Invertebrate Anatomy OnLine
Haemopis marmorata
and Hirudo medicinalis ©
Leeches
4jul2006
Copyright 2003 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises, a glossary, and chapters on supplies and laboratory techniques are also available at this site. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Annelida P, Clitellata, Hirudinomorpha C, Hirudinea, Euhirudinea, Arhynchobdellida O, Gnathobdelliformes sO, Hirudinidae F (Fig 13-7A)
Annelida P
Annelida consists of the segmented worms in the major taxa Polychaeta (bristleworms), Oligochaeta (earthworms and relatives), Branchiobdellida (crayfish ectosymbionts), and Hirudinea (leeches) with a total of about 12,000 known species in marine, freshwater, and terrestrial environments. The segmented body is composed of an anterior prostomium, a linear series of similar segments, and a posterior pygidium. The prostomium and pygidium are derived from anterior and posterior ends of the larva whereas the intervening segments arise through mitotic activity of mesodermal cells in the pygidium.
The body wall consists of a collagenous cuticle secreted by the monolayered epidermis. A connective tissue dermis lies beneath the epidermis. The coelom is lined by a mesothelium which may be specialized to form the body wall muscles. Most annelids have chitinous bristles, or chaetae, secreted by epidermal cells, that project from the body. The coelom is large, segmentally compartmented, lined by mesothelium, and well developed in polychaetes and oligochaetes but reduced in leeches. Successive coelomic spaces are separated by transverse bulkheads known as septa which consist of double layers of mesothelium with connective tissue in between. The right and left sides of each segmental coelom are separated by longitudinal mesenteries which, like septa, are double layers of mesothelium with connective tissue between.
The gut is a straight, regionally specialized tube that begins at the mouth at the anterior end and extends for the length of the body to end at the anus on the pygidium. It penetrates each septum and is supported by dorsal and ventral mesenteries. Like that of most invertebrates, the gut consists of ectodermal foregut, endodermal midgut, and ectodermal hindgut. The nervous system consists of a dorsal brain in or near the prostomium, a pair of circumpharyngeal connectives around the anterior gut, and a double, ventral nerve cord with paired segmental ganglia and nerves. The hemal system of most annelids is a set of tubular vessels, some of which are contractile and serve as hearts. The hemal system is absent or greatly reduced in leeches. The system includes a dorsal longitudinal vessel above the gut in which blood moves anteriorly, a ventral longitudinal vessel below the gut, in which blood moves posteriorly, and paired segmental vessels that connect the dorsal and ventral vessels. The digestive, hemal, and nervous systems are continuous and pass through the segments.
Respiration is accomplished in a variety of ways. In some, the general body surface is sufficient but gills are present in most polychaetes, many leeches, and a few oligochaetes. Excretory organs are metanephridia or protonephridia and typically one pair is present in each segment. These osmoregulatory organs are best developed in freshwater and terrestrial species. The sexes are separate in polychaetes but oligochaetes and leeches are hermaphroditic. In the ancestral condition paired submesothelial clusters of germ cells were present in each segment and released developing gametes into the coelom. In derived taxa reproductive functions tend to be confined to a few specialized genital segments. Gametes mature in the coelom or its derivatives and fertilization is external. Gametes are shed through ducts derived from metanephridia or by rupture of the body wall. Spiral cleavage follows fertilization. Clonal reproduction is common.
Clitellata
Clitellata, the sister taxon of Polychaeta, includes earthworms, leeches, and related taxa. The head is reduced and its sensory functions minimized. Clitellates lack the parapodia characteristic of polychaetes although chaetae may be present. Also lacking are head and pygidial appendages. Present however, is a girdle-like band of secretory epidermis, the clitellum, near the anterior end of the worm. The brain has moved out of the prostomium into a more posterior segment. These worms are hermaphroditic and the gonads and reproductive equipment are restricted to a few specialized genital segments. Development is direct, without a larva.
Hirudinomorpha
Hirudinomorpha includes leeches and leechlike worms. The body has a terminal posterior sucker and a dorsal subterminal anus. An anterior sucker is present in all but one species (Acanthobdella peledina). Chaetae are lacking (except for Acanthobdella). Hirudionomorpha is the sister taxon of Oligochaeta and includes branchiobdellids and true leeches.
Hirudinea
Hirudinea is the sister taxon of branchiobdellida and includes the true leeches (Euhirudinea) plus Acanthobdella peledina.
Euhirudinea
True leeches are specialized annelids derived from oligochaete ancestors. Leeches may be aquatic, amphibious, or terrestrial but most are aquatic in freshwater habitats. All are carnivores, either parasitic or predatory. The parasites consume blood, usually that of vertebrates.
Leeches have anterior and posterior suckers, no chaetae, and a body composed of the prostomium and 33 segments. The underlying segmental organization of these annelid worms is obscured by superficial rings around the body. Parapodia, chaetae, and head appendages are absent.
The hemal system is reduced or absent and its role in fluid transport has been taken over by coelomic channels. The coelom lacks septa or mesenteries and is reduced to four longitudinal channels. Most of the coelomic space is filled with botryoidal tissue derived from mesothelium.
