Invertebrate Anatomy OnLine
Molgula manhattensis ©
Sea Squirt
5jul2006
Copyright 2001 by
Richard Fox
Lander University
and
Edward Ruppert
Clemson University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises, a glossary, and chapters on supplies and laboratory techniques are also available at this site. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
ChordataP, Tunicata sP, Ascidiacea C, Pleurogona O, Stolidobranchia sO, Molgulidae F (Fig 27-12, 29-32)
Chordata P
Chordata is characterized by a suite of apomorphies including a dorsal hollow nerve cord, notochord, pharyngeal gill slits, and a post anal tail (Fig 29-1). The ancestor was a fishlike deuterostome that swam using alternating contractions of right and left longitudinal axial muscles to create undulations of the body. The flexible, incompressible notochord prevented these contractions from compressing the body while allowing lateral deflection. The chordate central nervous system is a hollow, median, longitudinal nerve cord formed in the embryo by an invagination of surface ectoderm whose original function was probably sensory reception. Paired pharyngeal gill slits connect the lumen of the pharynx with the exterior and originally functioned in suspension feeding with respiration being added later. A muscular tail posterior to the anus is, although commonplace in chordates, an unusual feature not found in other taxa. It is an extension of the axial musculature and is the chief locomotory organ. An additional apomorphy is the endostyle, a region of pharyngeal endoderm, that secretes iodated compounds, either mucus or hormones.
Tunicata (= Urochordata) sP
Tunicates are highly derived and less like the ancestral chordates than are cephalochordates or vertebrates. At some time in the life cycle all possess a notochord, dorsal hollow nerve cord, pharyngeal gill slits, postanal tail, and endostyle but only the gill slits and endostyle are present in adults. Tunicates use the pharyngeal gill slits for suspension feeding. The larva is much more chordate-like than the adult and resembles a tadpole or fish, has all the chordate apomorphies, and is known as the tadpole larva. Metanephridia are absent and coelom is reduced to a pericardial cavity and gonads. As in cephalochordates the gut is dominated by an enormous pharynx surrounded by a water-filled atrium but unlike cephalochordates, it is U-shaped with the mouth and anus anterior. Tunicates may be benthic or planktonic and solitary or colonial. All are marine.
Tunicata is traditionally divided into Ascidiacea (the benthic sea squirts in three taxa; Aplousobranchia, Phlebobranchia, and Stolidobranchia), Thaliacea (the pelagic salps), and Appendicularia (the pelagic larvaceans). Recent molecular evidence and reevaluation of morphological evidence, however, suggests that Ascidiacea is paraphyletic and Tunicata should be reorganized into three different higher taxa (Fig 29-32). In this reorganization Stolidobranchia would be one higher taxon. Phlebobranchia plus Thaliacea would be the second taxon. Aplousobranchia plus Appendicularia is the final tunicate taxon. For now, however, the traditional classification will be followed.
Ascidiacea C
Ascidiacea is usually taken as representative of Tunicata, at least for the purposes of introductory laboratory exercises. Ascidians, or sea squirts, are sessile filter feeders that, as adults, bear little resemblance to their chordate relatives. Ascidians have a living, external, cellular exoskeleton, or tunic, underlain by epidermis. The tunic resembles connective tissue, except it isoutside the epidermis, and consists of cells, a secreted extracellular matrix, and ground substance. Much of it is a cellulose-like polysaccharide. In many ascidians blood vessels cross the epidermis to enter the tunic, a feature found in no other animal.
The gut is U-shaped and both openings are anterior, with the anus dorsal to the mouth. The gut is dominated by an enormous pharynx whose wall is perforated by numerous tiny gill slits. The pharynx is surrounded by a water-filled atrium into which the gill slits open and which itself opens to the sea. It is both respiratory organ and filter-feeding device. Water and food particles enter the pharynx and the water passes through the gill slits to the atrium and then out the siphon. Food, entangled in mucus secreted by the endostyle, remains in the gut and passes posteriorly to be digested.
The hemal system includes a heart, vessels, and blood spaces in the connective tissue. The heart is enclosed in a pericardial cavity derived from the ancestral coelom. The pattern of blood flow resembles that of the cephalochordates and early vertebrates except that the heart reverses direction periodically and the blood thus flows in both directions through the system. Ascidians have no structure recognizable as a kidney.
Ascidians are simultaneous hermaphrodites and the gonoducts open into the atrium. Some ascidians are solitary and may be relatively large. Others are colonial with tiny individual zooids in a common tunic.
Pleurogona O
The gonad*s) are on the inner surface of the body wall. The epicardium is absent or represented by a renal sac.
Stolidobranchia sO
Phlebobranchia, Aplousobranchia, and Stolidobranchia are the three higher ascidian taxa. In stolidobranchs the gonads are on the inside surface of the body wall beside the pharynx and not in the gut loop. No epicardium is present but Molgulidae has renal sacs, which are derived from the epicardium. The neural gland is dorsal to the cerebral ganglion. The stolidobranch pharyngeal lining is strongly pleated and has transverse and longitudinal blood vessels. Molgulidae has spiral gill slits. Stolidobranchs can be solitary or colonial. The plane of the tadpole tail is vertical.
