Invertebrate Anatomy OnLine
Lolliguncula brevis ©
Brief Squid
4jul2006
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises can be accessed by clicking on the links to the left. A glossary and chapters on supplies and laboratory techniques are also available. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
MolluscaP, Eumollusca, Conchifera, Ganglioneura, Rhacopoda, Cephalopoda C, Coleoidea sC, Decabranchia SO, Teuthoidea O, Myopsida sO, Loliginidae F (Fig 12-125, 12-88)
Mollusca P
Mollusca, the second largest metazoan taxon, consists of Aplacophora, Polyplacophora, Monoplacophora, Gastropoda, Cephalopoda, Bivalvia, and Scaphopoda. The typical mollusc has a calcareous shell, muscular foot, head with mouth and sense organs, and a visceral mass containing most of the gut, the heart, gonads, and kidney. Dorsally the body wall is the mantle and a fold of this body wall forms and encloses that all important molluscan chamber, the mantle cavity. The mantle cavity is filled with water or air and in it are located the gill(s), anus, nephridiopore(s) and gonopore(s). The coelom is reduced to small spaces including the pericardial cavity containing the heart and the gonocoel containing the gonad.
The well-developed hemal system consists of the heart and vessels leading to a spacious hemocoel in which most of the viscera are located. The kidneys are large metanephridia. The central nervous system is cephalized and tetraneurous. There is a tendency to concentrate ganglia in the circumenteric nerve ring from which arise four major longitudinal nerve cords.
Molluscs may be either gonochoric or hermaphroditic. Spiral cleavage produces a veliger larva in many taxa unless it is suppressed in favor of direct development or another larva. Molluscs arose in the sea and most remain there but molluscs have also colonized freshwater and terrestrial habitats.
Eumollusca
Eumollusca, the sister taxon of Aplacophora, includes all molluscs other than aplacophorans. The eumolluscan gut has digestive ceca which are lacking in aplacophorans, the gut is coiled, and a complex radular musculature is present.
Conchifera
Conchifera, the sister taxon of Polyplacophora, includes all Recent molluscs other than aplacophorans and chitons. The conchiferan shell consists of an outer proteinaceous periostracum underlain by calcareous layers and is a single piece (although in some it may appear to be divided into two valves). The mantle margins are divided into three folds.
Ganglioneura
Most Recent molluscs are ganglioneurans, only the small taxa Aplacophora, Polyplacophora, and Monoplacophora are excluded. Neuron cell bodies are localized in ganglia.
Rhacopoda
The mantle cavity is posterior in the ancestor although it may be secondarily moved to an anterior position by torsion. This taxon includes gastropods and cephalopods.
Cephalopoda C
Cephalopods are derived, bilaterally symmetrical molluscs specialized for rapid, well-coordinated motion. They tend to be large, active raptors with well-developed nervous systems, sensory equipment, and complex behavior patterns. Convergent evolution has produced many features analogous to those of vertebrates. The taxon includes about 700 extant species of squids, cuttlefishes, nautilus, and octopods.
The body is elongated dorso-ventrally rather than along the antero-posterior axis. The foot is modified to form eight or more, suckered, prehensile appendages used for food capture. In almost all living cephalopods the shell is either reduced and internal or absent. Gills are present in a mantle cavity ventilated by muscular contraction rather than cilia. The hemal system includes systemic and accessory hearts. Blood is confined to vessels, which are lined by a cellular endothelium, and there is no hemocoel. The nervous system is highly cephalized and consists of a ring of coalesced ganglia around the anterior gut.
Coleoidea sC
Coleoids are cephalopods with ten or fewer arms, two gills, and two nephridia. The shell is internal or vestigial. Included are squids, cuttlefishes, octopods, and vampyromorphs.
Decabranchia So
Decabranchs have eight arms and two long specialized tentacles. These are the squids and cuttlefishes, which is most cephalopods.
Teuthoidea O
These are the squids. The body is long and tubular, the shell is an internal, dorsal, longitudinal skeletal rod.
Myopsida sO
The eye is covered by a transparent corneal membrane. Most are coastal.
Laboratory Specimens
This exercise is based on the common southeastern squid, Lolliguncula brevis (Fig 12-70A), but can also be used with Loligo and, with some modification, Illex and Sepia. Living, frozen, or preserved specimens can be used. If the study is performed on a living specimen, most internal structures can be seen with no dissection other than opening the mantle cavity. Conduct the study in a dissecting pan of seawater or magnesium chloride (if living), or tapwater (if frozen or preserved) using magnification as needed.