The excretory system is metanephridial but derived. The nervous system is typically annelidan. Like oligochaetes, leeches are hermaphroditic and have a clitellum. Cross-fertilization occurs and the eggs are enclosed in a hard proteinaceous cocoon. Cleavage is spiral and development is direct.
Arhynchobdellida O
Arhynchobdellids have no hemal system and coelomic channels are responsible for fluid transport. Freshwater, amphibious, and terrestrial.
Natural History
The use of leeches such as Hirudo for medical purposes has regained favor in recent years. Leeches are now used routinely and with considerable success to maintain arterial blood flow to traumatized areas with impaired venous drainage. They are used by plastic surgeons in the repair of skin flaps or severed digits. The leech provides the drainage necessary to sustain and encourage arterial flow to the traumatized region. The leech anticoagulants hirudin and hementin are administered to cardiac patients to inhibit clot formation and to destroy existing clots respectively. At least two companies, Leeches USA and Biopharm UK, have been established to provide the medical profession with medicinal leeches and pharmaceuticals derived from leeches.
Haemopis is an opportunistic predator and scavenger feeding on a wide variety of small animals, both living and dead. Among its prey are earthworms, other leeches, insects, small crustaceans, snails, slugs, and amphibian tadpoles and eggs. Haemopis is amphibious and at night crawls many meters from the water to feed on small terrestrial animals and carrion. One species is semiterrestrial.
Hirudo, Macrobdella, and Philobdella consume vertebrate blood a type of feeding known as sanguivory. Macrobdella decora is the American medicinal leech and in North America it is the leech that most often attacks mammals, including humans. Hirudo medicinalis has not successfully colonized North America.
Laboratory Specimens
This exercise is written for Haemopis marmorata , the American horse leech, and Hirudo medicinalis, the European medicinal leech, and can be used equally well for either species. Both species belong to Arhynchobdellida O and are similar. Hirudo is a sanguiferous species that sucks blood whereas Haemopis is predaceous and will eat almost any small animal it can capture. Haemopis is often used for fishing bait and can, in season, be purchased at bait shops. Hirudo and Haemopis are sometimes placed in separate families (Hirudinidae, Haemopidae).
This exercise may be conducted using preserved or living specimens of either Haemopis or Hirudo. Live, anesthetized, dissections are preferable to preserved.
Study and dissection of leeches must be conducted under magnification. Place a large preserved leech in a dissecting pan of tapwater. If living specimens are used, the leech should initially be placed in 5% ethanol. After about 15 minutes the 5% solution should be replaced with 15% ethanol. The animal should be kept in the 15% solution for the remainder of the study.
External Anatomy
The leech body is elongate and dorsoventrally flattened (Fig 1, 13-70). The shape is highly variable from species to species and sometimes it may be nearly cylindrical or short and fat (Fig 13-82). The body is covered by a thin cuticle and the integument is pigmented, often with bright colors but sometimes drab gray or black.
Suckers
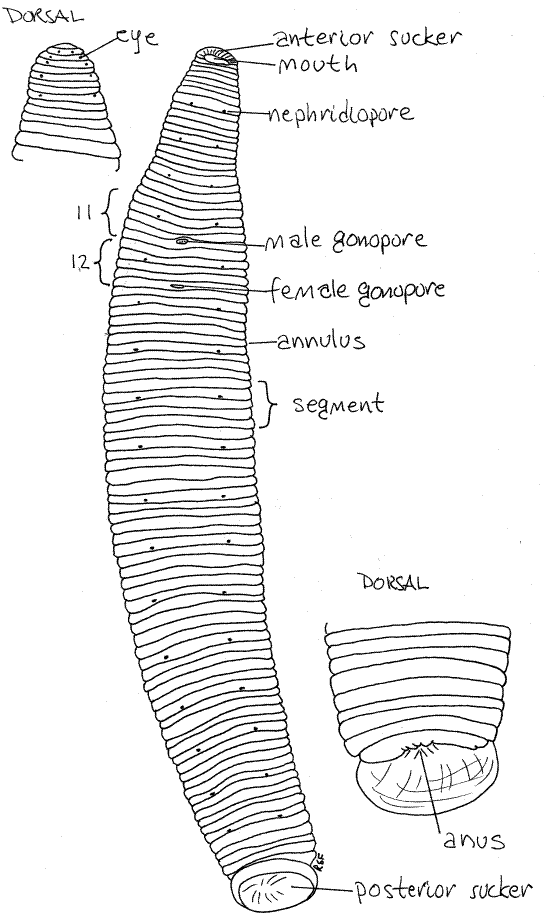
A large posterior sucker is present at that end of the body and a smaller anterior sucker at the opposite end (Fig 1, 13-70). Both are equipped with gland cells that secrete adhesive. The large preoral chamber occupies the interior of the anterior sucker. The posterior wall of this chamber is a transverse sheet of tissue known as the velum. The mouth is a small, triradiate opening in the center of the velum (Fig 13-76B).
Segmentation
The body of all leeches consists of the prostomium plus 33 segments of which 26 are body segments and seven form the posterior sucker (Fig 1). The anteriormost part of the body is the prostomium but it is not a segment. The prostomium and first five segments are the head and the anterior sucker is derived from their ventral parts. The head segments can be seen dorsally but ventrally they are obscured by the sucker.