Laboratory Specimens
Molgula manhattensis, the sea grape, is a solitary sea squirt of shallow water on the coast of most of Europe and Britain (Norway to Portugal) and on the Atlantic and Gulf coasts of the United States from Maine to Texas. This exercise is written specifically for Molgula but could also be used with several other genera.
Instructors wishing to use locally collected sea squirts have several choices depending on their geographic location. Of the two common solitary ascidians on the southeastern coast of the Unites States, Styela plicata (Stolidobranchia: Styelidae) and Molgula manhattensis, the latter is the best subject for dissection even though it is much smaller than Styela. The thick tunic of Styela is very difficult to remove and is a formidable deterrent to dissection. Ascidia interrupta (Phlebobranchia: Ascidiidae) occurs in the northeastern United States and is present in some parts of the Southeast (North Carolina and Florida). It is a good choice when available. Molgula, Ascidia interrupta or Ciona intestinalis can be used in the northeastern United States. Ciona intestinalis (Phlebobranchia: Cionidae) is one of the most widely distributed ascidians. It is common on the northern European and North American Atlantic and Pacific coasts. Ciona is often used in teaching laboratories and another exercise in this series describes its anatomy ( Invertebrate Anatomy OnLine ).
On the west coast of North America Ascidia ceratodes is almost identical to Ascidia interrupta but is smaller, 50-70 mm. It occurs from British Columbia to the Gulf of California in shallow water and is usually common, sometimes abundant. Pyura hastata (Stolidobranchia: Pyuridae) is about the size of Molgula manhattensis and occurs from Alaska to southern California where it can be common.
Because it is smooth, globular, translucent, and often grows in clusters, Molgula manhattensis goes by the common name of "sea grape" (Fig 29-12G). Large specimens are about 2 cm in diameter but much of this mass is tunic and seawater so the animal inside is closer to 1 cm. The dissection is best performed on living specimens but preserved material can be used if necessary. A dissecting microscope should be used throughout and living specimens should be relaxed in isotonic magnesium chloride.
External Anatomy
Place a large Molgula in a culture dish of seawater (or tapwater if preserved) on the stage of the dissecting microscope and examine with the lowest power. If it is alive and you are careful to avoid disturbing it, the animal will probably relax, extend its siphons, and begin pumping water through its pharynx.
Siphons
The body is usually ovoid and slightly flattened from side to side (right to left). The two siphons that protrude conspicuously from one end of the body are easily found but not so easily told apart. Try to distinguish them at this time but do not despair if you cannot be sure which is which. You will be able to tell them apart later when you get inside.
The buccal siphon is situated at the end of the oval outline of the animal whereas theatrial siphon is close to it but on one side of the oval. If you have a living specimen and allow it to extend the siphons they can more easily be distinguished. The opening of the buccal siphon is surrounded by six small conical lobes whereas that of the atrial siphon has four larger lobes. These lobes can be seen when the siphons are not extended but are not as obvious then. The atrial siphon is usually the longer and narrower of the two.
Orientation
If you have identified the two siphons, you can use them as landmarks to orient the animal. The buccal siphon is anterior and the atrial siphon is dorsal. The end of the sphere opposite the buccal siphon is posterior and the side opposite the atrial siphon is ventral. The plane of symmetry passes through both siphons and divides the animal into right and left halves.
Feeding
Be sure you understand the feeding process before dissecting your specimen. The buccal siphon opens into the anterior end of the gut, most of which is an enormous pharynx with minutely perforated walls. The perforations are the gill slits, or stigmata. The pharynx is surrounded by an invaginated seawater space known as the atrium. The atrium connects with the exterior through the atrial siphon.
The walls of the pharynx are covered by a moving sheet of mucus that forms a fine-meshed sieve (Fig 29-15D). The mucus is secreted by the endostyle, located mid-ventrally in the pharynx. The mucus is moved dorsally from the endostyle by frontal cilia on the walls of the pharynx so it covers the walls and gill slits.
A water current is generated by lateral cilia in the gill slits. Water and food particles enter the buccal siphon and move into the pharynx lumen. The water passes through the mucous net, through the gill slits, and into the atrium. It then returns to the sea through the atrial siphon.
Food particles too large to pass through the mucous net are retained in the pharynx. The mucus is gathered by the dorsal lamina on the dorsal midline of the pharynx. Cilia of the dorsal lamina move the mucus and food posteriorly into the stomach where they are digested. The gut empties via an anus into the atrium.
The gonads also empty into the atrium. Digestive wastes and gametes are carried out of the atrium by the feeding current as it exits the atrial siphon.
