Invertebrate Anatomy OnLine
Lumbricus terrestris ©
Earthworm
28may2007
Copyright 2003 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises, a glossary, and chapters on supplies and laboratory techniques are also available at this site. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Annelida P, Clitellata, Oligochaeta C, Haplotaxida O, Lumbricina sO, Lumbricoidea SF, Lumbricidae F (Fig 13-7A)
Annelida P
Annelida consists of the segmented worms in the major taxa Polychaeta (bristleworms), Oligochaeta (earthworms and relatives), Branchiobdellida (crayfish ectosymbionts), and Hirudinea (leeches) with a total of about 12,000 known species in marine, freshwater, and terrestrial environments. The segmented body is composed of an anterior prostomium, a linear series of similar segments, and a posterior pygidium. The prostomium and pygidium are derived from anterior and posterior ends of the larva whereas the intervening segments arise through mitotic activity of mesodermal cells in the pygidium.
The body wall consists of a collagenous cuticle secreted by the monolayered epidermis. A connective tissue dermis lies beneath the epidermis. The coelom is lined by a peritoneum which may be specialized to form the body wall muscles. Most annelids have chitinous bristles, or chaetae, secreted by epidermal cells, that project from the body. The coelom is large, segmentally compartmented, lined by peritoneum, and well developed in polychaetes and oligochaetes but reduced in leeches. Successive coelomic spaces are separated by transverse bulkheads known as septa which consist of double layers of peritoneum with connective tissue in between. The right and left sides of each segmental coelom are separated by longitudinal mesenteries which, like septa, are double layers of peritoneum with connective tissue between.
The gut is a straight, regionally specialized tube that begins at the mouth at the anterior end and extends for the length of the body to end at the anus on the pygidium. It penetrates each septum and is supported by dorsal and ventral mesenteries. Like that of most invertebrates, the gut consists of ectodermal foregut, endodermal midgut, and ectodermal hindgut. The nervous system consists of a dorsal brain in or near the prostomium, a pair of circumpharyngeal connectives around the anterior gut, and a double, ventral nerve cord with paired segmental ganglia and nerves. The hemal system of most annelids is a set of tubular vessels, some of which are contractile and serve as hearts. The hemal system is absent or greatly reduced in leeches. The system includes a dorsal longitudinal vessel above the gut in which blood moves anteriorly, a ventral longitudinal vessel below the gut, in which blood moves posteriorly, and paired segmental vessels that connect the dorsal and ventral vessels. The digestive, hemal, and nervous systems are continuous and pass through the segments.
Respiration is accomplished in a variety of ways. In some, the general body surface is sufficient but gills are present in most polychaetes, many leeches, and a few oligochaetes. Excretory organs are metanephridia or protonephridia and typically one pair is present in each segment. These osmoregulatory organs are best developed in freshwater and terrestrial species. The sexes are separate in polychaetes but oligochaetes and leeches are hermaphroditic. In the ancestral condition paired submesothelial clusters of germ cells were present in each segment and released developing gametes into the coelom. In derived taxa reproductive functions tend to be confined to a few specialized genital segments. Gametes mature in the coelom or its derivatives and fertilization is external. Gametes are shed through ducts derived from metanephridia or by rupture of the body wall. Spiral cleavage follows fertilization. Clonal reproduction is common.
Clitellata
Clitellata, the sister taxon of Polychaeta, includes earthworms and their allies and the leeches. The head is reduced and its sensory functions minimized. Clitellates lack the parapodia characteristic of polychaetes although chaetae may be present. Also lacking are head and pygidial appendages. Present however, is a girdle-like band of secretory epidermis, the clitellum, near the anterior end of the worm. The brain has moved out of the prostomium into a more posterior segment. These worms are hermaphroditic and have gonads and reproductive equipment restricted to a few specialized genital segments. Development is direct, without a larva.
Oligochaeta C
Most of the 3500 known species of oligochaetes are small annelids found in benthic aquatic habitats, mostly freshwater, but about 200 species occur in the sea. The taxon also includes the large terrestrial earthworms, some of which reach lengths of three meters.
Laboratory Specimens
Of the many terrestrial earthworms in Lumbricidae, Lumbricus terrestris, the night crawler, is the best known, mostly because it is a favorite fishing bait. Because of its availability and large size, it is a good subject for a laboratory study of oligochaete anatomy. Living worms are readily available from bait shops and are easily anesthetized in 5-7% non-denatured ethanol in tapwater. Worms should be placed in the anesthetic about 15-30 minutes prior to the beginning of the laboratory period. Preserved worms may be used if necessary but the following description applies specifically to fresh specimens. This is a rare instance in which living material is less expensive and easier to obtain than preserved and it should be taken advantage of. Living, anesthetized worms are far, far superior to preserved worms for the study of oligochaete anatomy.
External Anatomy
Study an anesthetized living Lumbricus in a dissecting pan of 5-7% ethanol on the stage of the dissecting microscope. The liquid must be deep enough to completely cover the worm.