Most segments are superficially subdivided by circumferential rings into false segments, or annuli, which make it difficult to recognize the true segments. Most segments have five annuli but those at the anterior and posterior ends have fewer. It is not necessary that you recognize the boundaries of each segment.
Figure 1. Ventral view of the leech, Haemopis marmorata. The insets are dorsal views of the anterior and posterior ends. leech23L.gif
Sense Organs
Haemopis and Hirudo have five pairs of small black eyes at the anterior end on the dorsal surface of the head (Fig 1, 13-76B). They form an arc around the curve of the end of the worm but may be difficult to see against the darkly pigmented integument. If you cannot see them, try placing the worm, venter down, on the stage of the dissecting microscope and lay a glass slide over the head to compress it. The eyes should now be visible. Each eye is a deep pigment cup filled with photoreceptor cells and each points in a different direction.
Several tiny sensory papillae (sensilla) are located on the middle annulus of each segment (Fig 13-70). Those of Hirudo are easy to see, especially on the ventral surface, but those ofHaemopis marmorata usually cannot be seen. There are seven papillae on each sensory annulus. Three are dorsal, three ventral, and two lateral.
The papillae have receptor cells sensitive to chemicals, light, and water movement. The eyes, which were seen earlier, are papillae specialized for photoreception. Mechanoreceptive neuron endings are scattered over the body surface. Sanguivorous leeches are sensitive to water movement and respond positively to disturbances in hopes it was caused by prey.
Gonopores
Leeches are hermaphroditic with the male and female systems opening independently of each other. The two relatively large gonopores are on the ventral midline in the anterior quarter of the body (Fig 1, 13-70). The male gonopore (on segment 11) is anterior to that of the female. During copulation the penis is everted through this opening and inserted into the female gonopore on segment 12. (Preserved Hirudo often have the penis everted.) The female gonopore is a little smaller and less obvious than the male pore. (Members of Macrobdella have a cluster of copulatory pores on the ventral surface a few annuli posterior to the female gonopore.)
Anus
The anus is on the dorsal midline immediately anterior to the edge of the posterior sucker (Fig 1).
Clitellum
The leech clitellum is present only during periods of reproductive activity and even then it is inconspicuous. When present, it is located on segments 10-13 and these segments together are the clitellar region. The epithelium of this region is slightly swollen and paler during the reproductive season but at other times it is indistinguishable from the rest of the body. Gland cells here secrete a hardened, proteinaceous cocoon into which fertilized eggs are deposited.
Nephridiopores
Tiny, paired, segmental nephridiopores are present laterally on the ventral surface of most of the body except the extreme anterior and posterior ends (Fig 1, 13-70). The pores are inconspicuous but are marked by a tiny pale spot on the posterior edge of the second annulus of the segment and are spaced five annuli apart for most of the length of the body. (The nephridiopores are sometimes hard to find in preserved Hirudo.)
Internal Anatomy
" Anchor the worm temporarily with a #1 insect pin through the posterior sucker and open the body with a middorsal longitudinal incision. Start the incision at the posterior end of the worm by pinching the dorsal body wall with a pair of fine forceps and then cut through the fold of body wall with fine scissors. Insert the point of the scissors in the resulting opening and cut from sucker to sucker along the dorsal midline. The body wall is thick and muscular and you must cut entirely through it without damaging the organs in the interior.
Most of the interior of the worm is packed with dark, reddish-brown botryoidal tissue derived from the coelomic mesothelium and containing coelomic remnants. It is homologous to the chlorogogen of oligochaetes.
Opening the leech and cleaning away the botryoidal tissue to reveal the organs is a time-consuming task but one that must be performed carefully. Your efforts will be rewarded by a specimen in which the gut, excretory, nervous, and reproductive systems are clearly visible and easily studied. Once the packing is removed little additional dissection is necessary.
Coelomic Channels
Watch for branches of the dorsal longitudinal coelomic channel lying in the botryoidal tissue (Fig 2). In life they are red and resemble blood vessels. These channels will be destroyed as you remove the botryoidal tissue. The conspicuous, dorsal coelomic channel on the dorsal midline of the gut is easily seen if it is filled (in life) with red coelomic fluid (Fig 2). If you avoid cutting everything ventral to it, you will not damage the gut or other organ systems.
Body Wall
The thick and muscular body wall begins with the epidermis (Fig 13-72A). Gland cells in the epidermis secrete a thin cuticle and abundant mucus. The cuticle is layered, composed of protein and mucopolysaccharide and is replaced every few days as it wears away.
Inside the epidermis is a thin connective tissue dermis where the chromatophores responsible for body pigmentation are located. Internal to the dermis are the body wall muscles including the relatively thin outer layer of circular muscle, a thin layer of oblique muscles, and the thick, inner layer of longitudinal muscles. The fibers of the longitudinal muscles are easily seen in the cut edges of the body wall. The cuticle, epidermis, dermis, circular and oblique fibers cannot be distinguished from each other in gross dissection. Abundant slender, threadlikeradial muscles extending from the body wall to the viscera. Dorsoventral muscles span the thickness of the worm between dorsal and ventral surfaces and are responsible for maintaining the flat shape.
