor of tropical rainforests or southern hemisphere temperate wet forests where they are nocturnal, negatively phototactic predators of small arthropods. Slime, ejected up to 30 cm from pores on the oral papillae, is used to entangle the prey (or would-be predators). The mandibles then lacerate the prey to provide an entrance point for injection of a toxic saliva. Hydrolytic enzymes in the saliva digest the prey and the resulting liquid is sucked into the gut by the muscular pharynx. The prey can be much larger than the velvet worm.
Reproductive strategies are widely varied in Onychophora. Velvet worms are gonochoric, fertilization is internal, sperm are transferred in spermatophores, and this transfer may be direct or indirect. Females usually have a seminal receptacle and males have a seminal vesicle, both for the storage of sperm, either allochthonous or autochthonous, respectively. In species with indirect sperm transfer males deposit spermatophores on the body of the female after which the sperm penetrate the body wall and make their way to the ovary where fertilization takes place. In most species females produce large yolky eggs and development is direct, producing miniature adults. Species may be oviparous or viviparous. Viviparous females, either matrotrophic (with a placenta) or lecithotrophic, gestate embryos in the uterus for about a year. Sexual dimorphism is minimal except that females are larger than males and have more legs. Onychophorans may reach lengths of 15 cm but most are smaller, 3-4 cm, the smallest being about 5 mm.
Laboratory Specimens
It is assumed that most studies of onychophoran anatomy will rely on preserved specimens supplied commercially and that in most courses the study will be limited to external anatomy. Few laboratories in North America will have the opportunity to study these animals alive. Preserved onychophorans are available from Ward’s Natural Science Co. as “peripatus”, with the name being used in its broadest sense as a common name for all onychophorans. These specimens may or may not actually belong to the genus Peripatus and in my experience they do not. Specimens I have purchased from Ward’s belong to Peripatopsidae, which does not include Peripatus. Generic identity makes little difference for a study of general gross anatomy, however, and this exercise can be used with representatives of either family.
The study should be conducted with the specimen immersed in liquid (water, 40% isopropanol, or 80% ethanol) in a small dissecting pan on the stage of a dissecting microscope. An anchovy fillet tin with a thin layer of dissecting pan wax poured on the bottom is ideal for this purpose. Specimens used only for the study of external anatomy can be reused from year to year but those dissected for examination of internal anatomy are ruined for further use and new specimens must be provided for each class. It is assumed that in most courses the study will be of external anatomy only.
External Anatomy
Examine a specimen immersed in fluid in a small dissecting pan on the stage of a dissecting microscope with low power, about 7X.
Papillae
Observe the body surface with higher power, about 45X. The body and appendages are covered by large and small papillae arranged in closely spaced rings, orannuli encircling the body or appendages. The papillae are covered by tiny scales, which are difficult to discern at this power, and are sensilla with various functions, chiefly chemo- and mechanoreceptive. The larger papillae bear a terminal chitinous sensory bristle (Fig 15-2B) and are mechanoreceptive (tactile). The bristle is best seen by looking at a papilla in side view against the dark background of the wax bottom of the pan. The smaller papillae lack the bristle. Sensilla on the antennae and lips are chemoreceptive.
Figure 1. A velvet worm (From Sedgwick, 1910). Onychop10.gif

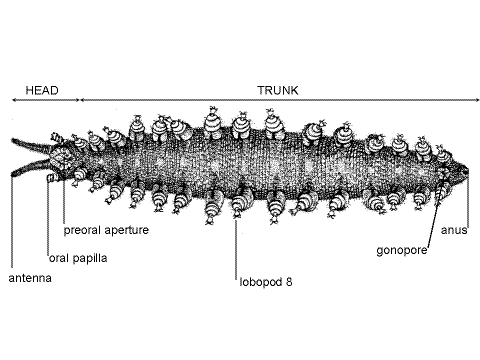
Tagmata
The onychophoran body is divided into a small anterior head and a much larger posterior trunk (Fig 1, 2) but the demarcation between the two is indistinct and both have paired segmental appendages. The body is not visibly segmented and, with the exception of the segmentally arranged lobopods, segmentation is not apparent externally. The external nephridiopores, coxal organs, and crural papillae are also segmentally arranged, but inconspicuous.
