Invertebrate Anatomy OnLine
Metridium senile ©
Anemone
10jul2006
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises can be accessed by clicking on the links to the left. A glossary and chapters on supplies and laboratory techniques are also available. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Cnidaria P, Anthozoa C, Zoantharia sC (=Hexacorallia), Actiniaria O, Sagartiidae F (Fig 7-42, 7-75)
Cnidaria P
The cnidarian body consists of a central blind sac, the coelenteron (= gastrovascular cavity), enclosed by a body wall comprising two epithelia, the outer epidermis and the inner gastrodermis (Fig 7-1, 7-2). A gelatinous connective tissue layer, the mesoglea, lies between the two epithelia. The mouth opens at one end of the coelenteron and marks the oral end. The mouth is at the tip of a process, the manubrium that elevates it above the oral surface. The opposite pole is the aboral end. The imaginary line connecting the oral and aboral poles is the axis of symmetry around which the radial symmetry of the body is organized. The mouth is usually surrounded by one or more circles of tentacles.
The defining cnidarian feature is, of course, possession of stinging cells, or cnidocytes (Fig 7-8). Characteristic of the epidermis, they are also sometimes found in the gastrodermis. Cnidocytes contain an explosive organelle, the cnida, which, upon proper stimulation, inverts and ejects a slender, often barbed and toxic thread in the direction of prey or predator (Fig 7-9). Three types of cnidae are found in cnidarians (Fig 7-10). Nematocysts (in nematocytes), spirocysts (in spirocytes), and ptychocysts (in ptychocytes). All toxic cnidae are nematocysts whereas spirocysts are sticky, and the everted tubules of ptychocysts are used for constructing feltlike tubes. Most cnidae are nematocysts and these are present in all three higher cnidarian taxa. Spirocysts and ptychocysts are found only in Anthozoa.
The basic body plan described above can be manifest as a swimming medusa or attached polyp. In some taxa only one generation is present whereas in others both are found. A life cycle featuring alternation of sexual, swimming medusae with benthic asexual polyps is typical of many cnidarians.
All cnidarians are carnivores feeding on live prey which they usually capture using tentacles armed with cnidocytes. Digestion occurs in the coelenteron which is typically equipped with ciliated canals for distribution of partly digested food. Cnidarians are ammonotelic and diffusion across the body and tentacle surface eliminated the ammonia from the body. Gas exchange is across the general body surface. The nervous system is a plexus of basiepithelial neurons serving sensory and motor systems (Fig 7-6). Most cnidarians are gonochoric. The life cycle typically includes a planula larva. Cnidarians are chiefly marine but the well-known Hydra is an exception.
Anthozoa C
This heterogeneous taxon includes stony corals, sea fans, anemones, sea pansies, and others. Anthozoa is the largest cnidarian taxon, with over 6000 species of exclusively marine polyps. Anthozoans are always polyps, with no medusa in the life cycle. The polyps can be solitary or colonial. The polyps tend to be large, with extremes up to 1 m in diameter, and colonies, such as the corals, may be even larger. The mesoglea contains cells and all three types of cnidae are present, the only taxon for which that is the true. The mouth opens into a flattened, ectodermal invagination known as the pharynx, which in turn opens into the coelenteron. Many have endosymbiotic zooxanthellae that figure importantly in their biology. The coelenteron is compartmentalized by longitudinal septa and gonads are gastrodermal.
Zoantharia sC
This, the largest anthozoan taxon with 4000 species, includes sea anemones and stony corals. The septa that partition the coelenteron are in multiples of six and usually occur in pairs. The gonads are in the septa. Cnidae consist of nematocysts, spirocysts, and ptychocysts with spirocysts and ptychocysts being found nowhere else.
Actiniaria O
Actiniaria, the sea anemones, are solitary and have the morphology typical of anthozoan polyps (Fig 7-20). Anemones are relatively large polyps, compared with hydrozoans and scyphozoans, with most being less than 10 cm long and 5 cm in diameter but some are much larger. Anemones can dramatically alter their size and shape by inflating (with seawater) or deflating (Fig 7-19). About 1350 species are known. Most anemones attach to a firm substratum by the pedal disc (Fig 7-22) but many inhabit soft substrata and do not attach (Fig 7-21). The periphery of the coelenteron is partitioned by abundant longitudinal, sheetlike septa which greatly increase the surface area for absorption of food and its subsequent intracellular digestion. The central region of the cavity is unpartitioned and is the site of extracellular digestion. The free edges of the septa border the central region and secrete hydrolytic enzymes into it. Polymorphism and alternation of generations are absent. Most are gonochoric.