Coelom
The coelom is reduced by invasion of mesenchymal botryoidal tissue and connective tissue (Fig 13-72A). These tissues fill almost all of the ancestral coelomic space. The remnants of the coelom form a fluid transport system that resembles, and functions like, a hemal system. In modern leeches the coelom is represented by the four longitudinal, vessel-like, coelomic channels, tiny coelomic "capillaries" in the botryoidal tissue, and sacs enclosing the gonads. The nephridia and gonoducts are coelomoducts.
There are four longitudinal coelomic channels in Haemopis and Hirudo (Fig 2). These are the dorsal, ventral, right lateral and left lateral coelomic channels. The dorsal channel, mentioned above, lies on the midline atop the gut and the ventral channel is a tube around the ventral nerve cord. It is ventral to the gut.
Figure 2. Dorsal dissection of the anterior end of the leech, Haemopis. Botryoidal tissue has been removed and much of the reproductive system has been omitted. Nephridia of the right side are omitted. Leech30L.gif
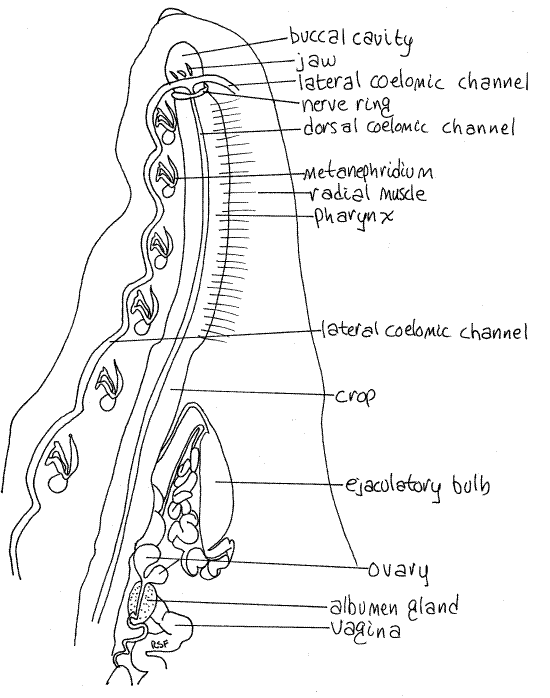
The two lateral channels are muscular and contractile and function as "hearts" to propel coelomic fluid through the system. The four channels are connected with each other anteriorly and posteriorly and all have numerous segmental branches. The channels have a cellular lining derived from mesothelium and contain hemoglobin.
The two lateral channels are sinuous tubes embedded in the sides of the ventral body wall where they pass immediately lateral to the nephridia (Fig 13-72A). They are usually visible, without further dissection, for at least part of their length and are easy to see in living specimens, if they still contain red coelomic fluid.
Digestive System
Sanguivorous leeches, such as Hirudo and Macrobdella, have large jaws with strong muscles and sharp teeth for cutting through mammalian skin. The predator, Haemopis, consumes its prey whole, and has weak jaws with a few blunt teeth or sometimes lacks jaws. Sanguivorous leeches feed irregularly and have anatomical and biochemical adaptations for storing food for hundreds of days. Predaceous leeches feed frequently, typically every few days, and digest food rapidly.
The gut is a long, white or gray, regionally specialized tube lying on the midline and extending from mouth to anus (Fig 3). (The gut of Hirudo may appear red if it contains a recent blood meal but its walls are white or gray.) The annelid gut comprises an ectodermal foregut, endodermal midgut, and ectodermal hindgut.
Expose the gut to view by grasping the sides of the incision with forceps and pulling them gently apart. Pin the walls open along the length of the worm. This will rupture some of the branches of the coelomic channels.
The gut is the dorsalmost large structure in the interior but it, like everything else, is embedded in botryoidal tissue and it adheres tightly to the body wall. The borders of the gut may not be obvious when you first open the body.
" Beginning at the posterior end locate the gut and free it for its entire length from the surrounding botryoidal tissue. Do this carefully with fine forceps and scissors. Use the scissors to cut longitudinally between the sides of the gut and the adjacent body wall. Grasp the two cut edges of the body wall with forceps and pull them apart and away from the gut. Stop if this tears the gut. Try to avoid cutting through the walls of the gut or of its diverticula. (If you puncture the gut of Hirudo you may see the remains of its last meal. If so, note that the blood is still fluid and has not coagulated.) Do not cut anything ventral to the gut yet but note, and then cut as necessary, the many muscles crossing the interior. The dorsoventral muscles run from the ventral body wall to the dorsal body wall and radial muscles extend from the body wall to the viscera. Be careful of the small, white, wormlike metanephridia lying ventrally beside the gut (Fig 2).
The mouth is an opening through the velum into the buccal cavity, the first chamber of the gut tube (Fig 2, 3, 13-76C). The walls of the buccal cavity are thin, delicate, and transparent. Depending on the species there may be jaws embedded in the walls of the buccal cavity (Fig 4).