Figure 1. Ventral view of a female Lolliguncula from Beaufort, North Carolina. Ceph47La.gif
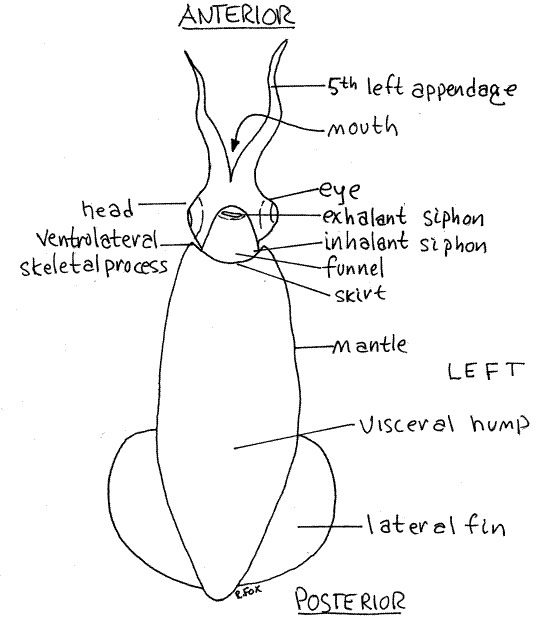
Orientation
The squid body is elongate, streamlined, and shaped rather like a torpedo (Fig 1). It is derived from the squat, limpet-like shape of the ancestral molluscs but has undergone a major shift of the primary body axis (Fig 12-62). In most bilaterally symmetrical animals, including the ancestral mollusc, the chief body axis is antero-posterior but in cephalopods it is dorsal-ventral. The cephalopods have modified the ancestral plan by lengthening this axis so that the head and appendages are at the ventral, rather than the anterior, end of an elongate animal. This shift can be a source of confusion for students who expect animals to have their heads at the anterior end.
The easiest way to avoid this confusion is to rename the axes of squids according to their function rather than their morphology. Following this convention, the end with the head and appendages will be referred to as anterior (Fig 1), even though it is really ventral. The opposite, pointed end will be called posterior (it is really dorsal).
The long straight edge with the funnel will be ventral (really posterior) and the opposite edge, which corresponds with the anterior end of the ancestral mollusc, will be called dorsal. Adoption of this convention eliminates confusion because the animal appears to conform to other bilateral animals and forward is anterior, rear is posterior, up is dorsal and down is ventral. Right and left are not affected. All remaining references to direction in this exercise follow this convention.
External Anatomy
Place a squid in a dissecting pan of suitable size and immerse it in the appropriate fluid (isotonic magnesium chloride if living or tapwater if preserved).
Visceral Hump
The body is composed of the anterior head and a long, conical, posterior visceral hump. The head bears 10 long appendages. The tubular funnel is ventral to the head (Fig 1, 12-79). The column of tissue connecting the head and the visceral hump is the neck.
The visceral hump is the long narrow cone that forms the remainder of the body. Its thick muscular walls are the mantle, which encloses the large mantle cavity and the visceral mass. The open water space within the mantle is the mantle cavity. The organs in the core of the visceral hump make up the visceral mass.
The pointed apex of the hump is the posterior end of the animal and it bears a pair of lateral fins. The fins are thin sheets of muscle and connective tissue displaced a little to the dorsal side of the cone.
You now have all the landmarks you need to orient your specimen and identify its three principal axes. Use the landmarks to determine anterior-posterior, dorsal-ventral, and right-left according to the convention. Find the plane of bilateral symmetry.
The free anterior margin of the mantle is the skirt , or collar (Fig 1). It is shallowly scalloped and bears three short, acute, anterior skeletal points. One point is on the middorsal process on the dorsal midline and is the anterior tip of the rudimentary internal shell. The other two points are the two ventrolateral skeletal processes and are the external manifestations of endoskeletal cartilages. The head and funnel are attached to the skirt at these three points.
Ventrally the funnel, which is derived from the foot of the ancestral molluscs, emerges from the mantle cavity (Fig 1, 12-64). It lies below the head on the ventral midline and is theexhalant siphon through which water exits the mantle cavity. Contractions of circular muscles in the mantle force jets of water from mantle cavity out the funnel. The animal relies on this for propulsion and respiration. Water enters the mantle cavity via two lateral inhalant siphons between the mantle and funnel (Fig 1).
The mantle cavity contains the two gills and the outlets of the digestive, excretory, and reproductive systems (Fig 2). The respiratory current enters, passes over the gills and outlets, and then exits. The siphons have valves to insure that water flows in the correct direction.