Laboratory Specimens
This exercise is written for preserved specimens of Metridium. If living specimens are to be used then the Aiptasia/ Metridium exercise should be used instead of this one. Metridium senile is a common anemone in the northeastern United States and in northern Europe. It inhabits shallow, subtidal water and lives attached to rocks. It is a relatively large species. It does not harbor zooxanthellae. Metridium has abundant short tentacles.
Anatomy
External Anatomy
Examine a preserved specimen with the dissecting microscope. The specimen should be in a deep dissecting pan or culture dish and immersed in tapwater.
The body of a typical anemone is a cylindrical column with a crown of tentacles at one end and an adhesive pedal disk at the other (Fig 1). The column is hollow and contains the coelenteron, or gastrovascular cavity.
Each end of the column is a disc. The basal, attached end of the column is the aboral end, terminating in the pedal disc. It is usually attached to a firm substratum such as rock or shell.
The oral end is free in the water and terminates in the oral disc. The mouth lies in the center of the oral disc. In Metridium there is a circumferential fold, or collar, around the column that divides the column into two regions. The collar overhangs a deep fold, the fossa. The collar covers the oral disk when the anemone contracts and withdraws its tentacles.
Clusters of very fine unbranched tentacles encircle the oral disc and cover its surface. The tentacles are hollow evaginations of the body wall and contain extensions of the coelenteron. Their epidermis contains cnidocytes used for stinging.
The mouth is an elongate slit in the center of the oral disk. It is expanded slightly at each end. These expansions are the ends of vertical, ciliated grooves called siphonoglyphs. Most Metridium specimens have two siphonoglyphs but some have only one and some may have three.
Siphonoglyph cilia generate a constant flow of water into the gastrovascular cavity even when the mouth is closed. This maintains hydrostatic pressure in the cavity against which the musculature can act to keep the column turgid. The mouth opens into a short, flattened tube, the pharynx, which opens into the coelenteron. With the blunt probe open the mouth so you can see the pharynx. The siphonoglyphs are at opposite edges of the flat pharynx.
Many anemones, Metridium included, have numerous blisterlike openings scattered about the lower part of the column. These are cinclides through which the animal expels long thread-like acontia. The epithelium of the acontium bears large numbers of cnidocytes. Many anemones do not have acontia or cinclides.
The elongate mouth and flat pharynx destroy the otherwise perfect radial symmetry of anemones and confer upon them a biradial symmetry. Such a body plan has two planes of symmetry rather than the several planes of a radially symmetrical animal or the single plane of a bilaterally symmetrical one. Both planes of symmetry are longitudinal and are at right angles to each other. Both include the aboral-oral axis, which is the axis of symmetry.
Figure 1. Metridium exile. Most of the acontia have been truncated for clarity. Redrawn and adapted from Hyman (1940)and Ruppert, Fox, Barnes (2004). Anthozoa62L.gif
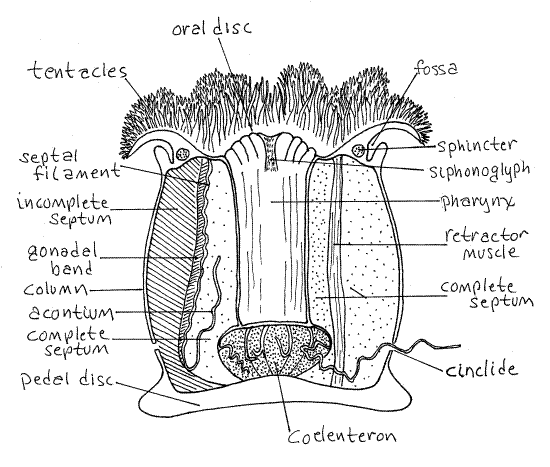
One plane passes through both siphonoglyphs whereas the other is perpendicular to it and divides the mouth into two ends, each with a siphonoglyph. Some anemones have only one siphonoglyph and are thus bilaterally symmetrical with only one plane of symmetry.
Figure 2. Make sketches of oral disks to show how the shape of a straight mouth with two siphonoglyphs imposes biradial symmetry on the oral disk and how a single siphonoglyph results in bilateral symmetry. anthozoa76L.gif.
Internal Anatomy
& Place the specimen in a wax-bottom dissecting pan if it is not already in one. Insert the point of a pair of heavy scissors through one of the siphonoglyphs into the pharynx and cut through the body wall in the plane passing through the two siphonoglyphs. Cut all the way to the pedal disk. At the sink, rinse the coelenteron with a gentle stream of water. &
Observe that the upper end of the animal consists of a flattened tube, the pharynx, within a larger tube, the body wall. The space between is the coelenteron. The pharynx extends only part of the way down the column and is connected with the body wall by sheets of tissue, the septa (sometimes called mesenteries).