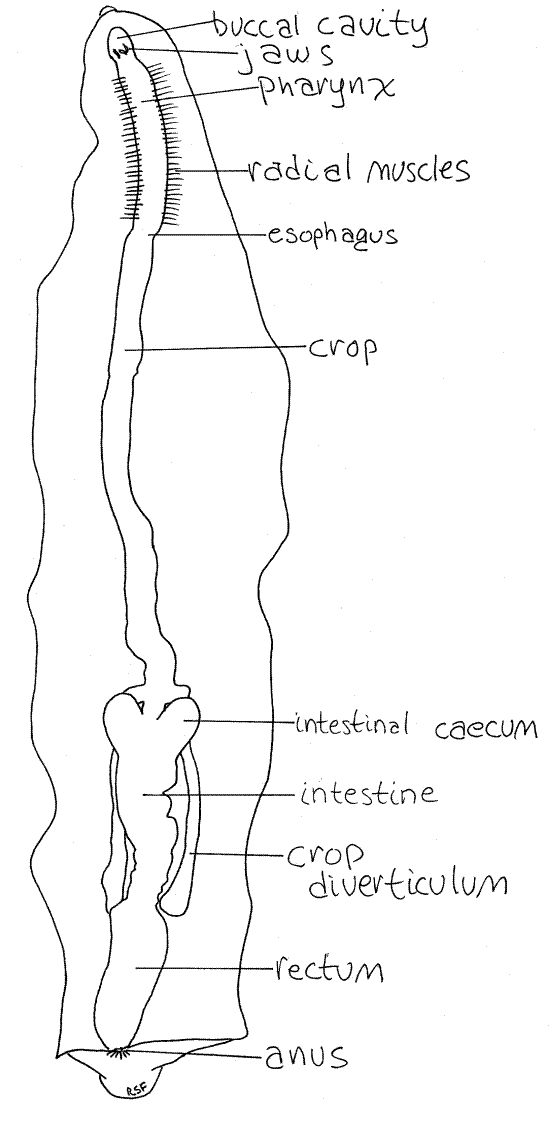
Figure 3. Dorsal dissection of Haemopis showing the gut. All other organ systems have been removed. Leech24L.gif
Posteriorly the buccal cavity opens into the muscular pharynx. In Haemopis the pharynx is long and straight (Fig 3) but in Hirudo it is short and oval (Fig 5, 13-77A). Note the manyradial muscles extending from the pharynx to the body wall. Contraction of these muscles dilates the pharynx.
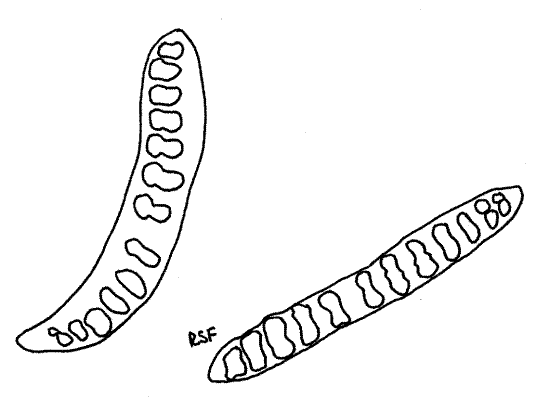
Figure 4. Jaws and teeth of Haemopis marmorata. Leech26L.gif
In Hirudo the white salivary gland is mingled with the radial muscles making it difficult to see the pharynx (Fig 5). Secretions of the salivary cells of Hirudo contain hirudin (an anticoagulant), an anesthetic, a spreading factor (hyaluronidase), a vasodilator (similar to histamine), antibiotics, and mucus. The salivary gland is not a discrete organ and its cells are epidermal gland cells whose individual ductules open independently on the jaws.
In Haemopis note, but do not damage, the conspicuous circumenteric nerve ring encircling the anterior end of the pharynx (Fig 2). (The circumenteric nerve ring of Hirudo is hidden by the salivary gland. Carefully cut away the salivary gland and radial muscles on one side to reveal it.)
Without damaging the nerve ring, open the buccal cavity with a middorsal incision and find the three hard jaws (Fig 4), of Haemopis marmorata or Hirudo. (Haemopis grandis has no jaws or teeth).
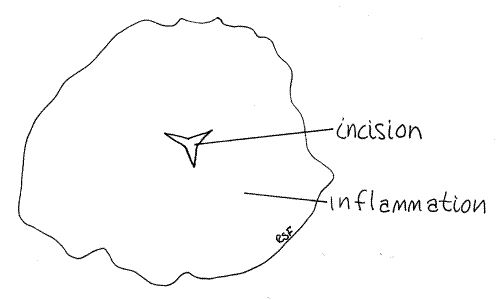
>1a. Remove one or more of the jaws and make a wholemount for observation with the compound microscope. Note the teeth on the cutting edges. There are 10-20 teeth in Haemopismarmorata and up to 100 in Hirudo. In Hirudo the jaws are large and the dorsal one is the most obvious from your viewpoint. It looks like a white cone pointed anteriorly. It is in the posterior buccal cavity. Its anterior edge bears a sharp ridge of up to 100 very fine sharp teeth. The bite of Hirudo is a characteristic triradiate incision made by the three scalpel-like jaws (Fig 6). <
The posterior end of the pharynx constricts somewhat to become the esophagus (Fig 3). The short esophagus immediately widens to become the long, thin-walled, transparent crop. The buccal cavity, pharynx, and esophagus make up the foregut and they are lined internally with cuticle.
The midgut comprises the crop and intestine. The crop extends through the middle region of the body and then ends where it joins the intestine (Fig 3). The crop is a storage organ and is very large in sanguivorous species which depend on widely spaced meals.