>1a. Arrange your specimen with its ventral side up in a dish and cover it with liquid. Use your thumb and first two fingers to grasp the anterior visceral hump in the region of the mantle cavity. In doing so your thumb and second finger should press gently on the right and left sides of the mantle cavity while your forefinger presses on the ventral surface, between your thumb and second finger. While watching the funnel, press gently on the sides of the mantle cavity with thumb and second finger. This will expand the mantle cavity and draw water into it through the lateral inhalent siphons. Release the sides and press gently on the ventral surface with your forefinger. This will decrease the volume of the cavity and force water out of the funnel. You will see the jet of water emerge from the funnel. Use a pipet to release a cloud of dye/seawater mixture near one of the inhalent siphons while pumping. A jet of colored water will soon emerge from the funnel. <
Head and Foot
Arrange the squid in the pan so the dorsal surface is uppermost and look at the anterior end. The region known as the head (Fig 1, 12-62B) is the combined head and foot of the ancestral mollusc and the name Cephalopoda (ceph = head, pod = foot) alludes to this combination. The foot of cephalopods is divided into numerous suckered, flexible, appendages, either arms or tentacles. Squids and cuttlefishes have a total of 10 such appendages and are accordingly referred to as decapods (Decabranchia). Eight of their appendages are relatively shortarms, and two are much longer tentacles (Fig 6, 12-64). Octopods have a total of eight appendages all of which are arms.
Squids have five appendages on each side numbered from dorsal to ventral. Starting dorsally on the right, find and assign numbers to the five right appendages. Appendage 1 is the dorsalmost appendage and is the shortest (Fig 6, 12-64). Appendages 1, 2, 3, and 5 are arms. Each arm bears two rows of suckers.
The fourth appendage is a tentacle and it is much longer than any arm. It is contractile so its length is variable. It is expanded distally to form a club with four rows of suckers (Fig 12-64, 12-74A). Except for the club, the tentacles do not bear suckers. The clubs are used to capture food which is transferred to the arms for manipulation.
The left fifth appendage of males is slightly modified to form a hectocotylus arm, which is used to transfer spermatophores to the female (Fig 2, 12-85B). Its suckers have reduced cups and enlarged pedicles. The pedicles are long, fingerlike, and arranged in a comblike row at the end of the arm. Determine the sex of your specimen by examination of the left fifth arm.
With magnification look at a large sucker near the base of an arm. The oldest and largest suckers are proximal on the arm and new suckers are added to the growing distal tip. The suckers are usually not symmetrical and each consists of a deep, muscular cup with a toothed, chitinous ring for reinforcement (Fig 12-76A). These rings are easily dislodged in dead fresh or frozen specimens and several may be lying about on the bottom of the dissecting pan. They may be absent in preserved material.
The cup is attached to the arm via a slender, muscular, stalklike pedicle. The distal end of the pedicle is expanded to form a disk-shaped piston that can be seen in the bottom of the cup. When in contact with a firm surface, the rim of the cup seals against the surface. Contraction of the pedical withdraws the piston and generates a suction inside the cup. The cups continue to function even in dead (fresh) specimens and sometimes even in preserved specimens.
The arms and tentacles are borne on a ring of muscle that surrounds the mouth. Look at the anterior end of the head, en face, using magnification if necessary. Spread the 10 appendages apart so you can see the mouth in the center of the circle of arms. It is a large opening equipped with a pair of dorsal and ventral brown or transparent, proteinacous jaws, forming a beak resembling that of a parrot (Fig 6, 12-63, 12-64).
Figure 2. The tip of the left fifth (hectocotylus) arm of a male Lolliguncula. Ceph48L.gif

The mouth is surrounded by two concentric circular membranes (Fig 6). The outermost is the buccal membrane. It covers the bases of the appendages and has seven small, suckered points on its margin. The second and innermost ring is the peristomial membrane near the mouth. (In female Loligo there is a horseshoe-shaped depression on the inner surface of the buccal membrane where spermatophores are attached during courtship. The equivalent area of Lolliguncula is inside the mantle cavity.)
The eyes of cephalopods are remarkably similar to those of vertebrates. Each consists of an outer, transparent cornea which is the lateral wall of the head and is separated from the remainder of the eye by a water chamber (Fig 7, 12-82). The iris lies internal to the cornea and the pupil is a circular opening in its center. A small silver fold of the iris extends into the otherwise circular pupil. This fold extends farther into the pupil under conditions of bright light and retracts in dim light. It is analogous to the vertebrate iris diaphragm.
Posterodorsal to the eye is a low narrow ridge, the olfactory crest which bears chemosensory epithelium. Water entering the mantle cavity passes over the crests.
The visceral mass is a thick solid, central core extending from the head through the roof of the mantle cavity to the posterior end of the body (Fig 5). Posteriorly it expands to occupy most of the visceral hump.