Pull the two sides of the animal apart and pin them firmly to the wax with stout pins. This will hold the coelenteron open. Be sure the tissues are completely submersed in water.
Examine the specimen without magnification. Find the opened pharynx. It is a flat, pleated tube extending from the mouth into the coelenteron. Find its aboral end. Slip the blunt probe under the free margin of the pharynx into the space between the pharynx and the body wall.
Find some of the numerous septa that extend from the body wall into the coelenteron (Fig 1, 7-16). Much of the space in the cavity is filled with these septa. Your best view of an isolated septum is near the pedal disk. The septa extend as partitions from the pedal disk to the oral disk, near midbody they disappear from view behind the pharynx.
The largest septa are the complete septa which, in the oral end of the coelenteron, extend from the body wall to the pharynx. Use the blunt probe to reveal some complete septa at the edge of the pharynx.
Examine two successive complete septa at their junction with the pedal disk. Move them away from each other and find the smaller incomplete septa between them. Incomplete septa also extend from oral to pedal disc but do not extend far enough into the coelenteron to reach the pharynx.
Follow one complete septum upward from the pedal disk until its free edge becomes a heavily convoluted septal filament. At the point where the convolutions begin a long free filament, the acontium, arises and extends threadlike into the coelenteron.
Place the dissecting pan on the stage of the dissecting microscope and support it with blocks of wood so it cannot tilt and spill. Examine the inner surface of the body wall with low power. The free, unattached, inner edges of the septa are thickened and are the septal filaments. Each filament is composed of three side by side lobes running the length of the filament from the end of the pharynx to the pedal disk (Fig 7-18). They are best developed near the pharynx. They are longer than the distance between the end of the pharynx and the pedal disk and consequently are wavy and folded on themselves, especially near the pharynx.
Look at the aboral end of a filament with the highest power of the dissecting microscope. You will be able to see clearly the three lobes of which it is composed. Consult your textbook to review the functions of the three lobes.
A single, long, thread-like acontium arises from the aboral end of the middle lobe of each septal filament. Much of the space in the coelenteron is filled with filaments and acontia. Follow the free edge of a septum down into the column, noting that it becomes less convoluted near the pedal disk. The acontia arise from the lower, unconvoluted portion of the septum. Acontia have abundant cnidocytes.
In addition to the complete septa there are numerous incomplete septa that arise from the inner wall of the column (as do the complete septa) but these do not extend all the way across the coelenteron and do not join the pharynx. Look at the spaces between the complete septa and find some of the smaller incomplete septa arising from the body wall.
Spread and flatten one of the complete septa as much as you can and look for the retractor muscle (Fig 1, 7-18A,B). This is a broad, thin, longitudinal band of muscle running the length of the septum from oral to pedal disk. Its fibers are visible but its greater thickness may not be appreciable. Contraction of these muscles shortens the column and withdraws the tentacles and oral disk into the column. There is a circumferential sphincter muscle of circular fibers around the oral end of the column to close the oral end over the retracted tentacles (Fig 1).
Anemones have circular muscles in the body wall of the column which pressurize the coelenteron. In addition, there are radial muscles in the septa. These run from pharynx to the body wall and by contracting open the pharynx and mouth. During contraction, the anemone opens the mouth and contracts the longitudinal muscles to expel the water from the coelenteron.
Metridium is hermaphroditic but each individual produces only one kind of gamete at any one time. The gonads are longitudinal bands of gamete-producing tissue on the edges of the septa just peripheral to the septal filaments and central to the longitudinal muscles (Fig 1, 7-18A). The gonads are usually located on the incomplete septa but are not always present.
*Hyphenated call-outs, such as this one, refer to figures in Ruppert, Fox, and Barnes (2004). Those without hyphenation refer to figures embedded in this exercise.
References
Barham EJ, Pantin CF . 1951. The organization of the muscular system of Metridium. Quart. J. Micros. Sci. 92:27-54.
Fautin DG, Mariscal RM. 1991. Cnidaria: Anthozoa. Pp. 267-358 in F.W. Harrison & J.A. Westfall (eds). Microscopical Anatomy of Invertebrates, vol. 2. Wiley-Liss, New York.
Hyman LH. 1940. The Invertebrates: Protozoa through Ctenophora, vol. I. McGraw Hill, New York. 726 p.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Stephenson TA. 1928. The British Sea Anemones. Ray Society.
Supplies
1 Metridium
1 dissecting pan
1 dissecting microscope
2 wooden blocks to support the dissecting pan on the microscope stage