>1a. Add some seawater/carmine suspension to the water in the dish and gently swirl the carmine about so it is dispersed throughout the water in the dish. Under magnification and against a lighted background (transmitted light), observe the openings of the siphons and look for the movement of carmine, or other particles, into or out of the siphons. You should be able to discern a steady flow of water into the buccal siphon and another stream out of the atrial siphon.
The incurrent flow will be easier to see because of the carmine particles in the water but the outflow should have no particles and will be best detected by watching for the displacement of the water surrounding the opening of the atrial siphon. Why are there no carmine particles in the outflow? <
>1b. Use a Pasteur pipet to deliver a little 0.1% methyl green/seawater or carmine/seawater directly to the opening of the extended buccal siphon. The dye will probably enter the siphon, be detected by sense organs, and then be forcibly ejected as the animal contracts the body wall muscles and empties the pharynx and atrium through the siphons. <
>1c. Slip the tip of a pipet or hypodermic needle into the buccal siphon and inject a little 1% methyl green/seawater into the closed pharynx. Remove the needle and allow the animal to recover from the experience, reextend its siphons, and begin pumping water again. When the pharyngeal cilia begin beating, the dye will be seen exiting the atrial siphon. Be sure to distinguish between the slow gentle ciliary current and the abrupt, forceful muscular current. How is it that dye will cross the pharynx and leave the atrial siphon when carmine particles will not?
>1d. Pick up the squirt and hold it over a towel well away from your microscope and books, and squeeze it gently. Make sure the siphons are not pointed at the microscope when you do this. If it is healthy, it should contract its body wall muscles and eject streams of water from one or both siphons as it empties the pharynx and atrium. Much of the volume of a functioning, feeding sea squirt is seawater and the animal will become much smaller when that water is expelled. <
Tunic
Molgula is covered by a thick, translucent tunic, to which the name “tunicate” alludes. The tunic is the outermost layer of the body wall which consists of tunic and mantle. The mantle, in turn, consists of the epidermis, connective tissue, circular and longitudinal muscles. The tunic is a secreted exoskeleton but is far from being dead and inert as exoskeletons typically are. It is composed of a matrix of cellulose (tunicin), protein fibers, cells, and proteoglycan ground substance. In many tunicates it also contains blood vessels. The tunic is unique in being a living exoskeleton that grows as the animal grows and does not require molting.
Its cells are derived from the connective tissue mesenchyme of the body wall, not from the epidermis which underlies it. It is thought that the cellulose, however, is secreted by the epidermis. The tunic is sometimes thought of as an external connective tissue lying outside the epidermis. The epidermis, muscles and internal connective tissue of the mantle lie beneath the tunic and cannot be seen yet.
Look closely at the surface of the tunic. It is probably fouled with silt, sand, and debris. There may be other animals, including small Molgula, growing on it. Some areas of the surface bear small, hairlike extensions of the tunic, called papillae that are used to hold sand and other particles to the surface. A roughened, irregular area or projection, usually at the posterior end, marks the former point of attachment of the tunic to the substratum. Sometimes the substratum is another tunicate, often of the same species.
Remove the sea squirt from the dish and scrub it gently with a toothbrush to remove most of the adhering debris. Return it to the dish. Most of the remaining debris will probably be in the vicinity of the siphons, which is where the papillae are best developed. The papillae should be easier to see now and some of the viscera should be visible through the body wall. Easiest to see are the intestine and orange or brown pyloric gland (= liver) associated with the stomach wall at the posterior end of the dorsal surface.
Internal Anatomy
" Your next task is to remove the tunic without damaging the animal within. This is not difficult but it must be done with care and in accordance with the instructions. The soft body of a sea squirt is connected to the tunic only at the siphons so it is fairly easy to remove the animal once the tunic has been opened.
Be sure you know the locations of the sagittal plane and midline. Remove the animal from the dish and hold it gently between your thumb and forefinger. With a sharp scalpel, make a median longitudinal incision through the tunic (only) beginning at one siphon and extending around the perimeter of the animal to the other siphon. Keep the incision on the midline all the way around the dorsal, posterior, and ventral perimeter. Use short, repetitive, slicing strokes of the scalpel and do not put unnecessary pressure on the animal with either your fingers or the scalpel.
As soon as you make the first penetration completely through the tunic, the soft body will ooze through the break in the tunic. Avoid cutting in the region of the break as it is important that you do not cut the soft body. Continue cutting the tunic on either side of the breakthrough and work your way completely around the specimen.
(Sometimes the pressure from your fingers will cause the entire animal to squeeze out through the buccal siphons. This is not the same as oozing out through a cut in the tunic and, while not disastrous, it is not desirable. If it happens, the animal will be intact and usable but inside out. It must be separated from the tunic and turned right side out before you can use it. Studying an inside out animal without realizing it can be very confusing. Such an animal will be enclosed in its own pharynx.)