Body Wall
The body is covered by thin, easily shed epidermis underlain by a thicker connective tissue dermis. Complex, multicellular chromatophores are located in the outer layer of the dermis (12-83A,B,C). (If you are looking at living or fresh material the body is probably speckled with expanded chromatophores.) Each chromatophore contains a single pigment in a sac and a wide range of colors has been reported from various squid species including brown, black, violet, blue, yellow, orange, and red. Chromatophores are capable of rapidly changing the color of the animal.
>1b. Observe the color of a living squid in an aquarium. Watch while it swims undisturbed and then interfere with it in some way and see if the chromatophores respond (12-83B,C). Observe the squid undisturbed against pale and dark backgrounds. Watch the squid interact with other species, including prey, and with conspecifics. <
>1c. Observe the chromatophores of a living specimen with the dissecting microscope. Watch the chromatophores enlarge and contract. Can they become so small you cannot see them? What colors of chromatophores are present? <
>1d. Use a dissecting microscope to examine the chromatophores of a fresh, or recently frozen, squid. Note their size and the range of sizes. What colors of chromatophores are present? <
>1e. The dermis contains iridiocytes in its deeper layers. These flat cells contain guanine rods that diffract light. They are abundant near the eye where they produce a shimmering iridescence around the ink sac. If you have a living or fresh specimen, examine the iridiocytes with the dissecting microscope and observe their characteristic iridescence. <
" With scissors make a longitudinal, ventral incision a little to the right or left of the ventral midline of the visceral hump. Begin at the skirt and cut posteriorly all the way to the posterior end of the hump to open the mantle cavity as in Figure 3. Use strong pins inserted into the wax to hold the cavity open.
Examine the cut surface of the ventral mantle wall. Most of its thickness is muscle composed mostly of circular and radial fibers. There are connective tissue sheaths on both sides of the muscle layer and the dermis lies outside the outer sheath.
Squids possess an endoskeleton composed of the dorsal, internal, chitinous shell, or pen, and other elements constructed of chondroid, a clear, firm, cartilage-like material (Fig 12-64). The pen is located on the dorsal midline of the visceral hump and is embedded in the mantle. It is shaped like a feather with the broad vane located posteriorly. The vane protects the dorsal surface of the visceral mass. The shaft of the feather points anteriorly and its tip forms the middorsal process of the mantle skirt. The endoskeleton consists of 11 cartilages, including cranial, fin, funnel, and orbital cartilages, as well as several supports for the skirt.
>1f. Later, when completely finished with your dissection, make a shallow, longitudinal, middorsal incision and remove the pen. Note its featherlike shape. You may press the pen in a thick book, like a flower, and keep it indefinitely. <
Mantle Cavity
The mantle cavity and its contents are now visible as a result of the incision just completed. In addition, much of the internal anatomy can also be seen through the thin body wall lining the mantle. (In living specimens the transparency of the body wall makes it even easier to see the internal anatomy.)
The large mantle cavity extends posteriorly almost to the end of the visceral hump. Note that the funnel opens into the mantle cavity anteriorly and that its posterior margin is thin and flexible. The mantle skirt is joined via three interlocking junctions to the funnel and head (Fig 3).
Figure 3. A female Lolliguncula with the mantle cavity open and viewed ventrally. Ceph49La.gif
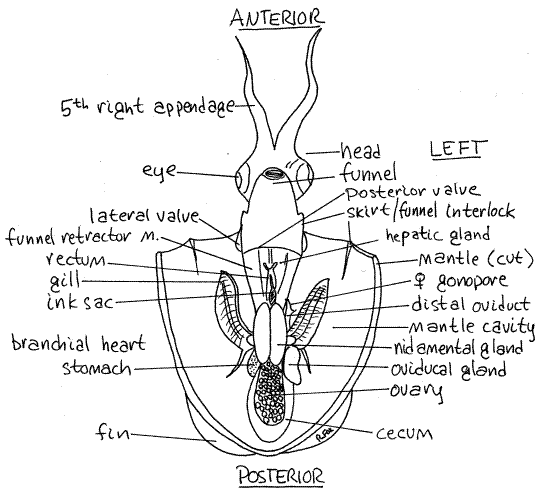
The one-way valves guarding the inhalant siphons are just dorsal to the lateral interlocks. They are flexible, thin, posteriorly directed flaps of tissue that permit entry of water during inhalation but close to prevent its exit.
" Open the funnel with a midventral, longitudinal incision along its entire length and look inside it. The thin, flexible sheet of tissue just inside the anterior opening is a one-way valve to prevent the intake of water through the funnel during inhalation (Fig 12-64).
