Invertebrate Anatomy OnLine
Farfantepenaeus aztecus
Brown Shrimp ©
27jun2006
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises can be accessed by clicking on the link to the left. A glossary and chapters on supplies and laboratory techniques are also available. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Arthropoda P, Mandibulata, Crustacea sP, Eucrustacea, Thoracopoda, Phyllopodomorpha, Ostraca, Malacostraca C, Eumalacostraca, Caridoida, Decapoda O, Dendrobranchiata sO, Penaeidea iO, Penaeoidea SF, Penaeidae F, (Fig 16-15, 19-67, 19-90)
Arthropoda P
Arthropoda, by far the largest and most diverse animal taxon, includes chelicerates, insects, myriapods, and crustaceans as well as many extinct taxa such as Trilobitomorpha. The segmented body primitively bears a pair of jointed appendages on each segment. The epidermis secretes a complex cuticular exoskeleton which must be molted to permit increase in size. Extant arthropods exhibit regional specialization in the structure and function of segments and appendages but the ancestor probably had similar appendages on all segments. The body is typically divided into a head and trunk, of which the trunk is often further divided into thorax and abdomen.
The gut consists of foregut, midgut, and hindgut and extends the length of the body from anterior mouth to posterior anus. Foregut and hindgut are epidermal invaginations, being derived from the embryonic stomodeum and proctodeum respectively, and are lined by cuticle, as are all epidermal surfaces of arthropods. The midgut is endodermal and is responsible for most enzyme secretion, hydrolysis, and absorption.
The coelom is reduced to small spaces associated with the gonads and kidney. The functional body cavity is a spacious hemocoel divided by a horizontal diaphragm into a dorsal pericardial sinus and a much larger perivisceral sinus. Sometimes there is a small ventral perineural sinus surrounding the ventral nerve cord.
The hemal system includes a dorsal, contractile, tubular, ostiate heart that pumps blood to the hemocoel. Excretory organs vary with taxon and include Malpighian tubules, saccate nephridia, and nephrocytes. Respiratory organs also vary with taxon and include many types of gills, book lungs, and tracheae.
The nervous system consists of a dorsal, anterior brain of two or three pairs of ganglia, circumenteric connectives, and a paired ventral nerve cord with segmental ganglia and segmental peripheral nerves. Various degrees of condensation and cephalization are found in different taxa.
Development is derived with centrolecithal eggs and superficial cleavage. There is frequently a larva although development is direct in many. Juveniles pass through a series of instars separated by molts until reaching the adult size and reproductive condition. At this time molting and growth may cease or continue, depending on taxon.
Mandibulata
Mandibulata is the sister taxon of Chelicerata and in contrast has antennae on the first head segment, mandibles on the third, and maxillae on the fourth. The brain is a syncerebrum with three pairs of ganglia rather than the two of chelicerates. The ancestral mandibulate probably had biramous appendages and a J-shaped gut, posterior-facing mouth, and a ventral food groove. The two highest level mandibulate taxa are Crustacea and Tracheata.
Crustacea sP
Crustacea is the sister taxon of Tracheata and is different in having antennae on the second head segment resulting in a total of 2 pairs, which is unique. The original crustacean appendages were biramous but uniramous limbs are common in derived taxa. The original tagmata were head but this has been replaced by head, thorax, and abdomen or cephalothorax and abdomen in many taxa. Excretion is via one, sometimes two, pairs of saccate nephridia and respiration is accomplished by a wide variety of gills, sometimes by the body surface. The nauplius is the earliest hatching stage and the naupliar eye consists of three or four median ocelli.
Eucrustacea
Eucrustacea includes all Recent crustaceans except the remipedes. The taxon is characterized by a primary tagmosis consisting of heat, thorax, and abdomen although the derived condition of cephalothorax and abdomen is more common. Eight is the maximum number of thoracic segments.
Thoracopoda
In the ancestral thoracopod the thoracic appendages were turgor appendages used for suspension feeding in conjunction with a ventral food groove. Such appendages and feeding persist in several Recent taxa but have been modified in many others.
Phyllopodomorpha
The compound eyes are stalked primitively although derived sessile eyes occur in many taxa.
Malacostraca C
Malacostraca includes most of the large and familiar crustaceans such as crabs, shrimps, lobsters, crayfish, isopods, and amphipods. Primitively the trunk consists of 15 segments, eight in the thorax and seven in the abdomen but in most Recent species the abdomen has only six segments. The female gonopore is on the eighth thoracic segment and the male on the sixth.
Decapoda O
The largest and most familiar crustaceans belong to Decapoda. The 10,000 species of crabs, shrimps, crayfishes, lobsters, and their relatives are decapods. The first three segments of the decapod thorax are fused with the head to form a cephalothorax and their appendages are maxillipeds. The remaining five pairs of thoracic appendages bear simple or chelate walking legs. The resulting ten legs accounts for the name “decapod”. A large carapace extends posteriorly from the head and is fused dorsally with all eight thoracic segments. Laterally the overhang of the carapace encloses the branchial chamber with the gills. The most primitive decapods (shrimps, lobsters, and crayfishes) have well developed abdomens whereas the most derived taxa (true crabs in Brachyura) have reduced, almost vestigial, abdomens (Fig 19-24).