After you have extended the incision completely around the animal, from one siphon to the other, the two halves of the tunic will peel away revealing the soft animal, enclosed in the mantle, within. The process may remind you of peeling a grape.
If the two sides will not separate easily from each other, use the dissecting microscope to look for small areas where the two halves of the tunic remain attached to each other and carefully cut them without cutting the adjacent body. Whereas the tunic is now free of the body, the rest of the body wall (mantle) is still intact and should remain so for the time being.
When you separate the two sides of the tunic, you will notice that the animal is firmly attached to the tunic only at the two siphons. If you are observant, you may notice a delicate, transparent blood vessel running from the body to the tunic on each side of the tunic. Working under magnification, use your fine forceps to pull gently the siphonal tissues and free them from their attachment to the tunic.
Note the texture and consistency of the tunic and then set it aside. Place the animal in a small dissecting pan of seawater deep enough to cover it. An empty sardine can with a wax bottom makes a good dissecting pan for Molgula. The animal should not be placed in magnesium chloride until after the heart has been studied. If you are dissecting a preserved animal, cover it with tapwater.
Mantle
Relocate the buccal and atrial siphons and orient the animal with the right side facing upward, toward you. The animal is still enclosed in the remaining body wall, or mantle, and only the exoskeleton has been removed so far. The mantle is primarily connective tissue and muscle with the thin epidermis over its surface. The muscles are poorly developed in Molgula. They are best developed around the siphons where they form sphincters to close the siphons (Fig 29-19). The muscle layer consists of widely separated transverse, longitudinal and oblique strands. With carefully adjusted light and a dark background, you can see these strands in the mantle. The viscera are partly obscured by the translucent mantle but the outlines of most of the organs can be seen through it. Do not remove the mantle at this time, but be aware that because of it you are not getting a clear view of the organs.
Right Side
Look at the animal's right surface with the buccal siphon on your right and away from you as in Figure 1. Most of the tissue inside the mantle is the pharynx but it is difficult to observe with the mantle intact.
Figure 1. The right side of Molgula manhattensis with the tunic removed (after van Name, 1945). Uro79L.gif
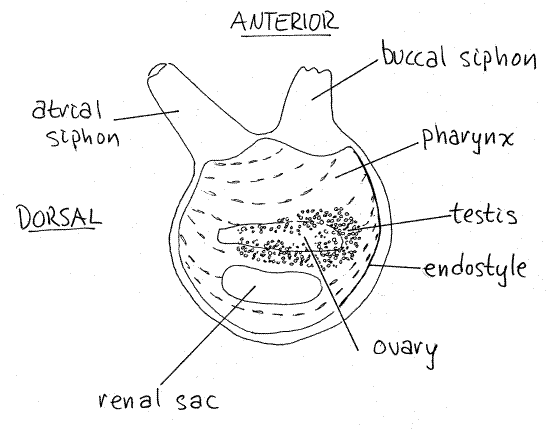
The most conspicuous feature on this side is the renal sac (= renal organ, kidney). The renal sac is a unique and characteristic feature of Molgulidae. It is thought to be homologous to the epicardium of other ascidians and the epicardium may be a remnant of the coelom. The renal sac is slightly curved and has a longer greater curvature and shorter lesser curvature.
The renal sac is a turgid, transparent, sausage- or bean-shaped sac located in the posterior half of the squirt (Fig 1, 29-22C). It has no duct connecting it to the exterior. It is a storage kidney that accumulates and stores uric acid crystals, which are easily seen as a whitish precipitate in the sac. Tunicates cannot break down the uric acid from nucleic acid metabolism and it is stored for life by various mechanisms in different species. Only molgulids have a single, large renal sac for this purpose. The nitrogen from protein metabolism is lost more conventionally as ammonia across the permeable surfaces of the body. No excretory organ has been found in ascidians.
Sea squirts are hermaphroditic and Molgula has gonads consisting of an ovary and testis on each side. The gonads of stolidobranchs are embedded in the part of the mantle that forms the wall of the atrium. The right hermaphroditic gonad is located on the anterior, lesser curvature of the renal sac, between the sac and the siphons, and almost touching the sac (Fig 1). It is composed of both ovary and testis. Its long axis is roughly parallel to the animal's dorso-ventral axis and is parallel to that of the renal sac and it is about the same length and width as the sac.
The gonads appear through the translucent tissues of the mantle as an indistinct whitish lobed organ. The long ovary is surrounded by the testis. A white oviduct extends along its long axis. The testis is composed of many small spherical lobules that may remind you of eggs, which they are not. The testis is located on the periphery of the ovary, along both its margins on the left but only on the posterior margin on the right. You will see the gonads more clearly after you remove the mantle.
Heart
The heart is located on the concave lesser curvature (anterior margin) of the renal sac, on the right side of the animal (Fig 29-22C). It is best to locate it now, before removal of the mantle. It is between the renal sac and the right gonad and extends along most of the length of the lesser curvature of the renal sac. It is enclosed in a coelomic space, the pericardium, but that is not apparent (Fig 29-21).