The thin, posterior, ventral margin of the funnel is another one-way valve. During exhalation, water pressure pushes this flexible tissue against the ventral skirt thus closing the valve and preventing the exit of water from any place other than the funnel.
The valves assure that water enters only through the two inhalant siphons and exits only via the funnel. This provides the necessary power for swimming and insures that clean, incoming water will flow over the gills before it reaches the gonopores, kidney pores, ink sac, and anus. Review the valves and their role in controlling the flow of water into and out of the mantle cavity.
The funnel can be aimed by the squid to control the direction of motion. Aiming is controlled by a pair of large, longitudinalfunnel retractor muscles that look like white cords extending posteriorly from the funnel beside the visceral mass (Fig 3, 12-79). These muscles insert on the sides of the funnel and originate on the broad posterior end of the pen.
Another pair of cordlike muscles, thicker than the funnel retractors and dorsal to them, contributes to the roof of the anterior visceral mass. These are the cephalic retractor muscles that originate on the pen and the skirt cartilages and insert on the posterior surface of the cranium. These and other muscles connect the head with the visceral hump and adjust the position of the head.
Study the structures visible in the visceral hump (Fig 3, 12-79). The two large gills are located laterally, beside the visceral mass in about the middle of the mantle cavity. Each is long and feathery and composed of numerous non-ciliated gill filaments projecting from a longitudinal central axis. The axis is attached by a thin sheet of tissue to the roof of the mantle cavity.
At the base of each gill is a small oval branchial heart (Figs 3, 4, 5, 12-80B). These are accessory hearts that supply the gills with unoxygenated blood. The systemic heart, which will be identified shortly, supplies the rest of the body. If this seems unusual, consider that mammals also have a systemic heart (the left atrium and ventricle) to supply the body and an accessory heart (the right atrium and ventricle) to serve the respiratory organs.
Female Reproductive System
If your specimen is a female, much of the anterior mantle cavity will be occupied by two large, white, oval nidamental glands which are not present in males (Fig. 3). These glands secrete part of the egg envelope.
The ovary is a translucent, lobulated mass lying in a coelomic space (the gonocoel) in the posterior end of the visceral mass. Its size is variable. The oviduct exits the left side of the ovary and extends anteriorly. It swells enormously to become the large, white oviducal gland on the left, just posterior to the left branchial heart (Figs 3, 4).
The oviduct extends from the oviducal gland to open into the mantle cavity via the female gonopore located anterior to the left branchial heart. There may be a cluster of bristlelike, white spermatophores attached to the dorsal mantle wall on the left side of the mantle cavity. If present, they will be medial to the left gill and anterior to the opening of the oviduct. The spermatophores are placed here during courtship. (In Loligo the spermatophores are placed on the horseshoe organ of the buccal membrane.)
Remove the nidamental glands (if your specimen is female) before continuing your examination of the mantle cavity. Be careful that you do not damage any of the organs to which they are attached.
Male Reproductive System
If your specimen is a male there will be no nidamental glands but you will see a single white spermatophoral gland on the left of the visceral mass just posterior to the left branchial heart (Fig 5). Spermatophores, resembling small white sticks may be visible within the gland (Fig 12-85A).
Figure 4. Ventral view of a female Lolliguncula with the mantle cavity opened. The nidamental glands have been removed. Ceph50La.gif
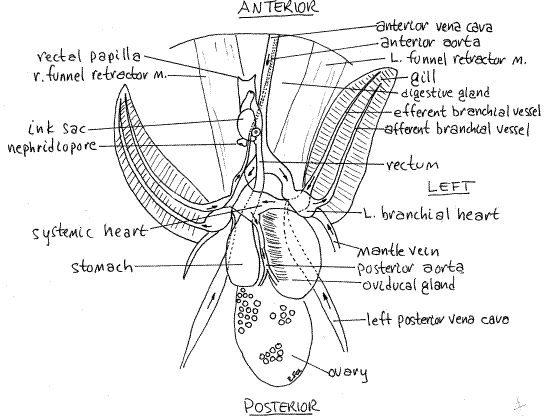
The testis is a bright, white, flat, ovoid mass lying in a coelomic space on the roof of the posterior visceral mass. (In living specimens it should be faintly visible through the numerous translucent tissues in the area but it will not be apparent in preserved material.) Its gonoduct lies on the left and leads anteriorly from the coelom to the spermatophoral gland where spermatozoa are packaged into spermatophores. The sperm duct leaves the gland and runs to a long, wide, thin-walled spermatophoral sac where spermatophores are stored. The penisexits the sac and lies on the left side of the rectum just anterior to the left branchial heart. The male gonopore is at the tip of the penis.