Orientation
Examine the external features of the worm (Figs 1, 2). The anterior end is usually larger than the posterior and is round in cross section whereas the posterior tends to be flattened dorsoventrally. The dorsum is dark and rounded while the venter is pale and slightly flattened. The dark dorsal blood vessel can be seen through the body wall on the dorsal midline. Find right-left, dorsal-ventral, anterior-posterior, and the plane of symmetry.
Figure 1. A dorsal view of the anterior end of Lumbricus. The segments are numbered. Oligo54L.gif
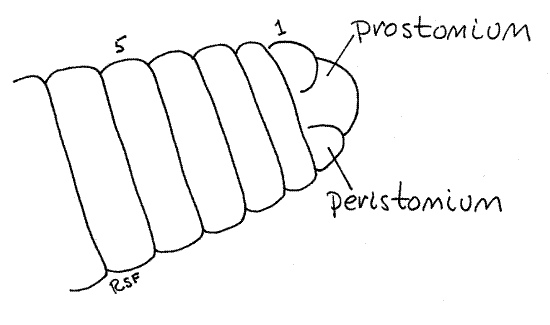
Clitellum
A band of thickened secretory epithelium, the clitellum (clitell = saddle), girdles the body near the anterior end. The clitellum secretes a mucous cocoon, into which gametes and albumen are released and where fertilization occurs.
Segments
The body is segmented and each segment is separated from its neighbors by a distinct circumferential groove. The anteriormost true segment is the peristomium (peri = around, stome = mouth; Fig 1, 13-59A). Anterior to the peristomium is a small dorsal lobe, the prostomium (pro = before) that, for embryological reasons (it is not a product of the teloblast cells), is not considered to be a segment. The peristomium encircles the large, ventral mouth and is an almost complete ring around the body (Figs 2, 4, 13-59A). The prostomium fits into a small, dorsal notch in the peristomium. The prostomium is a small lobe dorsal to the mouth and anterior to the peristomium. The segments are numbered anterior to posterior beginning with the peristomium.
Count the preclitellar segments in your specimen. ____________ How many segments contribute to the clitellum? _____________ Compare your counts with those made by other students. Does the number of segments in each region appear to be constant or variable? ____________
The segments posterior to the peristomium are complete rings, without notches, and extend uniformly for the entire length of the worm. The posteriormost division of the body is thepygidium, which encircles the anus at the posterior tip of the worm. Like the prostomium, and for the same reasons, the pygidium is not considered to be a true segment.
Chaetae
The eight small chaetae on each segment (except the first) are usually visible with adequate magnification (25X). Chaetae are small chitinous bristles emerging from pores in the integument on the ventral half of the worm. The chaetae are arranged in four pairs, two on each side (Fig 6, 13-61A). Each segment has a pair of lateral chaetae and a pair of ventral chaetae on each side. The chaetae are used as anchors when burrowing to hold parts of the worm against the so that elongation of the animal results in controlled, usually forward, motion.
The chaetae are equipped with protractor and retractor muscles and are retractile. In your specimen they may be withdrawn in some, or even all, of the segments. Careful examination, however, should reveal some segments with extended chaetae. Chaetae are amber or brown, slightly curved, and short.
Because
the chaetae are sometimes used as landmarks to describe the position of
other structures, it is useful that they be given names. The
four chaetae of each side of the segment are designated with the names chaeta a,
chaeta b, chaeta c, and chaeta d moving
from ventral to dorsal (Fig 6).
Figure 2. A ventral view of Lumbricus. The worm has been rolled slightly to reveal part of the right side. Oligo55L.gif
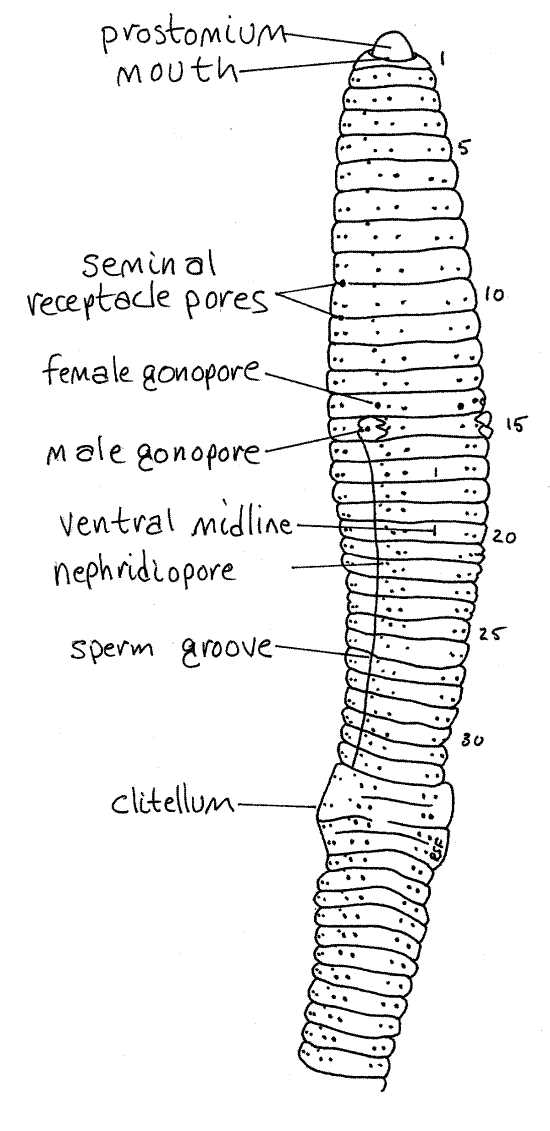
>1a. Rinse an unanesthetized worm with tap water and hold it in your hand on the stage of the dissecting microscope. Focus on the lateral body wall and watch the chaetae. You may see the animal retract or protract some of its chaetae while you watch. <
>1b . Run your fingers along the ventral and lateral surfaces of your anesthetized specimen from anterior to posterior and then from posterior to anterior. Try to feel the chaetae and determine if they point anteriorly or posteriorly. <
Pores
A little dorsal and anterior to chaeta b on each side of most segments, is a small opening, the nephridiopore, near the anterior edge of the segment (Fig 2). This is the external opening of a metanephridium, or kidney, a pair of which being present in most segments. The nephridiopores are closed by sphincters and are very difficult to see when closed.