Figure 5. Dorsal dissection of the leech, Hirudo medicinalis, emphasizing the digestive system. leech25L.gif
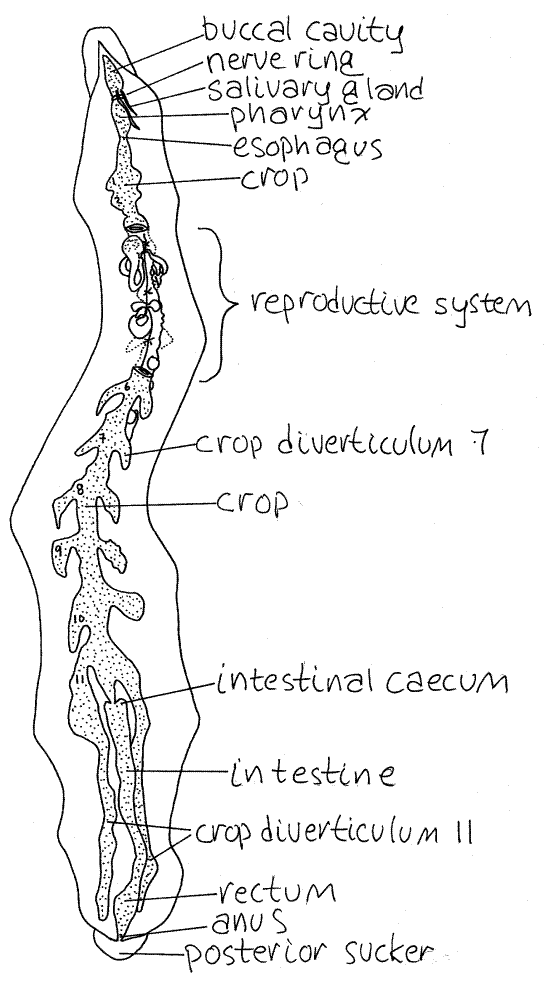
The crop bears paired lateral crop diverticula to increase its storage capacity. There are 11 pairs of diverticula in the sanguivore, Hirudo (Fig 5) but only one in the carnivore,Haemopis (Fig 3). The diverticula of Haemopis are at the extreme posterior end of the crop whereas those of Hirudo are arranged segmentally along its entire length. In both species the posterior diverticula are long, unbranched, blind tubes that adhere closely to the ventral body wall.
Figure 6. Hirudo bite on human skin. Leech27L.gif
An obligate symbiotic bacterium, Aeromonas hydrophila, inhabits the gut of Hirudo and produces an antibiotic that prevents the growth of other bacteria that would otherwise prevent the long term storage of blood and cause it to decompose. Aeromonas also assists in digestion of the blood and may provide vitamins for its host.
The intestine follows the crop and bears two intestinal ceca at its anterior end (Figs 3, 5). In Haemopis the ceca are large and bulge from the anterior end of the intestine but those ofHirudo are tiny and inconspicuous.
The walls of the intestine and its ceca are thick and opaque in contrast to those of the crop and its internal surface is thrown into deep transverse folds. The intestine secretes exopeptidases and is the region of hydrolysis and absorption. Leeches have no lipases, carbohydrases, or endopeptidases. In sanguivorous species the protein (globin) of the hemoglobin molecule is the chief food.
The intestine opens into the rectum, or hindgut, whose walls are thin, diaphanous, and translucent. The rectum opens to the exterior via the dorsal anus.
Reproductive System
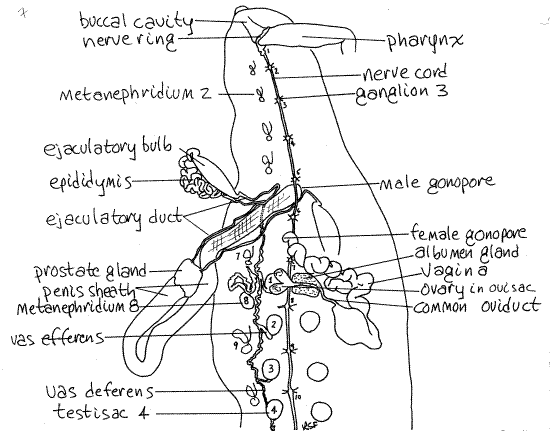
Complete male and female reproductive systems are present in each individual. The copulatory organs, both male and female, of Haemopis are large and dominate the vicinity of segments 10-18 (Figs 2, 7). Those of Hirudo are much smaller and cannot be seen until the gut is moved aside (Fig 8, 13-78B) and are largely restricted to segments 11 and 12 (Fig 5).
" With fine scissors and forceps cut the connective tissue and muscles holding the gut in position, freeing enough of it so you can move it aside and reveal the reproductive organs. Be careful that you do not damage these organs.
Figure 7. Dorsal dissection of the anterior end of Haemopis emphasizing the reproductive system. The male system has been moved to the left and the female to the right. Nephridia, with the exception of # 8, are stylized. Leech28La.gif
Female
All parts of the female system are localized in the vicinity of the middle of the crop. The ovaries are paired but the rest of the system is single.