>1g. Make a wetmount of a spermatophore in seawater and examine it with the compound microscope. (If your specimen is living, you may see active spermatozoa exiting the open end.) It may be necessary to tear the spermatophore apart to see the sperm. <
Posterior Digestive System
Most of the posterior gut is visible without dissection but the anterior end, from mouth through esophagus, is embedded in the head and visceral mass and cannot be seen at this time (Fig 12-77).
Figure 5. Ventral view of a male Lolliguncula with its mantle cavity opened. Ceph51La.gif
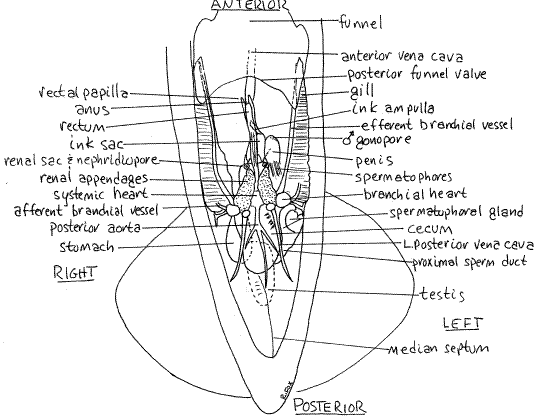
The stomach is a muscular sac on the right immediately posterior to the right branchial heart (Figs 3, 4, 5). It is relatively thin-walled and may be difficult to recognize. It may contain granular material and is dorsal to and posterior to the heart.
The large gastric cecum opens from its medial side and extends posteriorly, on the left (Figs 3, 5). When expanded the gastric cecum is thinner walled and much larger than the stomach. Its walls are contractile, however, and its size varies inversely with the thickness of its walls. The inner surface of the gastric cecum wall bears small finlike folds. The stomach, intestine, esophagus, digestive gland, and gastric cecum join at a complex junction with a five-way valve.
The intestine exits the valve and runs anteriorly in the visceral mass from which it emerges as the rectum. The rectum is a small tube attached to the dorsal midline of the mantle cavity (Figs 3, 4, 5). The anus is at its anterior end.
The digestive cecum of cephalopods is divided into two parts (Fig 12-77). The large digestive gland (= hepatic gland, or liver), lies dorsally on the midline in the visceral mass between the funnel retractor muscles (Fig 4). It is not obvious unless dissected. It is a large, long, off-white organ extending from the posterior end of the head to the anterior end of the stomach. It is wide anteriorly, pointed posteriorly.
The pancreatic gland, or pancreas, is a separate, much smaller, U-shaped region of the digestive cecum located anterior to the stomach where it is surrounded by the kidneys. Ducts from the digestive gland run to the pancreas and from there to the gastric cecum. Together the two portions of the digestive cecum secrete an acidic mixture of peptidases, amylases, and lipases into the gut.
>1h. Visualization of the gut can be improved by injecting dye into the rectum via the anus, if the gut walls are intact. <
Ink Sac
Lying beside the rectum is the ink sac (Figs 3, 4, 5, 12-77, 12-79). Iridiocytes in its walls give it a shiny, metallic appearance. Its transparent duct can be seen extending anteriorly from it to empty into the rectum just posterior to the anus. The walls of the duct do not contain iridiocytes and ink contained within is clearly visible.
Hemal System
The single systemic heart lies on the midline of the ventral surface of the visceral mass between the branchial hearts (Figs 4, 5, 12-80). It is enclosed in a thin pericardium and consists of a central muscular ventricle into which the two efferent branchial vessels drain.
The systemic heart pumps blood to the body via the anterior and posterior aortae (Fig 4). Unoxygenated blood returning from the body goes to the branchial hearts via an anterior vena cava and two posterior vena cavae. Each branchial heart pumps blood to a gill via an afferent branchial vessel on the lateral edge of the gill. The blood is oxygenated in the gill and leaves via the efferent branchial vessel on the medial margin of the gill to go to the systemic heart (Fig 4, 12-80B).
>1i. Visualization of the major blood vessels can be enhanced by injection with toluidine blue or other dye. Use a hypodermic syringe with a fine needle to inject the systemic heart or an aorta and one of the vena cavae. <
Nephridia
The paired renal sacs (nephridia, or kidneys) are located beside the midline just anterior and ventral to the systemic heart (Fig 5). They are modified metanephridia and are associated with the pericardial cavities of the branchial hearts and the anterior vena cavae. The renal sacs cover most of the organs and structures in the area between the systemic heart and the posterior end of the ink sac but may be difficult to discern. Each opens to the mantle cavity via a nephridiopore located on the sides of the base of the rectum, immediately posterior to the ink sac. Each pore is in the center of a small donut-shaped papilla (Figs 4, 5). In females the donut is associated with the white and orange accessory nidamental gland but in males it is transparent.