Earthworms are simultaneous hermaphrodites and each individual has complete female and male reproductive systems including separate external gonopores. With the exception of the male gonopores, these openings are small and difficult to find. They are best demonstrated with a bent microneedle, which can be used to probe the surface of the cuticle until it slips into the previously invisible opening.
On the ventral surface of segment 15 are conspicuous swellings associated with the openings of the male vas deferens. The openings themselves are more difficult to see but each is flanked by two low, glandular, transverse ridges, or lips, that are easily seen. The openings are the male gonopores (Fig 2, 13-9A). During copulation sperm from the testes exit the body via these pores. A shallow, inconspicuous sperm groove extends posteriorly from each male gonopore to the clitellum and lies immediately lateral to chaeta b.
The two female gonopores, on segment 14 lateral to chaeta b, are the openings of the oviducts. These can best be demonstrated by probing with a microneedle directed anteriorly. Sometimes they are easy to see and sometimes not. Eggs from the ovary are released from these pores.
The two pairs of seminal receptacles belonging to the female system are located in segments 9 and 10 and their ducts open laterally in the grooves between segments 9-10 and 10-11 respectively (Fig 2, 13-59A). The four seminal receptacle pores may be difficult to find. Look for them beside chaetae c and d.
All segments posterior to 12 bear a tiny coelomic pore located on the dorsal midline in the groove between adjacent segments. These are almost impossible to see but can be demonstrated relatively easily with the microneedle. They are often easier to see along the flatter posterior portion of the worm. They open into the coelom and are used to leak coelomic fluid onto the surface of the animal to keep it moist. The body surface is the respiratory surface and it must be moist. Each pore has a sphincter to prevent unnecessary fluid loss.
Internal Anatomy
" Remove the worm from the dissecting pan and, while holding it in one hand, use your finest scissors to make a shallow, dorsal longitudinal incision a little to one side of the dorsal midline. Start by pinching the body wall with forceps and then cut through the pinch with the scissors. This initial cut should begin immediately anterior to the clitellum and initially should extend anteriorly for 2-3 segments. Be very careful that you do not cut the bright red middorsal blood vessel that can be seen through the body wall on the midline.
>1c. Press the incision against a glass slide to transfer some of the coelomic fluid to the slide then return the worm to the dissecting pan. To the slide, add a drop of 0.05% aqueous methylene blue and a coverslip. Use the compound microscope to study the coelomic corpuscles, or coelomocytes. Many are amoeboid and bear numerous short, narrow filopods, which move very slowly, whereas others have no pseudopods. Some of the cells in the coelom are free chlorogogen cells called eleocytes. <
Extend the dorsal incision anteriorly from the clitellum to the prostomium and posteriorly to about segment 30-40. Be sure it is a little to one side of the dorsal midline. Reflect the body wall and pin it to the wax with #1 insect pins as you go. Be sure the worm is in a convenient position on the dissecting pan before you pin it. Plan ahead so that you will be able to observe any part of the worm with the microscope after it is pinned. Insert the pins at 45 ° angles so you have plenty of working room above the worm. It is impossible to conduct the dissection if the pins are inserted vertically. Be sure the worm is completely immersed in 5-7% ethanol.
The body cavity, or coelom, is partitioned by septa, which are transverse sheets of thin tissue that extend from the body wall to the gut tube. You will have to cut many septa (carefully, with fine scissors) before you can move the body wall aside. Be careful that you do not cut any of the large red blood vessels although it is inevitable that you will cut a few of the smaller vessels.
Body Wall and Coelom
Look at the cut edge of the body wall and, with magnification, identify its major layers. Outermost is the thin, tough, iridescent cuticle composed of collagen fibers secreted by theepidermis beneath it (Figs 3, 6, 13-59B). The epidermis is a monolayered columnar epithelium containing numerous sensory and secretory cells. A subepithelial nerve plexus lies below the epidermis.
A thick and conspicuous layer of circular muscle lies inside the epidermis. In fresh preparations the circular muscle is reddish brown. The circular muscle fibers are divided into rings by the circumferential grooves between segments.