The diaphanous, transparent walls of the heart are the inner walls of the pericardium and are difficult to recognize as such unless the heart is beating (Fig 29-21B). Waves of contraction run from one end of the heart to the other every few seconds. The billowing walls of the pulsating heart look like a diaphanous curtain waving in a gentle breeze. If the animal is in seawater you may see the heart beat but not if it is in magnesium chloride.
>1e. Count and record the beats, or peristaltic waves, per minute. The heart of urochordates is unusual in that it periodically pauses and then reverses direction. Note the direction in which the peristaltic waves propagate and watch for a few minutes to see the beat reverse direction. What is the approximate length of time between reversals? <
The body cavity of ascidians is a hemocoel consisting of open, unlined sinuses and vessel-like channels. The body is supplied by the heart via two blood channels, or vessels (Fig 29-21A). Each end of the heart opens into one of these channels. The ventral end of the heart connects via some of the ventral channels to the ventral side of the pharynx, endostyle, and test. The dorsal end of the heart connects, via the dorsal channel, to the dorsal side of the pharynx, the viscera, and the test. Both parts of the hemal system end in the same fine capillary-like channels in the pharynx. Each half of the system alternately serves as veins, then arteries, and then veins again as the heart periodically reverses its beat.
When you have finished your study of the heart, you may replace the seawater in your dissecting pan with isotonic magnesium chloride if you wish. The heart will stop beating in magnesium chloride. It will continue beating for hours in seawater.
Left Side
Turn the animal over and look at the left side (Fig 2). The most conspicuous feature on this side is the postpharyngeal gut which appears as a hairpin loop along the periphery of the animal. It begins on the dorsal side near the atrial siphon with a short esophagus which you cannot see now.
Figure 2. The left side of Molgula manhattensis with the tunic removed (after van Name, 1945). Uro80L.gif
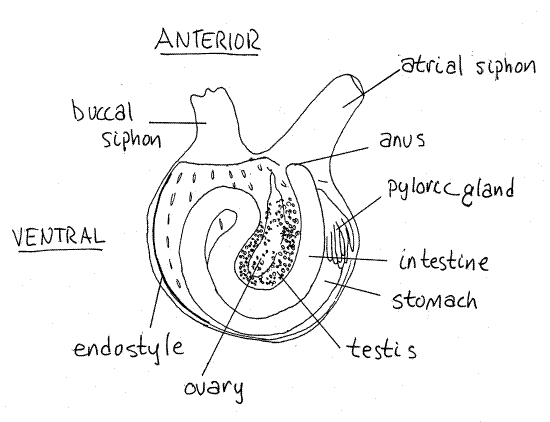
The esophagus leaves the pharynx and empties into the anterior end of the stomach which is almost completely hidden by the large, conspicuous, brown or orange (in life) pyloric gland.
The stomach exits the pyloric gland and begins the outer leg of the hairpin loop of the intestine (Fig 2, 29-15A). The stomach is swollen proximally but tapers gradually to become theintestine. The intestine extends ventrally and then doubles back on itself, passes by the stomach to empty via the anus into the atrium, neither of which are visible at this time. The left gonadis located in the interior of the gut loop. Its long axis roughly coincides with the animal's antero-posterior axis.
" Turn your specimen back over so the right side faces you. Insert the finest point of your finest scissors in the atrial siphon and cut through its mantle wall to open the atrium. Continue cutting through the mantle in a ventral direction across the body anterior to the right gonad. Do not cut the gonad.
At the ventral end of the gonad turn the incision posteriorly around the gonad and renal sac and then dorsally along the posterior margin (greater curvature) of the renal sac. Stop cutting when you reach the dorsal end of the sac.
The gonad and renal sac are now freed on three sides but remain intact and attached dorsally, at the base of the atrial siphon. The oviduct has not been harmed. The atrium has been opened and is most of the space you see surrounding the organs. Deflect the renal sac and right gonad dorsally, cutting the delicate, transparent blood vessels that connect them to the body as necessary. With most of the right body wall deflected, look again at the lesser curvature of the renal sac and see if the heart is still beating. If it is, it will be easier to see from this, the right, side.
Gonads
Both gonads are now visible. With fine scissors snip the ventral end off the tip of the right gonad (including part of the ovary and testis). Set the tip aside for the moment and inspect the cut surface of the ovary and note that it is hollow. Carefully insert the tip of a minuten nadel in it. Look at the opposite, dorsal end of the ovary and find the wide, short oviduct where it exits the ovary and is free of it. Follow the oviduct to its terminus at the female gonopore where it empties into the atrium. Insert a nadel into the gonopore and see that it slips easily into the lumen of the hollow ovary. The ovarian lumen is a remnant of the coelom. Eggs are shed into it, move through the oviduct, enter the atrium, and are then released through the atrial siphon.