Figure 2. Ventral view of a typical velvet worm (From Pflugfelder, 1968). Onychop16.gif
Head
The three-segmented head bears three pairs of appendages; antennae, mandibles, and oral papillae (Fig 3, 15-1A). The large median opening on the ventral surface of the head is the best called the preoral aperture, although it looks like a mouth and is usually referred to thus. It opens into the shallow depression, the preoral cavity (= prebuccal depression), which precedes the true mouth. The true mouth opens from the inside wall of the preoral cavity into the pharynx and the preoral cavity is not part of the gut. The mandibles protrude into the preoral cavity and the salivary glands empty here. Students familiar with arthropod anatomy will see the similarities between this arrangement and the important, but less well defined, preoral cavity of insects, crustaceans, and chelicerates (Fig 21-7).
The onychophoran preoral aperture is better delimited and a more obvious chamber than that of arthropods and is surrounded by a circular outer lip and a semicircular inner lip (Fig 3, 15-1A) composed of scales. The lips close the preoral aperture. The scales of the inner lip completely surround the aperture whereas those of the inner lip arch around its anterior end. The preoral aperture may be partly or entirely closed in your specimen.
Figure 3. Ventral view of the head of a velvet worm. (From Sedgwick, 1910). Onychop7.gif
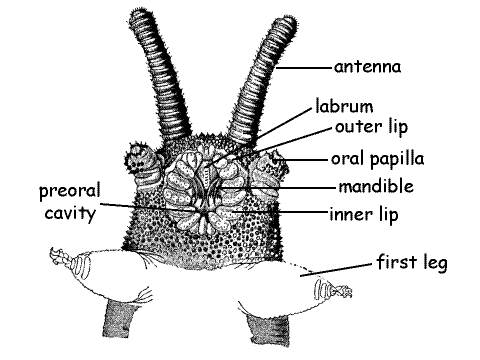
Labrum
The anterior labrum is the roof of the preoral cavity. Use fine forceps to push the lips apart and reveal the preoral cavity and its conspicuous labrum (= tongue). The muscular, median labrum is the dorsal anterior wall of the preoral cavity (Fig 3). It has a finely toothed mid-ventral ridge.
Antennae
Two conspicuous sensory antennae are present dorsolaterally at the anterior end of the head (Fig 1, 2, 3,15-1A). The antennae are conspicuously annulated and the annuli bear abundant chemo- and mechanoreceptive sensory papillae.
Mandibles
A pair of mandibles is located laterally in the walls of the preoral cavity (Fig 3, 15-1B). Each mandible consists of a base from which extend two sclerotized toothed jaws. Use your fine forceps once again to push the lips apart to reveal the curved brown sclerotized jaws. The jaws are posterior to the labrum and extend from the sides of the cavity into its lumen. Two jaws are present on each side but, if the mouth is partly closed, it will be difficult to see both.
Each mandible is equipped with a powerful and complex musculature. During prey capture, the preoral aperture is opened and the mandibles extended to lacerate the prey. The mandibles move independently of each other in longitudinal planes, from anterior to posterior. They do not oppose each other in the transverse plane. Saliva from a pair of salivary glands is released into the preoral cavity and then injected into the lacerated prey. Enzymes in the saliva begin digestion of the prey externally and the resulting liquid is pumped to the midgut by the pharynx.
Oral Papillae
The posteriormost pair of head appendages is the oral papillae. These lie laterally, more or less beside the preoral aperture (Fig 3, 15-1B). The distal end of each papilla bears the opening of the duct from one of the two large slime glands (Fig 5, 7). Like the antennae the oral papillae are sensory. Examine one apillae with high power of the dissecting microscope to see the sensory papillae. Note that they are equipped with a sensory bristle.
Eye
A tiny eye is located dorsally at the base of each antenna. The lens is visible externally. A retina and optic ganglion are internal. The eye looks like a translucent white bead embedded in the integument.
Trunk
The trunk may consist of up to 43 segments, depending on species, although there is no external evidence of segmentation (other than the lobopods). The anus is at the posterior end of the trunk.