Inside the circular muscle layer is a thick layer of white or gray longitudinal muscle. Unlike the circular muscles, the longitudinal fibers run without interruption across segmental boundaries. While it is not obvious in gross dissection, the longitudinal muscle mass is divided into several bundles that run the length of the animal. You may be able to see the divisions between these bundles on the inside of the body wall.
The innermost layer of the body wall is the somatic peritoneum. This is a thin, glistening layer of squamous epithelium that covers the inner surface of the longitudinal muscles. A similar layer of splanchnic peritoneum covers the surface of the visceral organs.
Figure 3. The anterior end of a dissected earthworm. The nephridia, except for those of segment 7, have been omitted from the drawing. Oligo56La.gif
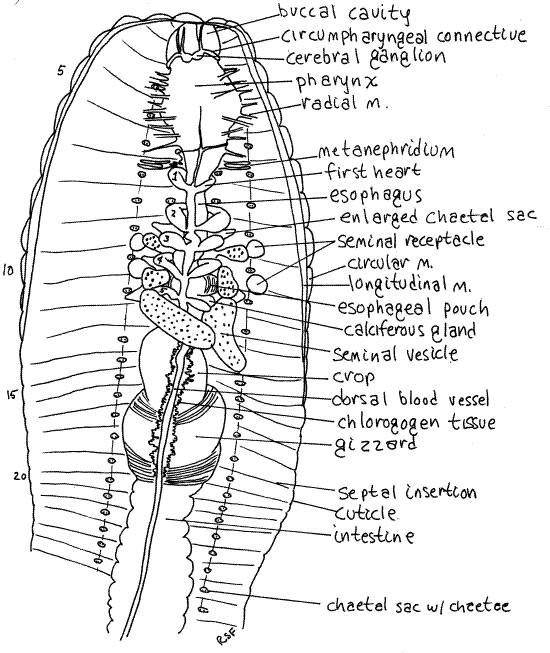
The open space enclosed by the two peritoneal layers is the body cavity, or coelom. It is divided into segmental coelomic compartments by double, vertical, transverse sheets of peritoneum, muscle, and connective tissue called septa. Septa are thin and transparent. Most of the septa were cut as you opened the body cavity but they may require additional cutting to reveal organs of interest. The septa are penetrated by the gut, hemal system, and nervous system. Longitudinal double sheets of peritoneum extend from the body wall to the gut tube and aremesenteries. The gut tube is suspended by the dorsal and ventral mesenteries.
In many areas the splanchnic peritoneum is specialized to form bright yellow or orange chlorogogen tissue (Figs 3, 6). Chlorogogen is involved in glycogen and lipid synthesis and storage, amino acid deamification, and the synthesis of ammonia and urea. It has a high iron concentration and may be the site of hemoglobin synthesis or breakdown. The color is due the lipid stored in it. Chlorogogen is derived from splanchnic peritoneum, which explains its position and distribution on the surface of the gut.
Chaetae
The four pairs of chaetae of each segment can be seen internally in four longitudinal rows along the floor of the coelomic cavity (Fig 3). The proximal heads of the chaetae are visible, with high magnification, as tiny paired black specks. Each pair is in a chaetal sac, which secretes the chaetae, and is attached to a set of tiny retractor and protractor muscles (Fig 13-59B).
The ventral chaetae of segment 26 are genital chaetae that help hold copulating worms together. Chaetae in some segments associated with reproduction may be reduced in size and the associated tissue modified for secretion.
Digestive System
Surface Features
Study the digestive system without damaging the hemal system. The gut is a straight tube extending from mouth to anus. It is regionally specialized to perform the several functions, including food procurement, storage, trituration, hydrolysis, absorption, reclamation, and feces storage and formation typical of complete digestive systems.
The annelid gut is composed of foregut, midgut, and hindgut of which the foregut and hindgut are ectodermal derivatives and lined with cuticle. The endodermal midgut does not secrete a cuticle. The mouth opens into the short, thin-walled buccal cavity in segments 1-3 (Fig 3, 4, 13-61B, 13-63A).
The pharynx is posterior to the buccal cavity (Figs 3, 4) in segments 3-5. The pharyngeal wall is thick and muscular. Numerous small radial muscles run from the pharynx to the body wall. Contraction of these muscles dilates the highly distensible pharynx.
Take note of, but avoid damaging, the conspicuous, white cerebral ganglia (brain) located atop the junction ofthe buccal cavity and the pharynx.
Posterior to the pharynx, the gut narrows to become the thin-walled esophagus in segments 6-12 (Figs 3, 4, 13-61B). The posterior end of the esophagus is hidden by six large, creamy white seminal vesicles that arch over it. Follow the esophagus through this region and note that it is not a simple tube as it is anteriorly.