>1f. Place the tip of the gonad in a drop of seawater (tapwater if using preserved material) on a slide, tease it apart, and make a wet mount with it. Examine the slide with the compound microscope and locate the large irregular eggs. Find an isolated egg and note that it is surrounded by numerous small follicle cells. Use high power to look at the remains of the testis that you teased apart. If you have a living specimen, you should see numerous tiny swimming sperm. Observe them and make a sketch of one of them. <
While the right gonad is deflected, look at its inner (left) surface and find the sperm duct, or vas deferens. It runs longitudinally along the margin of the testis and on the left surface of the ovary. It receives numerous dendritic branches from the testis and empties via several seminal papillae into the atrium, but not at the atrial siphon.
According to most accounts, Molgula manhattensis is an oviparous species in which gametes are released to the sea where fertilization and development occur. Some species ofMolgula are viviparous and retain eggs in the atrium where they are fertilized and develop. These species brood their embryos and release tadpole larvae from the atrium. Some viviparous species are easily confused with M. manhattensis and collections from the American northeast may contain some specimens of a viviparous species.
If your specimen has large eggs and/or embryos in the atrium, it is one of the viviparous species and you should take advantage of the opportunity to study development in species that brood their embryos. First, inform the instructor of your good fortune and be prepared to share the embryos with the rest of the class.
>1g. Make a wetmount with a supported coverslip, in seawater, of several of the embryos. Look at the embryos with the compound microscope. You can probably find all developmental stages from unfertilized eggs to tadpole larvae. All will be about the same size.
Unfertilized eggs will not have an elevated egg membrane but will have a large, conspicuous, clear, spherical nucleus. At 400X, yolk platelets can easily be seen in the cytoplasm of the unfertilized egg, and of all other stages, for that matter. Zygotes will resemble the unfertilized egg in being a single cell but they will have an elevated egg membrane. Look for two-cell, four-cell, and more advanced stages up to tadpole larvae.
The numerous small cells around the periphery of the embryo, but inside the egg membrane, are "test" cells. Their function and origin is not well understood.
Flattened follicle cells can be seen adhering to the outside of the egg membrane. These cells are spherical when they surround developing ova but flatten as the egg membrane develops.
Some of the embryos will probably be recognizable tadpole larvae curled within the egg membrane (Fig 29-24). The tail of the larva is wrapped around the body. The large vacuolated cells of the notochord can be seen easily. The black statolith in the larval cerebral vesicle can also be seen but no ocellus is present in Molgula tadpoles. Using 400X, focus on the surface of the embryo and you will see the numerous small individual cells of its epidermis. Their nuclei can also be seen.
Pharynx
The very large, thin, delicate pharynx underlies most of the mantle and extends dorsally and posteriorly from the buccal siphon. The open space surrounding the pharynx is the atriumand it is continuous with the sea outside via the atrial siphon, as you have demonstrated. The atrium is an ectodermal invagination that surrounds the pharynx on the dorsal and lateral, but not the ventral, surfaces.
" Gently tear or cut the numerous connections between the mantle and the pharynx on the right side but avoid damaging the pharynx. These points of attachment of the connective tissue of the mantle to the wall of the pharynx are the sites of entry of blood vessels into the pharyngeal wall. Remove the mantle from the right side of the specimen.
>1h. Make a wetmount of a small square of the mantle from the vicinity of the siphons and examine it with the compound microscope. Note the widely spaced, relatively weak transverse, longitudinal, and oblique muscle bands. <
With the mantle removed you will have a better look at the renal sac. See if the heart is still visible. If it is still beating take advantage of the clearer view you now have and make any observations you were unable to make earlier with the mantle still in place.
Once you have removed the mantle, the wall of the pharynx (variously known as the branchial basket, branchial sac, pharyngeal basket, or pharyngeal sac) will be exposed. Most of the tissue you see inside the mantle is the pharynx.
" Use fine scissors to open the pharynx by cutting through the right pharyngeal wall from the anterior buccal siphon to the posterior esophagus (Fig 29-15A). To do this insert one blade of a fine scissors in the buccal siphon and cut posteriorly on the right side until you reach the posterior end of the pharynx at the pyloric gland. Deflect and pin the pharyngeal wall and examine the interior. Relocate the opened buccal siphon at the anterior end and the opening to the esophagus at the opposite end.
The pharyngeal wall is perforated by minute gill slits (= stigmata). Find an area of the pharynx wall that is over a dark background and look at it with the highest power of your dissecting microscope. Against such a background you can barely make out the maze of tiny, elongate, curved gill slits in the wall. The gill slits of molgulids are spirals rather then the more usual ovals (Fig 29-26D).
Six longitudinal ridges or folds, the branchial pleats, are present on each side (right and left) of the inner wall of the pharynx. These pleats are an apomorphy of Stolidobranchia (Fig 29-26D). Several longitudinal blood vessels are contained within each pleat but the number varies. Transverse vessels of different sizes exit the longitudinal vessels and run between successive branchial pleats and their vessels. The largest ones are easily seen and are about the same size as the longitudinal vessels in the pleats. The pharynx is well supplied with blood vessels and functions as the respiratory surface as well as a filter for feeding.