The female system begins with the two, tiny, filamentous ovaries (Fig 7), each of which is enclosed in a small, spherical, translucent ovisac. The ovaries can be seen through the walls of the ovisacs, which are reduced coelomic spaces drained by short oviducts. Fertilization occurs in the sacs and oogenesis begins here.
A short oviduct exits each ovary. The two oviducts quickly join each other to become the common oviduct which immediately enters the anterior end of the translucent albumen gland. The common oviduct passes through the gland, receiving its nutritive secretions, and exits its posterior end.
Upon leaving the albumen gland the oviduct becomes convoluted and extends posteriorly to join the swollen proximal end of the vagina. The thick, highly convoluted distal end of the vagina extends anteriorly to the female gonopore on segment 12. (The vagina of Hirudo is much smaller than that of Haemopis and lacks the long convoluted distal region (Fig 8). It is comma-shaped.)
Figure 8. The reproductive system of Hirudo, in dorsal view. Leech29L.gif
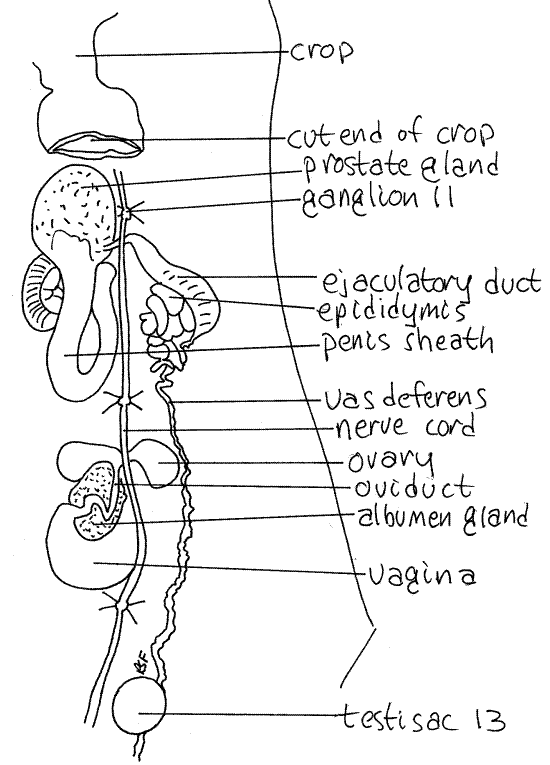
Male
The male reproductive system is scattered along most of the length of the body and most of its structures are paired (Fig 7). The reproductive system of Hirudo is shown in Figures 8 and 13-78B.
There are 9-11, usually 10, pairs of testes contained within small, spherical, coelomic testisacs and it is the testisacs, not the testes that you see in gross dissection. Testisacs are brown in Haemopis, white in Hirudo. The testisacs are arranged in right and left lateral rows beginning just posterior to the female gonopore and extending posteriorly almost to the end of the worm (Fig 7). They are hidden by the gut, other reproductive organs, and botryoidal tissue. Move these obstructions aside and find one of the testisacs, then remove the botryoidal tissue from its vicinity so you can see it clearly. Spermatogenesis occurs in the testisacs.
A small, convoluted, white vas efferens exits each testisac. The vas efferens quickly empties into the vas deferens. The vas deferens looks like the vas efferens but is long and longitudinal. There are two vasa deferentia, one receiving the vasa efferentia from the testisacs of the left side, the other for the right.
Each vas deferens runs anteriorly, hidden by a layer of connective tissue, then widens to become the epididymis (Fig 7). This is a bright white, thick, highly convoluted tube in which spermatozoa are stored. The vas deferens of Hirudo runs directly to the epididymis without reversing direction (Fig 8).
Each ejaculatory bulb is a large, firm, tough, spindle-shaped organ (Fig 7). The epididymis adheres to the border of the bulb and enters its posterior end. A long, slender, wavy, but not convoluted, ejaculatory duct exits the anterior end of the ejaculatory bulb and extends to the prostate gland.
The white prostate gland receives the ejaculatory ducts from the two ejaculatory bulbs (Fig 7). There is only one prostate gland and the remaining parts of the male system are unpaired.
The prostate is located at the proximal end of the large, U-shaped penis sheath, which connects with the male gonopore on segment 11 (Fig 8). The extensible penis is housed within the sheath and is extended through the male gonopore during copulation.
Copulation and Fertilization
During copulation the penis is everted from the male gonopore and inserted into the female gonopore of the partner. Sperm are transferred to the vagina of the partner. Insemination may be reciprocal but usually is not. Spermatozoa move up the oviduct to the ovisac. Fertilization occurs in the ovisac while the female cells are still oocytes. Fertilized oocytes pass down the oviduct and are coated with secretions of the albumen gland. Oogenesis is completed and development initiated occur in the cocoon. The developing embryos of Haemopis and Hirudo are nourished by albumen.
Epidermal gland cells of the clitellum secrete a ringlike, proteinaceous cocoon around the body. The cocoon slides anteriorly across the female gonopore from which it receives 5-10 fertilized eggs. The cocoon and its eggs continues to move forward and drops off the head. Deposited cocoons are ovoid. Development of the embryo occurs in the cocoon. The juveniles hatch from eggs as miniature replicas of the adults and there is no larval stage.