Figure 4. The anterior end of a dissected earthworm viewed from the dorsum. The blood vessels, septa, and nephridia have been removed to reveal the gut and reproductive system. Oligo57La.gif
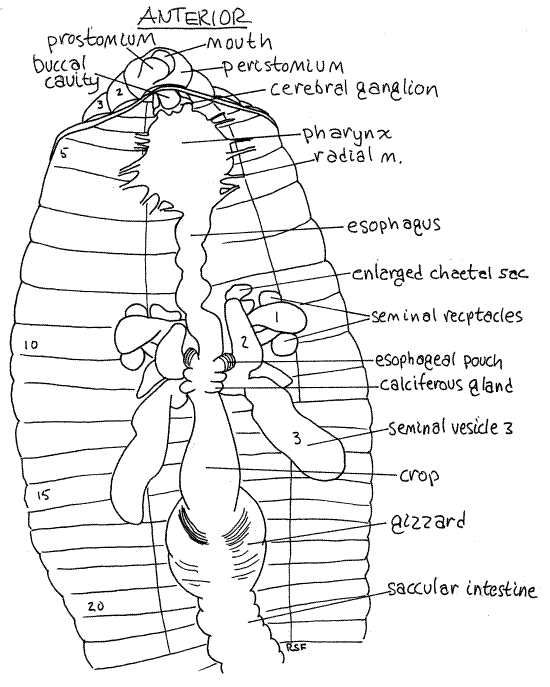
In about segments 10-11, the esophagus expands to become the wider esophageal pouches (Figs 3, 4). Each pouch is marked with fine stripes but is hidden under the seminal vesicles and septa.
Immediately posterior to the esophagus and esophageal pouches are two pairs of smaller expansions of the gut, the calciferous glands (or esophageal glands). These bulge laterally from the esophagus into the coelom and are usually white with an abundance of fine parallel red blood vessels They are hidden by the third seminal vesicle and are immediately posterior to the fifth heart. They remove excess calcium and carbon dioxide from the blood and secrete white crystals of calcium carbonate into the gut. Calciferous glands play a role in the regulation of calcium concentration and blood pH.
Posterior to the last pair of calciferous glands, the esophagus narrows again and then joins the large, bulbous, thin-walled crop in about segment 12 (Figs 3, 4, 13-61B). The crop extends posteriorly through several segments and is a food storage organ.
Posterior to the crop the gut becomes the gizzard (Figs 3, 4, 13-61B). This large expansion of the gut tube has thick, heavy, muscular walls and is the major site of mechanical digestion. In it food is ground (triturated) into smaller fragments. Its muscles have a rich blood supply and its transverse, slightly oblique, folds are conspicuous. The buccal cavity, pharynx, esophagus, crop, and gizzard constitute the foregut.
The gut narrows again posterior to the gizzard and becomes the intestine (or midgut, or stomach-intestine), which is the region for chemical digestion (hydrolysis) and absorption. The intestine is heavily invested with yellow chlorogogen tissue.
The intestine extends from the end of the gizzard almost to the anus and its dorsal wall is invaginated to form a typhlosole to increase surface area (Fig 6, 13-61A). The crease of the typhlosole contains chlorogogen and connective tissue. The typhlosole is not visible in this view.
The anterior intestine, or saccular intestine, is specialized for synthesis and secretion of enzymes and hydrolysis of food molecules. This region has thin walls that tend to form blisters. The remainder of the intestine is absorptive and has thicker walls.
The extreme posterior end of the gut is the rectum, or hindgut. It lacks a typhlosole and opens to the exterior via the anus. It is not visible at present.
It is not feasible to open the gut until after you have completed your study of the other organ systems. Postpone study of the gut lumen and return to it after you have finished the reproductive system.
Hemal System
The earthworm hemal system contains bright red hemoglobin which makes it very easy to study in living specimens. The pigment is in solution rather than being cellular.
In preview, the basic plan of the system includes two major longitudinal vessels, one dorsal and the other ventral to the gut (Fig 6, 3, 13-2, 13-61A). The two vessels are connected in each segment by paired segmental vessels that supply and drain the tissues of the segment. The dorsal vessel and five pairs of segmental vessels in the region of the esophagus are contractile and function as hearts.
Blood moves anteriorly in the dorsal vessel and is pumped to the ventral vessel by the ten segmental hearts. The blood then flows posteriorly in the ventral blood vessel. It leaves the ventral vessel via the segmental vessels of each segment, goes to the organs and then returns to the dorsal blood vessel.
The nervous system is provided with its own system of longitudinal blood vessels supplied by vessels from the ventral blood vessel.
Find the large dorsal blood vessel on the midline of the dorsal surface of the gut (Fig 3, 13-61B). Peristaltic waves of contractions of circular muscles in the dorsal vessel move the blood anteriorly. Watch the vessel as it contracts and see if you can tell which way the blood is flowing. One-way valves in this vessel prevent backflow. Sometimes these valves can be seen through the transparent walls of the dorsal vessel in the vicinity of the crop and gizzard.
Using 10-15X magnification, look ventral to the gizzard at the ventral blood vessel. This vessel is attached to the ventral side of the gut by the narrow ventral mesentery (Fig 6, 13-2).
Follow the dorsal vessel anteriorly to the esophagus and find the five pairs of large, contractile, segmental blood vessels which are the hearts (Fig 3, 13-61B). They encircle the esophagus and pump blood ventrally, which is the reverse of the other segmental vessels. Trace one of them from the dorsal vessel to the ventral vessel. If the hearts are beating, try to verify the direction of blood flow. The hearts may be hidden by the septa and nephridia of their segments. Remove these as necessary to reveal the hearts. Be careful you do not tear or cut the hearts. Should you do so, the hemal system will bleed out and the hearts and vessels will be much more difficult to see.