Internal Anatomy
" Further dissection is required to expose anterior portions of the digestive system and the nervous system. With a sharp scalpel bisect the animal with a clean vertical section coinciding with the median sagittal plane. Cut all the way through the animal from dorsal to ventral along its entire length so that it is divided into right and left halves (Fig 6). Study both halves as needed.
Figure 6. The left side of the squid, Lolliguncula, in median sagittal section. Musculature has been deflected to reveal the stellate ganglion. Ceph52La.gif
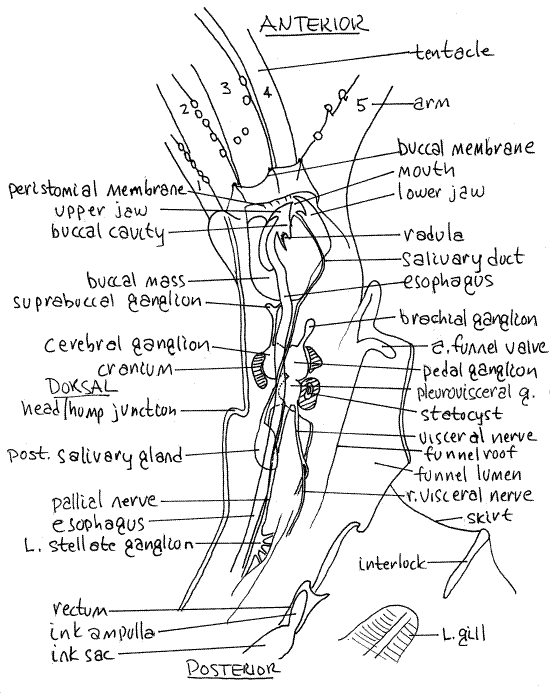
Anterior Digestive System
" The anterior digestive system is revealed by the sagittal section.
The two mandibles, or jaws, are embedded in the large, spherical buccal mass (Fig 6). They are dorsal and ventral to the mouth and are operated by the muscles of the buccal mass.
" Remove the dorsal jaw. The mouth opens into the buccal cavity in the interior of the buccal mass (Fig 6, 12-77). The two anterior salivary glands are located beside the buccal cavity and release a peptidase into it. They are not visible in sagittal section. The radular sac is a small, median pocket in the floor of the buccal cavity containing the small radula. The fine teeth of the radula are barely visible with the dissecting microscope.
>1j. Remove the radula, make a wet mount, and examine it with the compound microscope. Find its teeth Fig 12-78). <
The esophagus exits the posterior wall of the buccal cavity (Fig 6). It is a narrow, white tube that runs posteriorly through the nerve ring and cranium, then dorsal beside the rectum before curving sharply ventrally to enter the stomach.
The digestive gland occupies the ventral face of the visceral mass and extends the length of the mantle cavity, contributing to its roof (Fig 4). In life it is milky white.
The single, oval posterior salivary gland lies near the esophagus posterior to the nerve ring, embedded in the hepatic gland (Fig 6). A slender, white salivary duct runs anteriorly from the gland, paralleling the esophagus through the nerve ring, then along the ventral wall of the buccal mass to enter the buccal mass and open into the buccal cavity.
Nervous System
The nervous system is well-developed, bilaterally symmetrical, and strongly cephalized (Fig 6). It consists of 31 ganglia, their connectives, commissures, and nerves. It is supplied with information by sophisticated, well-developed sense organs and it coordinates a large and complex musculature. Cephalopods have a well-developed, centralized circumesophageal nerve ring, or brain, around the esophagus (Fig 6, 12-81).
The nerve ring is partially enclosed in a chondroid cranium (Fig 6) analogous to that of vertebrates. In sagittal section the cranium appears in three parts, one dorsal and two ventral. The dorsal piece forms the roof of the cranium and the anterior ventral piece forms the floor. The posterior ventral part is the otic capsule and contains a statocyst. Optic cups of chondroid are fused to the sides of the cranium and form orbits to support the eyes but they are not visible in sagittal section.
The nerve ring is composed of paired cerebral, pleural, pedal, and visceral ganglia in a ring around the esophagus. The ganglia are closely associated with each other (Fig 6, 12-81) with shortened commissures and connectives.
Three nerves arise from the fused pleurovisceral ganglion atop the otic capsule and extend posteriorly. Two of them, the pallial nerves, run to the stellate ganglia. The unpaired visceral nerve, extends posteriorly to innervate several posterior structures including the funnel retractor muscles, cephalic retractor muscles, rectum, ink sac, ctenidia, and parts of the reproductive system (Fig 6).