Lobopod
Each trunk segment is equipped with a pair of fleshy, muscular legs, or lobopods (= onychopods) that are serial homologs of, and resemble somewhat, the oral papillae and mandibles (Fig 2, 4). The conical legs are soft and fleshy like the parapodia of polychaete annelids and lack the articles or articulations characteristic of arthropod appendages. The leg is divided into a proximal base and a distal foot. Since there are no joints the base can bend at any place. The foot can be retracted into the base.
Select a leg that extends away from the body so you can observe it against the black background of the wax dissecting pan and examine it with high power (about 45X) to study its complex structure (Fig 4). It will probably be necessary to examine several feet in this manner in order to see all aspects.
Each foot bears two sclerotized claws, similar and homologous to, but smaller than, the jaws of the mandibles. In addition, three to five spinous pads are present. The pads contact the substratum when the foot is deployed in normal walking but the claws are usually elevated above the substratum.
Three to seven small, sensory distal papillae are present on the dorsal and lateral sides of the foot immediately proximal to the claws. In addition, less obvious basal papillae may be present on the narrow part of the foot just distal to the spinous pads (Fig 4). The base of the leg is ringed by annuli of sensory papillae as is the body itself.
Figure 4. A lobopod. A. ventral view, B. Anterior view. (From Sedgwick, 1910). Onychop19.gif
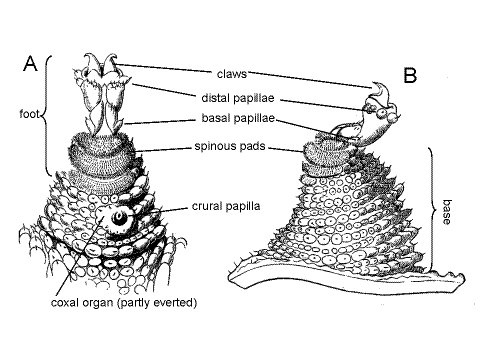
A short transverse groove is present ventrally at junction of the leg and body. It is perpendicular to the long axis of the body and parallel to that of the leg. The nephridiopore is in the proximal end of this groove. The crural and coxal gland openings, if present, are distal to the distal end of this groove (Fig 15-1C). The groove is easily found but the other structures are difficult to demonstrate. The nephridiopores are tiny, crural glands may not be present, and coxal organs may not be everted.
The limbs are equipped with muscles and each contains a diverticulum of the hemocoel, which functions as a hydrostatic skeleton. A valve at the base of each leg permits pressurization of the limb so that, in conjunction with its muscles, it can contribute to locomotion, moving forward and backward. The claws can be used to grasp the substratum during locomotion on rough surfaces and the spinous pads enhance traction against smooth surfaces. Onychophorans move slowly.
Crural Gland
In most taxa, paired, secretory, segmental crural glands are present in some legs. They may be on most legs or restricted to posterior legs, depending on taxon. They open via a pore on a crural papilla on the ventral surface of the base of the lobopod (Fig 15-1C). Little is known of their function.
Slime glands (discussed later) are modified crural glands. The two anterior accessory genital glands of males are the modified crural glands of the last pair of legs. They open via pores beside the gonopore and produce pheromones to attract females.
Coxal Organ
Coxal organs (= coxal vesicles) are paired, segmentally arranged organs in the lobopods. Each is an eversible sac that can be everted from a pore on the leg, apparently to absorb water from the environment. The pore is lateral to the nephridiopore at the base of the lobopod (Fig 15-1C).
Figure 5. Dorsal view of a dissected female velvet worm. The nerve cord and nephridia are omitted for clarity. (From Pflugfelder, 1968). Onychop12.gif
Internal Anatomy
" Open the body cavity (hemocoel) with a shallow dorsal incision made with fine scissors. Do not attempt this with a scalpel. The incision should be about 1 mm to the right or left of the dorsal midline to avoid damage to the heart, which lies immediately ventral to the midline. The incision should extend the length of the worm from just anterior to the anus to, but not into, the head. It should be no deeper than the thickness of the body wall. Pin the body wall aside using # 1 stainless steel insect pins inserted at 45 ° angles. Keep the specimen immersed in fluid. Avoid damaging the brain located dorsally in the head and the tubular heart on the dorsal midline. The pericardial sinus and heart may be damaged by this procedure but should remain recognizable.