Note the numerous branches of the dorsal and ventral blood vessels in the region of the crop and gizzard. The blood supply to the musculature of the gizzard is especially impressive. Note also the blood vessels to the body wall musculature, septa, nephridia, and seminal vesicles. These vessels are easy to see so long as they contain blood.
" Remove the hearts, dorsal blood vessel, nephridia, and septa from the area over the esophagus and reexamine that region of the gut, locating any organs you could not find earlier (Fig 4).
Respiratory System
Earthworms have no specialized respiratory structures and gas exchange takes place across the general epidermis which can be moistened when necessary with fluid from the coelomic pores. The integument is heavily vascularized to serve its respiratory function.
Nervous System
The earthworm nervous system is typically annelidan, consisting of a dorsal, anterior brain, circumpharyngeal connectives, ventral subpharyngeal ganglion and a ventral nerve cord with segmental ganglia, commissures, connectives, and segmental nerves (Fig 13-61-B, 13-62). The brain is farther posterior in oligochaetes than in polychaetes and is no longer in the prostomium.
The brain, consists of a pair of cerebral ganglia atop the anterior pharynx in segment 3 (Fig 3). If your previous incision exposed the buccal cavity, the brain can be seen without further dissection. Each ganglion is white and pyriform (pyri = pear, form = shape) and the two are connected across the midline by a short transverse cerebral commissure. The brain is the primary center for coordination of sensory and motor functions. It innervates the prostomium by a pair of prostomial nerves which can be seen on the dorso-lateral surface of the buccal cavity.
A large circumpharyngeal connective exits the side of each cerebral ganglion and runs ventrally around the pharynx to join the subpharyngeal ganglion ventral to the pharynx (Fig 13-62). Carefully cut away the tissue on one side of the pharynx to expose the connectives and the subpharyngeal ganglion. This ganglion serves the anterior three segments, including the peristomium, and is a major center for motor control.
The double, solid ventral nerve cord characteristic of annelids exits the subpharyngeal ganglion and extends posteriorly, on the ventral midline, for the length of the worm (Fig 5). The nerve cord swells in each segment to form a segmental ganglion from which arise three pairs of segmental nerves to nearby tissues (Fig 5). The segmental nerves are small and difficult to see.
There are no special sense organs in earthworms but the body surface, especially that of the head, bears receptor cells for taste, touch, light, and apparently vibration. The dorsum of the body is more sensitive to light than the venter. Unicellular photoreceptors are present in the epidermis and are most abundant on the prostomium. These photoreceptors are microscopic and cannot be seen in gross dissection.
Excretory System
Almost all segments of the earthworm possess a pair of complex metanephridia (Figs 3, 6, 13-65B). In fresh specimens these are easily seen on both sides of each segment. They are large, pale, and thin-walled. Each consists of a ciliated funnel, or nephrostome (nephr = kidney, stome = mouth), which opens from the coelomic cavity into a long, tortuously coiled tubule that penetrates the posterior septum of the segment and opens to the outside via a nephridiopore in the wall of the adjacent segment (Fig 13-61A, 13-65B). The wide distal region of the tubule functions as a bladder.
Reproductive System
Oligochaetes are simultaneous hermaphrodites and each individual contains complete and simultaneously functional male and female systems. The female system produces eggs and receives and stores sperm from the partner. The male system produces sperm and delivers it to a partner during copulation. The reproductive system is restricted to a few preclitellar segments (9-15). Most segments do not contain reproductive structures.
" Expose the reproductive system as follows. Free the anterior digestive system from pharynx to gizzard from its septal and circulatory connections with the body wall but leave the gut intact. Be careful that you do not harm the reproductive system in segments 9-15. Move the digestive tube aside and pin it out of the way, but do not cut or remove it. Some of the reproductive system is difficult to find but structures in bold type are easily identified.
Figure 5. Dorsal dissection of segments 9-16 of Lumbricus. The second right seminal receptacle has been removed. The sperm reservoirs are drawn as if transparent so the testes and sperm funnels are visible. Oligo58L.gif
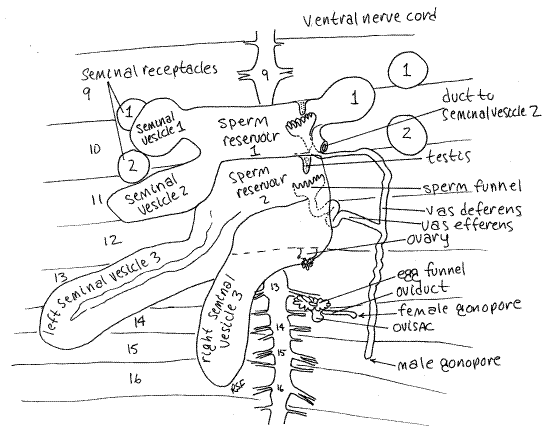
Male System
Look at the organs remaining in segments 9-15 (Figs 3, 4, 5, 13-61B, 13-67). Largest and most conspicuous are the three pairs of irregular, yellowish seminal vesicles belonging to the male system. Seminal vesicles store autosperm. (Autosperm is sperm produced by this worm.) The posterior pair is by far the largest and the anterior pair the smallest. These specialized coelomic spaces are the sites of maturation of sperm that originate in the tiny testes. The three pairs of vesicles are lateral outpockets of two large, median sperm reservoirs in segments 10 and 11 (Fig 5). The small, inconspicuous testes are in the reservoirs. The male gonoducts (sperm ducts, vasa deferentia) extend from sperm funnels in the reservoirs to the male gonopores on segment 15.