Nervous System
The leech nervous system is typical of annelids and may be seen in its entirety by simply moving the gut and median reproductive structures aside (Figs 2, 7, 8).
The brain consists of a pair of slightly swollen cerebral ganglia atop the anterior end of the pharynx (Fig 2). They are connected with each other by a short transverse commissure across the midline.
From the lateral borders of the brain arise the circumpharyngeal connectives, one from each side, which curve around the pharynx to join ventral to the gut at the largesubpharyngeal ganglion. The cerebral ganglia, commissure, circumenteric connectives, and subpharyngeal ganglion make up the circumpharyngeal nerve ring (Fig 2).
A pair of longitudinal nerve cords arises from the subpharyngeal ganglion and extends posteriorly on the ventral midline for the entire length of the animal (Fig 7). The two cords lie close together and together form the ventral nerve cord. Large, easily seen segmental ganglia are located along the nerve cord (Fig 7). The cord and ganglia are contained within the tubularventral coelomic channel, whose walls are dark but transparent. The nerve cord ends at the large terminal ganglion ventral to the posterior end of the rectum. Two pairs of segmental nerves arise from each segmental ganglion.
Osmoregulatory/Excretory System
Osmoregulation and excretion of metabolic wastes is accomplished by 17 pairs of large, elaborate metanephridia (Fig 2, 13-75), the first of which is in segment 7. The nephridia of freshwater leeches such as Haemopis and Hirudo are designed to eliminate large amounts of water while retaining salts. Their excretory role is secondary to osmoregulation.
The nephridia are small, twisted, white ducts arranged segmentally along the sides of the body in the ventral body wall beside the testisacs. Each includes a nephrostome, tubule, bladder, and nephridiopore. The nephrostomes project into the lateral coelomic channels (Fig 13-75) but are not connected to the tubule.
Hemal System
The hemal system is reduced in leeches and absent in the arhynchobdellidan Haemopis and Hirudo where it is functionally replaced by the coelomic transport system.
Respiratory System
Although some leeches have gills, the unspecialized general body surface is the gas exchange organ of Haemopis and Hirudo. Leeches ventilate the body surface by slow anterior to posterior undulations passing along the body. Gasses are distributed by hemoglobin in the coelomic fluid. Hirudo and at least some species of Haemopis enhance gas exchange by lying partially exposed to the atmosphere at the edge of the water. Leeches are facultative anaerobes.
Behavior
If living unanesthetized specimens are available take advantage of the opportunity to observe leech behavior. Leeches are active and accomplished swimmers that also creep in a characteristic motion described as "leechlike" (Fig 13-74). When creeping, the posterior sucker is attached to a firm substratum and the body extended anteriorly. The anterior sucker is then attached and the posterior sucker released. The body is shortened, the posterior end curled up under the body and moved anteriorly. The posterior sucker moves along the ventral surface. The posterior sucker is then attached to the substratum immediately beside the anterior. When both suckers are attached, the leech's body makes a hairpin loop high above the substratum. The anterior sucker is then released and the anterior end moved forward again.
>1b . Place a living leech in a large, shallow dish of pondwater and watch it move leechlike across the substratum using its anterior and posterior suckers. Consider the muscles and skeleton needed to accomplish this movement. <
>1c. Haemopis and Hirudo are excellent swimmers. When swimming the leech releases both suckers and flattens the body using its dorsoventral muscles. It then uses local contractions of dorsal and ventral longitudinal muscles to generate waves of undulations that move along the body from anterior to posterior. Place a living leech in a glass aquarium of pondwater and watch it swim. Describe the motion and the muscles necessary to swim. <
References
Bourne AG . 1884. Contributions to the anatomy of the Hirudinea. Quart. J. Micros. Sci. 24:419-506, pls. 24-34.
Brown FA . (ed) 1950. Selected Invertebrate Types. Wiley, New York. 597p.
Fernandez JV, Tellez V, Olea N. 1992. Hirudinea, in Harrison FW, Gardiner SL (eds.). Microscopic Anatomy of Invertebrates vol. 7 Annelida. Wiley-Liss, New York. 418p.
Klemm DJ. 1982. Leeches (Annelida:Hirudinae) of North America. U. S. Environmental Protection Agency Pub. EPA-600/3-83-025. 117p.
Mann KH. 1962. Leeches (Hirudinae) Their Structure, Physiology, Ecology, and Embryology. Pergamon, Oxford. 201p.
Pennak RW . 1989. Fresh-water Invertebrates of the United States, 3rd ed. Wiley, New York. 628p.
Sawyer RT. 1986a. Leech Biology and Behaviour, vol. I, Anatomy, Physiology, and Behaviour. Clarendon, Oxford. pp. 1-418. + index 33p.
Sawyer RT. 1986b . Leech Biology and Behaviour, vol. II, Feeding Biology, Ecology, and Systematics. Clarendon, Oxford. pp. 419-793. + index 33p.
Sawyer RT. 1986c. Leech Biology and Behaviour, vol. III, Bibliography. Clarendon, Oxford. pp. 799-1065.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Supplies
Dissecting microscope
Compound microscope
Small dissecting pan such as can be made from sardine Living or preserved Leeches, Haemopis or Hirudo
Dissecting set with microdissecting tools
# 1 stainless steel insect pins
5 % ethanol
15% ethanol