Figure 6. Cross section of the trunk of a velvet worm (From Pflugfelder, 1968). Onychop14.gif
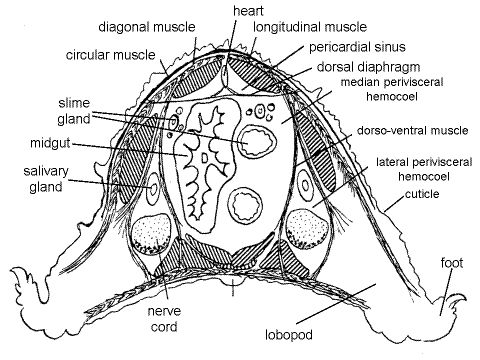
The body cavity is a hemocoel as it is in arthropods. It is divided into four compartments (Fig 6). A perforated dorsal diaphragm (= pericardial septum) separates the dorsal pericardial sinus from the much larger ventral perivisceral hemocoel, as in arthropods. The transverse fibers of the dorsal diaphragm can be seen by focusing carefully on the dorsal body wall. The longitudinal fibers of the inner muscle layer of the body wall can be seen dorsal to the diaphragm and pericardial sinus. The heart, probably appearing as a collapsed tube, but sometimes an obvious tube, circular in cross section, can be seen as a thickened band adhering to the dorsal surface of the diaphragm, on the dorsal midline.
Two sheets of dorsoventral muscles, whose fibers appear transverse from your viewpoint, divide the perivisceral hemocoel longitudinally into three, side-by-side compartments; two lateral perivisceral hemocoels and one median perivisceral hemocoel, of which the median hemocoel is by far the largest and is the one you have opened. The lateral hemocoels extend into the lobopods. The panarthropod hemocoel is not segmentally divided by transverse septa as is the coelom of a typical annelid.
The median perivisceral hemocoel contains the gut, slime glands and reproductive system whereas the lateral perivisceral hemocoels contain the salivary glands, ventral nerve cords, and nephridia. By focusing through the dorsoventral muscle sheet you can see into the lateral perivisceral hemocoel and find the salivary gland and ventral nerve cords as longitudinal white bands. The salivary glands are lateral to the nerve cords.
The fluid-filled hemocoel is a hydrostatic skeleton permitting the circular and longitudinal body wall muscles to antagonize each other in a locomotory system similar to that of annelids.
Digestive System
" Carefully extend the middorsal incision anteriorly through the head. The large, white, bilobed brain lies immediately under the body wall of the head. The incision must be shallow to avoid damage to the brain.
The gut extends from anterior mouth to posterior anus and consists of ectodermal foregut, endodermal midgut, and ectodermal hindgut, as it is common in bilaterians. The gut is preceded by the short preoral cavity which was discussed earlier.
The foregut comprises mouth, pharynx and esophagus. The mouth opens from the preoral cavity whose preoral aperture is visible on the ventral surface of the head. The mouth itself will not be seen in this dissection.
The pharynx can be seen extending posteriorly from under the brain (Fig 7, 15-3B). It is a short, slightly swollen, muscular pump used to bring liquefied food into the gut. The circular muscles of the pharyngeal wall can be seen with magnification. A pair of slender muscles extend from the anterior dorsal body wall to the posterior end of the pharynx. The pharynx connects with the enormous midgut via a very short (compared with the pharynx), narrow esophagus.
The midgut (Fig 5, 6, 15-3B), sometimes referred to as the stomach, intestine, or stomach-intestine, is a long, thick, irregularly creased tube extending most of the length of the trunk and accounting for most of the length of the gut. The midgut epithelium is secretory and absorptive and final digestion and absorption occur here.
The hindgut, or rectum, is short, sinuously curved, and narrow. It extends from the posterior midgut to the terminal anus. Find the anus and use a microneedle to demonstrate its continuity with the rectum.