Female System
Two pairs of small, spherical seminal receptacles are located in segments 9 and 10. These resemble the seminal vesicles but are smaller and more regular in shape.
Whereas the seminal vesicles are parts of the male system, the seminal receptacles belong to the female system. The vesicles are the sites of maturation of autosperm. The receptacles, in contrast, are the sites of storage of allosperm. Allosperm is sperm produced by, and received from, the copulatory partner of this worm. Short ducts penetrate the body wall and connect the seminal receptacles with external pores between segments 9-10 and 10-11.
A pair of inconspicuous ovaries is located in the anterior septum of segment 13 (Fig 5, 13-61B, 13-67). The egg funnel is on the posterior septum of this segment and connects via the oviduct with the female gonopore on segment 14.
>1d. Remove one of the seminal receptacles and place it in a drop of 0.75% saline solution on a slide. Cut it in half and cover it with a coverslip. Examine the preparation with the compound microscope. You may see masses of very active allosperm. Sperm are not always present in the seminal receptacles. Bright white seminal receptacles usually contain sperm whereas pale cream-colored ones frequently do not. <
Study of the remaining features of the reproductive system is beyond the scope of this exercise but its structures may be identified, at your discretion, with the help of Figures 5, 13-61B, and 13-67 if desired. A pair of tiny ovaries in segment 13 release eggs into the coelom where they mature. They are stores in a small pouch, the ovisac, which protrudes from segment 13 into segment 14. A pair of female gonoducts, open from the coelom as the egg funnels, and transport mature ova from the ovisacs to the female gonopores on segment 14.
Return now to the postponed study of the gut lumen and then come back to the cross section slide below.
Gut Lumen
" Return the gut to its original position and open it from mouth to anterior intestine by inserting the point of your fine scissors in the mouth and cutting posteriorly along the dorsal midline. Pay attention to the structure of the gut wall as you cut. Where appropriate, observe the contents and then remove them with jets of water from a pipet. The wall of the buccal cavity is thin, as you already know. The walls of the pharynx are much thicker and have heavy longitudinal folds that allow for expansion. The walls of the anterior esophagus are thin and unremarkable but posteriorly the esophagus expands to form the esophageal pouches. Posterior to the pouches are the two pairs of calciferous glands filled with calcium carbonate particles.
Open the crop. What is its function? What do you find inside it? The walls of the crop are thin and folded longitudinally to permit expansion. The gizzard has very thick muscular walls. Note the thick cuticular lining of the gizzard. If you carefully adjust the light, you will see that the cuticle is iridescent like the epidermal cuticle. The cuticle can be grasped with a fine forceps and stripped away from the underlying muscle and epithelium. Remove the cuticle and look more closely at the soft tissue beneath it. The lining is grooved and ridged. You may see several small stones in the gizzard. Their function is to assist in grinding the food.
Extend the incision posteriorly into the intestine but cut along the lateral wall rather than the dorsal. The anterior intestine has very thin walls and the walls tend to form outward blisters. This is the secretory saccular region of the intestine and is the site of chemical digestion. The typhlosole is a large rounded fold of the dorsal gut wall that hangs down into the gut lumen. In this region its walls bear conspicuous transverse folds (Fig 6).
Open the body wall all the way back to the anus. You need not be particularly careful with this procedure. Moving posteriorly from the saccular region, open the intestine at various places and find a region where the typhlosole is smooth and unfolded. The walls of the intestine are thicker here. This region is chiefly absorptive. More posterior still is a region where there is no typhlosole at all. This is the rectum. Its walls are a little thinner than those of the posterior intestine but are thicker than the saccular region.
Remove the gut and set it aside. With the gut out of the way clear tissues away from the ventral nerve cord to expose it to view. Trace it anteriorly to the brain.
Cross Section
>1e Study a commercially prepared slide of an earthworm cross section using the compound microscope. The section is probably through the anterior intestine (Fig 6, 13-61A). Look at the section first with low power (40X) and orient yourself. Find the body wall, coelom, gut wall, and gut lumen (Fig 6).
Look at the body wall with high power (400X) beginning on the outside of the worm and work inward. The outermost layer is the thin, noncellular cuticle (Fig 13-59B). The cellularepidermis lies immediately below the cuticle and is a monolayered epithelium containing abundant secretory and sensory cells. The secretory cells, which secrete the cuticle, are easily recognized by their large open secretory vesicles. The very thin line at the base of the epidermis is its basal lamina. A thin, inconspicuous, connective tissue dermis is situated just inside the basal lamina and cannot be distinguished from it.
The next layer is the relatively thick circular muscle of the body wall. The muscle fibers of this layer run perpendicular to the long axis of the worm and are seen here in longitudinal section. Their nuclei are clearly visible.