Find the dorsal and ventral midlines of the pharynx and locate the endostyle on the ventral midline (Fig 29-15A,B,C). It extends from the base of the buccal siphon to the posterior end of the pharynx. It is a narrow, wavy, ciliated groove lying between two wide ridges (Fig 29-15C). It is wide and conspicuous and easily recognized. It superficially resembles one of the many branchial folds in the pharynx wall but on closer inspection will be seen to be entirely different.
The endostyle is composed of ciliated and glandular cells. The glandular cells secrete an iodine-containing mucous net (Fig 29-15D) and the endostyle is homologous to the vertebrate thyroid gland (which is also an endodermal derivative of the floor of the pharynx). Frontal cilia on the inner surface of the pharynx move the mucous net dorsally to cover the inner wall and the gill slits. Water passes through the gill slits to the atrium but food particles are retained by the mucous net and remain in the lumen of the pharynx. The mucous net greatly reduces the pore size of the pharyngeal filter making it possible to utilize smaller food particles.
On the dorsal midline of the pharynx, opposite the endostyle, is the less conspicuous dorsal lamina ( Fig 29-15B). This is a narrow, longitudinal, ciliated ridge and gutter. It begins at the base of the buccal siphon and runs to the esophagus.
At the buccal siphon the dorsal lamina is joined by two circumferential ciliated ridges, the peripharyngeal bands that begin at the anterior end of the endostyle and run around both sides of the top of the pharynx at the base of the buccal siphon (Fig 3).
The mucus and trapped food moves dorsally from the endostyle and is gathered by the dorsal lamina and rolled into a string. The ciliary current of the lamina moves the mucus/food string posteriorly to enter the esophagus. In many tunicates (eg Ciona) the dorsal lamina is replaced by a longitudinal row of papillae, the dorsal languets, that perform the same function.
>1i. With fine scissors, remove a small piece of pharyngeal wall being sure to include a branchial pleat and some transverse vessels. Make a wet mount, being sure the tissue is not folded over itself, and examine it with the compound microscope. Note the spectacular array of short, curved gill slits. Look at them with 100X, then 400X. Find the conspicuous fringe oflateral cilia on the margins of the gill slits. These are the cilia that generate the flow of water in the buccal siphon, through the gill slits, and into the atrium. Regardless of length, the width of the gill slits is uniform and is about double the length of the lateral cilia. Another set of cilia, the frontal cilia, move the mucus net dorsally from the endostyle to the dorsal lamina. Frontal cilia are more difficult to see in this view as they extend vertically out of the plane of the slide and parallel to your line of vision so you are looking at them in end view.
Continuing your examination of the wetmount, look for longitudinal blood vessels in the branchial folds (Fig 29-26D). Look for transverse blood vessels leaving the longitudinal vessels and running perpendicular to them. Many sizes of transverse vessels are present and they branch repeatedly to supply the walls of the pharynx with blood. The spaces between the gill slits are the blood spaces and they contain abundant corpuscles. If the blood is moving, they are easily recognized for what they are and the extent of the blood vessels into all the connective tissue spaces between the gill slits is readily apparent also. <
>1j. Return to the dissected animal and use a Pasteur pipet to place some carmine particles suspended in seawater on the surface of the endostyle, pharyngeal wall, and dorsal lamina. Watch closely for ciliary currents. <
>1k. Place a drop of 1% methyl green/seawater on the inside surface (the surface facing you) of the left wall of the pharynx. Some of the dye may be moved dorsally by the frontal cilia but most of it should be moved rapidly through the walls of the pharynx by the lateral cilia.<
Neural Gland and Cerebral Ganglion
At the anterior end of the dorsal lamina, on the dorsal midline between the atrial and buccal siphons, is a large, spherical dorsal tubercle (Fig 3, 29-23). The two peripharyngeal bands that encircle the base of the buccal siphon pass around the dorsal tubercle to join the dorsal lamina.
The surface of the dorsal tubercle bears a C- or horseshoe-shaped groove that is the opening of a ciliated duct that leads to the neural gland (Fig 29-23). Through this opening seawater enters the neural gland from whence it is admitted to the hemal system to replace fluid lost from the blood vessels of the gills.
The opaque, oval neural gland is easily seen in the tissue dorsal to and close to the dorsal tubercle (Fig 3). It is about the same size as the tubercle. A homology between the neural gland and the vertebrate anterior pituitary gland, as well as the echinoderm madreporite and stone canal, has been proposed. Despite its name, the neural gland contains no neurons and has no nervous role.