Hemal System
The heart is a dorsal, muscular, segmentally ostiate tube that pumps blood from and to the hemocoel (Fig 6). It extends most the length of the body from the head to the level of the gonopore. It is enclosed in the dorsal pericardial sinus (= pericardial hemocoel) and adheres to the dorsal diaphragm. It is thus surrounded by blood, as in arthropods but very different from annelids. The heart is probably closed posteriorly but opens anteriorly into the head hemocoel without benefit of an aorta. Arteries and aortae are probably absent. The flow of blood through the pericardial sinus, ostia, heart, perivisceral hemocoel, and back to the pericardial sinus is like that of arthropods. The blood contains hemocytes and nephrocytes and is colorless.
Trachea
The onychophoran respiratory system consists of abundant tufts of very fine (1-3 m m in diameter) unbranched tracheae extending from spiracles on the body surface to the tissues. The midgut, brain, and muscles are especially well supplied by tracheae. The tiny spiracles are scattered over the body surface and there may be as many as 75 per segment. Tracheae and spiracles are difficult to demonstrate in gross dissection. Each spiracle opens into an atrium from which the tracheae radiate to the tissues. Spiracles are most abundant on the dorsal surface. Onychophoran spiracles, unlike those of most arthropods, cannot be closed thus rendering the animal susceptible to desiccation in dry environments. Consequently velvet worms are restricted to humid habitats.
Slime Gland
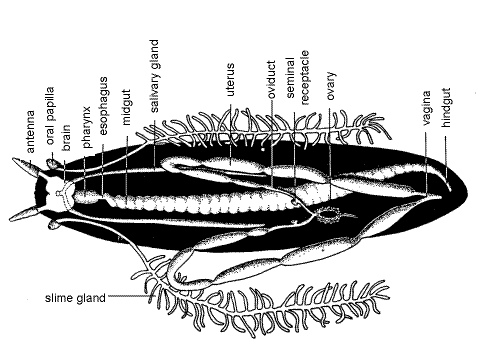
A pair of large, white slime glands lies in the median perivisceral hemocoel where they occupy most of the space not allotted to the gut. Each is a blind tube opening anteriorly at an aperture on an oral papilla. Posteriorly the gland is highly branched (Fig 5, 7, 15-3B).
Slime glands eject jets of milky white liquid slime from a pore at the tip of the oral papilla. The slime immediately, but temporarily, denatures to harden on contact with air. Within a few minutes the stickiness is lost. The fresh slime sticks tenaciously to arthropod exoskeletons but does not stick to the hydrophobic cuticle of the velvet worm. Slime glands are the modified crural glands of the segment of the segment of the oral papillae.
Onychophorans are gonochoric. The reproductive systems of male and female are similar in consisting of paired gonads, paired ducts leaving the gonads to coalesce to form a single common duct that opens at the posterior gonopore. Accessory genital glands are present in both sexes. The reproductive systems are in the median perivisceral hemocoel.
Female
The two tubular ovaries are wound together to form a single median mass near the posterior end of the body in the median perivisceral hemocoel (Fig 5, 15-3B). Small, oval, whiteeggs may be apparent in the combined ovaries. The ovary is suspended from the dorsal diaphragm by the ovarian ligament. From each ovary arises a slender white oviduct that passes anteriorly then turns sharply posteriorly and immediately expands to become a large uterus. Embryos, arranged like peas in a pod, may be present in the uteri of your specimen. If so, inform your instructor and be prepared to share with the class. A tiny pouch off each oviduct serves as a seminal receptacle for storage of sperm but these may be difficult to find (Fig 5).
Posteriorly the uteri pass ventral to the rectum, dorsoventral muscles, and nerve cords before joining to form the very short vagina, which opens at the gonopore (= genital aperture). The vagina is ventral to the rectum and hidden by it. Carefully tease away the dorsoventral muscles and connective tissue to reveal the vagina. The gonopore, also ventral to the rectum and vagina, is on the ventral midline between the last pair of legs (Peripatopsidae) or between the penultimate pair of legs (Peripatidae).