Two large stellate ganglia (stella = star), right and left, lie on the sides of the roof of the anterior mantle cavity (Fig 6). Each is located along the edge of the visceral mass. You can find them by pushing the funnel retractor muscles aside and looking along their dorsal edges. Each ganglion resembles a star and has several clearly visible giant axons radiating from it. These are motor axons to the muscles of the mantle.
Eye
Cephalopods are well known for their elaborate camera eyes which resemble those of vertebrates (Fig 7, 12-82). The eyeball is surrounded by the transparent cornea on the lateral surface and the sclera elsewhere.
The iris is visible inside the cornea on the lateral surface. It is a tough, thick layer with a silvery, opaque argentea layer containing iridiocytes. A layer of muscle in the iris causes the argentea to appear to undulate in living specimens. The pupil is an opening in the iris.
A small aquiferous pore located anteroventrad the eye opens into the lateral chamber, which is filled with seawater, between the cornea and the iris.
" Cut through the iris, beginning at the edge of the pupil, to reveal the interior, medial chamber, of the eye (Fig 7). The spherical lens is medial to the pupil. The lens is supported by ciliary ligaments and muscles that form the ciliary body. The inner wall of the interior of the eye is the sensory pigmented retina. It is a layer of alternating sensory and pigment cells analogous to the vertebrate retina and choroid.
Figure 7. Sketch of a cephalopod eye in section. Ceph53L.gif
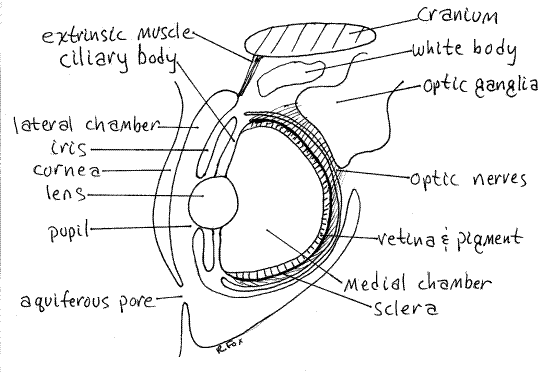
The tough, chondroid sclera forms a cup enclosing the eye.
The wall of the eye contains a thick layer of reflective iridiocytes called the argentea because of its silvery appearance (argente = silver). These prevent entry of light from any direction except through the pupil. There is a conspicuous (in life) white body of unknown function between the brain and the eye.
Nine extrinsic eye muscles extend from the eyeball to the cranium but are not thought to produce movement of the eye which is firmly fixed to the optic cup. Their function is not understood but they may compensate for changes in the shape of the eye produced by movements of the arms and head.
Behavior
>1k. If living squid are available, make observations of feeding behavior. In the laboratory, Lolliguncula will feed avidly on the grass shrimp, Palaemonetes, which is usually available in coastal areas in the range of the squid. Introduce living shrimp to the tank and carefully watch the response of the squid. Pay particular attention to chromatophore displays and the activity of the appendages. Look for differences in the roles of the arms and tentacles in food capture and feeding. <
References
Abbott RT. 1974. American seashells, 2 nd ed. Van Nostrand, Reinhold, New York. 663 pp.
Budelmann BU, Schipp R, von Boletzky S. 1997. Cephalopoda. Pp 119-414 in Harrison FA, Kohn AJ (eds), Microscopic anatomy of Invertebrates, vol A, Mollusca II. Wiley Liss, New York.
Bullough WS . 1958. Practical Invertebrate Anatomy (2 nd ed). MacMillan, London. 483p.
Hanlon RT, Messenger JB. 1996. Cephalopod behaviour. Cambridge Univ. Press, Cambridge. 232 pp.
Ho J-S . 1978. Laboratory Manual for Invertebrate Zoology Emphasizing Marine Forms. Hwong, Los Alamitos, Calif. 152p.
Pierce ME. 1950. Loligo pealeii, pp. 347-357 in Brown FA. (ed) 1950. Selected Invertebrate Types. Wiley, New York. 597p.
Purchon RD. 1977. The Biology of the Mollusca 2 nd ed. Pergamon, Oxford. 560p.
Schipp R, von Boletzky S. 1975. Morphology and function of the excretory organs in dibranchiate cephalopods. Fortschr Zool. 23:89-110.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Tompsett DH. 1939. Sepia. Liverpool Mar. Biol. Comm. Mem. 32:1-184.
Wilbur KM, Yonge CM (eds). 1964. Physiology of Mollusca vol. 1. Academic Press, New York. 473p.
Supplies
Dissecting microscope
Compound microscope
Dissecting pan, about 20X30 cm
Living or preserved squid
Dissecting set
Heavy dissecting pins