The nervous system of adult ascidians is very simple and the dorsal hollow nerve cord and sense organs of the larva are absent. The cerebral ganglion, or brain, is a long, narrow (fusiform), opaque, white (in life) structure lying between the dorsal tubercle and the neural gland (Fig 29-23). Its long axis parallels the dorsal lamina. The neural gland is dorsal to the cerebral ganglion in the stolidobranchs, whereas in most ascidians the positions are reversed and the ganglion is dorsal to the gland. A nerve cord exits each end of the ganglion and both branch soon after leaving the ganglion.
Numerous small, branched buccal tentacles surround the opening of the buccal siphon into the pharynx (Fig 3, 29-15A). These tentacles are sensory and, when stimulated with chemicals or touch, elicit contraction of the mantle muscles and expulsion of the water in the pharynx, along with the offending material. You may have seen the effect of these tentacles when you tried to introduce dyes or carmine into the buccal siphon. The tentacles also prevent the entry of large particles. The ring of tentacles marks the end of the buccal cavity and beginning of the pharynx.
Figure 3. View of the dorsal midline of the anterior end of the pharynx of Ciona intestinalis, viewed from inside the pharynx. The pharynx has been opened and its walls deflected. Uro77La.gif
Posterior Digestive System
Trace the dorsal lamina to the postero-dorsal corner of the pharynx and relocate the opening of theesophagus. In life the esophagus and its opening are white but are surrounded by the orange or brown mass of the pyloric gland (= liver, = hepatic gland). Insert the nadel into the opening of the esophagus and trace it posteriorly to the stomach.
" Remove the left wall of the pharynx so the entire double loop of the gut is exposed. You are looking at the right side of the gut. Find the opening of the esophagus again and insert the fine point of your fine scissors into it. Cut along the length of the esophagus, stomach, and intestine.
Look inside the lumen of the gut and observe that the pyloric gland is the thickened, folded, glandular epithelium of the stomach.
The stomach gradually narrows to become the intestine but the separation is indistinct. A large typhlosole, or longitudinal fold of the gut wall begins at the distal end of the esophagus and bulges into the intestine for almost its entire length.
The intestine may contain mucous strings originally fabricated by the dorsal lamina from mucus secreted by the endostyle. Trace the intestine for its entire length, opening it as you go. It eventually ends at the anus, which opens into the atrium at the base of the atrial siphon. Note that the typhlosole ends just short of the anus.
The inner, or right, surface of the left gonad is apparent in the lesser curvature of the gut loop. It is composed of a central, hollow ovary with a lobulated, peripheral testis on both margins.
Tadpole Larva
Examine a commercially prepared wholemount of a tadpole larva of unknown species. Very little internal structure is discernable in most commercial slides but you can see the general shape of the larva and a few structures. It consists of an ovoid anterior body with a long, slender, muscular, posterior tail. In some preparations a thick, transparent, almost invisible tunicsurrounds the body. The notochord is usually visible in the center of the tail. Within the body the cerebral vesicle (= sensory vesicle) should be apparent. It contains at least one (as inMolgula), but more often two, opaque spheres. One is a statocyst for gravity detection and the other is an eyespot for photoreception. The cerebral vesicle will become the cerebral ganglion of the adult. Two adhesive papillae are present at the extreme anterior end. These will be used to attach to the substratum when the larva leaves the plankton and begins its sessile adult life. The gut, with nonfunctional pharynx, endostyle, and intestine are usually an amorphous mass whose details cannot be recognized in these slides. Buccal and atrial siphons, although present, are neither open nor functional.
References
Alder J, Hancock A. 1907. The British Tunicata, vol II. Ray Society, London. 164p, 50pl.
Barrington , E.J.W. 1965. The Biology of Hemichordata and Protochordata . Oliver and Boyd, Edinburgh. 176p.
Berrill NK. 1950. The Tunicata, with an account of the British species. Ray Society, London. 354p.
Burighel P, Cloney RA. 1997. Urochordata: Ascidiacea, pp 221-347 in Harrison FW, Ruppert
EE (eds.) Microscopic anatomy of invertebrates. Wiley-Liss, New York. 537pp.
Kleinholz LH. 1950. Molgula manhattensis. in F. A. Brown, Selected Invertebrate Types. Wiley, New York. 597p.
Parker TJ, Haswell WA. 1921. A Text-book of Zoology, vol II. MacMillan. London. 714p.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Van Name WG. 1945. The North and South American ascidians. Bull. American Mus. Nat. Hist. 84:1-476, 31 pls.
Supplies
Dissecting microscope
Compound microscope
8-cm culture dish
seawater
Small dissecting pan (sardine tin with wax bottom)
# 1 stainless steel insect pins
Dissecting set with microdissecting tools
Isotonic magnesium chloride
Molgula manhattensis , living (preferred) or preserved.
0.1% methyl green/seawater
1% methyl green seawater
carmine/seawater suspension
toothbrush
Pasteur pipet
Hypodermic syringe
Living Molgula manhattensis are available from Woods Hole Marine Biological Lab. Preserved specimens from Wards Natural Science.