Male
The male reproductive system consists of a pair of testes and the ducts that drain them and is located in the median perivisceral hemocoel (Fig 7). Each tubular testis drains into an expanded spherical seminal vesicle where sperm are stored. The vesicle drains into a long convoluted vas efferens. The two vasa efferentia, one from each seminal vesicle, join posteriorly to form a single short vas deferens on the left side of the median hemocoel. Distally the vas deferens become the muscular ejaculatory duct which opens at the gonopore (= genital aperture). Where these ducts cross from one side of the body to the other they pass ventral to the nerve cords. Thus the right vas efferens passes ventral to both nerve cords to join the left vas efferens to form the vas deferens. The ejaculatory duct, which lies on the left, must pass ventral to the left nerve cord to reach the median gonopore.
Two pairs of accessory genital glands, anterior and posterior, are associated with the male system and open via pores near the gonopore. The anterior male accessory genital glands open via pores lying lateral to the gonopore. The posterior male accessory genital glands open by a pair of pores slightly posterior to the gonopore.
Salivary Glands
The two salivary glands are long, blind-ending tubes extending posteriorly from the head to about the middle of the trunk in the lateral hemocoels (Fig 5, 6, 15-3A, B). In a dorsal dissection such as yours, you will see them through the translucent layer of dorsoventral muscles. (Two white bands can be seen on each side ventral to the dorsoventral muscle layer. The lateral band is the salivary gland, the medial band is the nerve cord.) The two salivary ducts join anteriorly to form a short common salivary duct which empties into the preoral cavity but this junction will not be seen.
Salivary glands are the modified saccate nephridia of the segment of the oral papillae (segment 3). Like other nephridia they are located in the lateral perivisceral hemocoels.
The central nervous system is ladder-like, a phrase frequently used to describe annelid and arthropod nervous systems. Such a system consists of a dorsal brain, circumenteric connectives, and a pair of ganglionated ventral nerve cords connected by commissures to form a system that looks like a ladder with the nerve cords being the uprights of the ladder and the commissures the rungs. Because of its widely spaced cords and long commissures, the onychophoran system looks even more like a ladder than do those of annelids and arthropods (Fig 7, 15-3B).
" If you have not already done so, extend the dorsal body wall incision along the length of the head and into the base of an antenna. Do this without damaging the eyes on the head surface or the antennal nerves inside the antennae.
The brain consists of two large cerebral ganglia fused together on the midline in the dorsal head (Fig 7). Two large antennal nerves exit the brain anteriorly and extend into the antennae. Two large circumenteric connectives, one from each cerebral ganglion, exit the brain posteriorly and extend ventrally where each becomes a ventral nerve cord. Near the middle of the dorsal surface of the brain two short optic nerves exit the brain and extend to the optic ganglion and pigmented retina lying under the eyes. Mandibular nerves to the mandibles exit the circumenteric connectives.
Figure 7. A dissected male velvet worm in dorsal view. The accessory male genital glands, left nephridia, and left slime gland are omitted for clarity (Partly original and partly redrawn from several authors including Sedgwick (1910), Purcell (1900), and Storch and Ruhberg (1993). Onychop20L.gif
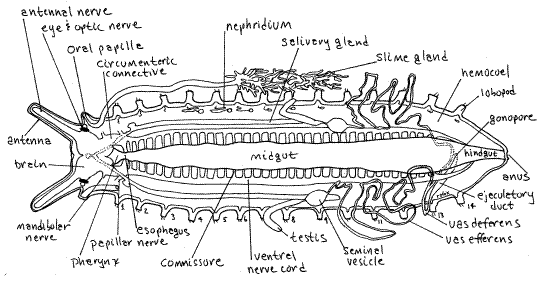
The first pair of ganglia (inconspicuous and not demonstrable) on the ventral nerve cord are those of the oral papilla and from them arise the papillar nerves. These two ganglia are connected by a thick transverse commissure, which is the first of the long series of rung-like commissures along the ventral nerve cord.
Excretory System
The excretory system consists of segmentally arranged pairs of saccate nephridia (= segmental organs) similar to those of arthropods. A typical such nephridium consists of an end sac, derived from an embryonic coelomic space (and with podocytes in its wall) and a tubule, derived from a metanephridium (Fig 15-3A).