The thickest layer of the body wall is the longitudinal muscle layer, which lies inside the circular muscle. Its fibers run parallel to the long axis of the worm and you see them here in cross section. The featherlike appearance of these fibers when in cross section is distinctive. The longitudinal muscle layer is divided into several bundles. A thin, squamous epithelium, thesomatic peritoneum, covers the inner surface of the longitudinal muscles. The large open space enclosed by the peritoneum is the coelom. Depending on the location of the section you may also see metanephridia, chaetal sacs, and segmental blood vessels.
Look at the gut tube in the center of the coelom using 100X. The dorsal wall of the anterior intestine is invaginated to form a large typhlosole which extends deep into the gut lumen. As a result, the gut lumen is U-shaped, with the opening of the "U" directed dorsally. The thick layer of cells bordering the lumen is the mucosal epithelium. Look closely at its free surface forcilia.
Figure 6. Cross section through the intestinal region of the earthworm, Lumbricus. The section is slightly oblique. Oligo59La.gif
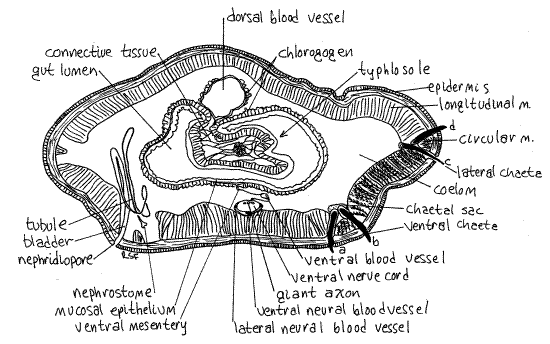
Moving from the mucosal epithelium toward the coelom there are, in order, connective tissue, circular muscle, longitudinal muscle, and peritoneum but in the intestinal wall these layers are poorly developed and probably cannot be distinguished from each other.
The epithelium of the foregut and hindgut secrete an extracellular cuticle that lines the lumen and is best developed in the gizzard. The development and function of the epithelium and of the muscle layers varies depending on the region of the gut.
Bordering the coelom, the outermost layer of the gut tube is the splanchnic peritoneum, which on the intestine is specialized as chlorogogen tissue. The chlorogogen forms a thick layer of large cells extending into the coelom from the peritoneum. Note that the space in the "U" of the typhlosole is nearly filled with chlorogogen and connective tissue.
Dorsal to the gut tube is the large dorsal blood vessel. Ventral to the gut is the smaller ventral blood vessel and large ventral nerve cord. The ventral vessel and the nerve cord are suspended from the gut by the ventral mesentery. At the dorsal surface of the nerve cord are three conspicuous giant axons.
A tiny longitudinal lateral neural vessel lies on each side of the nerve cord and a larger, also longitudinal, subneural vessel lies ventral to it. The lateral neural vessels are supplied by branches from the ventral blood vessel. They in turn supply the nerve cord with blood. The subneural vessel drains the nerve cord. <
References
Brooks WK . 1890. Handbook of Invertebrate Zoology. Bradlee Whidden, Boston. 352p.
Brown A . (ed) 1950. Selected Invertebrate Types. Wiley, New York. 597p.
Edwards CA, Lofty JR . 1977. The Biology of Earthworms. 2 nd ed. Chapman and Hall, London. 283p.
Freeman WH, Bracegirdle B . 1971. An Atlas of Invertebrate Structure. Heinemann, London. 129p.
Jamieson BGM ., 1992. Oligochaeta, Pp. 217-322 in Harrison FW, Gardiner SL (eds), Microscopic Anatomy of Invertebrates 7, Annelida. Wiley-Liss, New York.
418 pp. Laverack MS. 1963. The Physiology of Earthworms. Pergamon,
Oxford . 206p.
Parker TJ, Haswell WA . 1951. A Textbook of Zoology, vol 1, 6 th ed. MacMillan, London.
Pearse V, Pearse J, Buchsbaum M, Buchsbaum R . 1987 . Living Invertebrates. Blackwell (Boxwood), Palo Alto. 848p.
Rowett HGQ . 1957. Dissection Guides V. Invertebrates. Reinhart, New York. 56p.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Supplies
Dissecting microscope
Compound microscope
Living nightcrawlers, Lumbricus terrestris
Worm-size dissecting pan (kippered herring tin with wax bottom)
#1 stainless steel insect pins
5-7% non-denatured ethyl alcohol in tapwater
Dissecting set with microdissecting tools
0.75% saline solution
0.05% aqueous methylene blue
Cross section slides of Lumbricus through intestinal region
|
Item |
Source |
|
Live Lumbricus |
Local bait shop |
|
Preserved Lumbricus |
Ward’s, Carolina, others |
|
XS slides |
Triarch*, Wards, Carolina |
|
Median Sagittal section, anterior end |
Triarch |
|
Parasagittal section, anterior end |
Triarch, Ward’s |
|
Near median sagittal section, anterior |
Ward’s |
|
Nephridium wm |
Carolina, Ward’s, Triarch |
|
|
|
*Triarch has a large variety of prepared Lumbricus slides