The nephridia are in the lateral perivisceral hemocoels immersed in blood, which is ultrafiltered across their walls to form the primary urine in the sac. The primary urine is modified by selective absorption and secretion as it passes through the tubule to become the final urine. The distal part of the tubule may be expanded to form a bladder for storage of the final urine. The nephridium opens through a nephridiopore in a slit on the ventral surface of the base of each foot (Fig 15-1C).
The nephridia of the segments of legs 1-3 are reduced and smaller than all others (Fig 7). Those of in the segments of legs 4 and 5 are much larger than other nephridia, lack the bladder, and have nephridiopores distally on the leg. The remaining nephridia are smaller than nephridia 4-5.
Unpin the left anterior end of the specimen so you can locate legs 4 and 5 and then repin the specimen. Focus through the dorsoventral muscle layer in the vicinity of legs 4-5 to find the long convoluted tubules of the nephridia of leg segments 4-5. They can be seen between the salivary gland and the nerve cord. With fine scissors carefully make a longitudinal incision through the dorsoventral muscles in this region to improve your view of the nephridia.
References
Anon. 2003. Australian Museum Fact Sheet: Velvet Worms. www.amonline.net.au/factsheets/velvet_worms.ht m
Ax P. 2000. Multicellular animals, II. Springer, Berlin. 396pp.
Balfour FM. 1883. The anatomy and development of Peripatus capensis. Quarterly Journal Microscopical Science, 23:213-259, pls 13-20.
Barnes RB. 1993. Introduction to the Onychophora, Chilopoda, and lesser Protostomata, pp. 1-9 in Harrison FW, Rice ME (eds.). Microscopic anatomy of invertebrates, vol. 12 Onychophora, Chilopoda, and lesser Protostomata. Wiley-Liss, New York. 484pp.
Bullough WS. 1958. Practical invertebrate anatomy. MacMillan, London. 483 pp.
Freeman WH, Bracegirdle B. 1971. An Atlas of invertebrate structure. Heinemann Educational Books, London. 129 pp.
Kaestner A. 1968. Invertebrate zoology, II. Arthropod relatives, Chelicerata, Myriapoda. Wiley Interscience, New York. 472 pp.
Nielsen C. 2001. Animal evolution, 2 nd ed. Oxford University Press, Oxford. 563 pp.
Parker TJ, Haswell WA. 1951. The Onychophora, pp. 526-533 in A text-book of zoology, I, 6 th ed. MacMillan, London. 770 pp.
Pflugfelder O. 1968. Onychophora. Grosses Zoologisches Praktikum 13a:1-42, Gustav Fischer Verlag, Stuttgart.
Purcell WF. 1900. On the anatomy of Opisthopatus cinctipes, Purc., with notes on other, principally South African, Onychophora. Annals South African Museum 2(4):67-116.
Reid AL. 1996. Review of the Peripatopsidae (Onychophora) in Australia, with comments on peripatopsid relationships. Invertebrate Taxonomy 10:663-936
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Sedgwick A. 1888. A monograph of the species and distribution of the genus Peripatus (Guilding). Quarterly Journal Microscopical Science, 28:431-493.
Sedgwick A. 1910. Peripatus, pp. 3-26 in Harmer SF, Shipley AE. (eds). Cambridge natural History 5, Peripatus, Myriapods, and Insects pt 1. MacMillan, London. 584 pp.
Snodgrass RE. 1938. Evolution of the Annelida, Onychophora, and Arthropoda. Smithsonian Misc. Collections 97(6):50-149.
Storch V, Ruhberg H. 1993. Onychophora, pp. 11-56 in Harrison FW, Rice ME (eds.). Microscopic anatomy of invertebrates, vol. 12 Onychophora, Chilopoda, and lesser Protostomata. Wiley-Liss, New York. 484pp.
Supplies
Onychophoran, preserved or living
Dissecting microscope
Dissecting set with microdissecting tools
Small dissecting pan (anchovy tin with wax bottom)
#1 stainless steel dissecting pins