Laboratory Specimens
This exercise was written for the brown shrimp, Farfantepenaeus aztecus, but can be used equally well with any penaeid shrimp. On the North American east coast the most common penaeid shrimps are Farfantepenaeus aztecus (brown shrimp), F. duorarum (pink shrimp) and Litopenaeus setiferus (white shrimp). Other species, such as Litopenaeus vannamei is a Pacific species. The anatomy of the species is nearly identical and any of them can be used for this exercise.
Study a penaeid shrimp in a culture dish of liquid. It may be a relaxed living specimen, a fresh dead or a frozen specimen from the fish market, or a preserved one. Living specimens should be in isotonic magnesium chloride, fresh specimens in seawater, and preserved material should be in tap water. Most of the study should be made with a dissecting microscope.
External Anatomy
Tagmata
Note the overall shape of the animal (Fig 1, 19-1, 19-1, 19-25A). It exhibits the "caridoid facies", or typical primitive shrimp-like morphology. The animal is elongate and slightly laterally compressed, or flattened from side to side. It has appendages on all segments and the abdomen is muscular and powerful.
Head
The body consists of three tagmata; the anterior head, middle thorax, and posterior abdomen (Fig 19-1). The head consists of five segments but they are fused together, covered by a single sclerotized head capsule and cannot be distinguished externally. Each segment has a pair of appendages.
Anteriorly the head bears a pair of eyestalks beside the base of the median rostrum (Fig 1). The eyestalks articulate with the head and are movable. Distally each one bears a swollen, dark, convex cornea. The cornea is the outer, exoskeletal part of the compound eye. Under higher magnification (40x) it can be seen to be composed of multitudes of facets, or tiny lenses. Notice the orientation and extent of the cornea and speculate on the directions in which the shrimp has the most acute vision. Move the eyestalk up and down and observe (with a dissecting microscope) the complex interactions of the skeletal elements inside the eyestalk.
In all decapods the head and three anterior thoracic segments are fused together to form a cephalothorax. The head and entire thorax, not just the cephalothorax, are covered dorsally and laterally by a fold of body wall called the carapace (Fig 1).
Thorax
The eight segments of the thorax are not visible dorsally and laterally because they are covered by the carapace but some of them can be seen ventrally. The thoracic segments arethoracomeres and the appendages they bear arethoracopods.
In all decapod crustaceans the first three thoracomeres are fused with the head to form the cephalothorax. The appendages of these segments are modified to function more or less as mouthparts and are called maxillipeds. The remaining five thoracomeres are not fused with the head and are called "free thoracomeres", or pereomeres. Together these five free segments make up the pereon. Their appendages are known by the general name of walking legs, or pereopods. In all malacostracans, the pereon consists of the thoracic segments that are not fused with the head but the number (five in decapods) varies with taxon.
In decapod crustaceans the first pereopod (4 th thoracopod) bears a pincer, or chela, and is called the cheliped. In many decapods, e.g. crabs, it is large and powerful but in penaeid shrimps the chela is small and weak. In these shrimps the second and third pereopods are also chelate (bear a chela). The remaining two pairs of pereopods are not chelate and are said to be simple.
Figure 1. The brown shrimp, Farfantepenaeus aztecus. Redrawn from Farfante (1969).

Carapace
The crustacean carapace develops as an outgrowth of the posterior margin of the head (Fig 19-1). It grows posteriorly and laterally to cover a variable number of thoracic, and sometimes abdominal, segments. In the decapods it covers the entire thorax. Because it is covered by the carapace, the thorax appears to be a part of the head but, in fact, it is not. The carapace is a fold of the body wall that covers, and may be attached to, the thorax. In shrimps the carapace can be lifted to reveal the thorax under it. The right and left sides of the carapace are the branchiostegites that enclose the large lateral branchial chambers which house the gills (Fig 19-36, 19-3A).
The carapace is a double layer of body wall (Fig 5). The outer layer is relatively heavy and sclerotized whereas the inner layer is thin and unsclerotized. Sandwiched between the two layers is hemocoel.
The carapace bears a raised longitudinal keel, or carina, along the dorsal midline (Fig 1). The carina is flanked by a groove on each side. The carina extends anteriorly as the sharp, toothed rostrum.
Abdomen
The third tagma is the abdomen (Fig 1). In Malacostraca it has six segments and in the shrimps, lobsters, and crayfishes it is well developed and muscular. Its six segments are numbered, as are the eight of the thorax, from anterior to posterior. The abdominal segments are called pleomeres (= pleonites) and the first five pairs of appendages are pleopods(pleo=swimming).
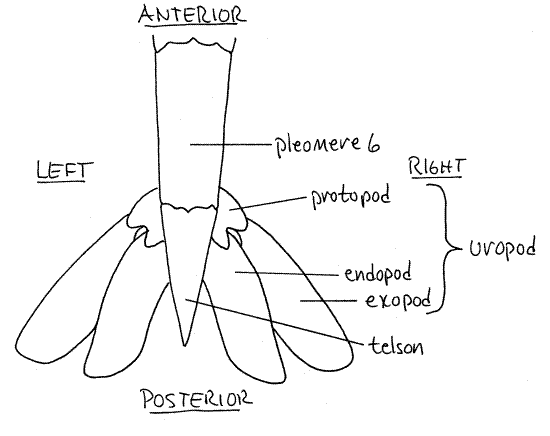
The sixth segment bears the uropods laterally and the unpaired median telson posteriorly (Fig 1). The telson is a narrow triangle. Its ventral surface bears a median slit-like anus near the base. The telson is not considered to be a true segment and it does not bear appendages.
Exoskeleton
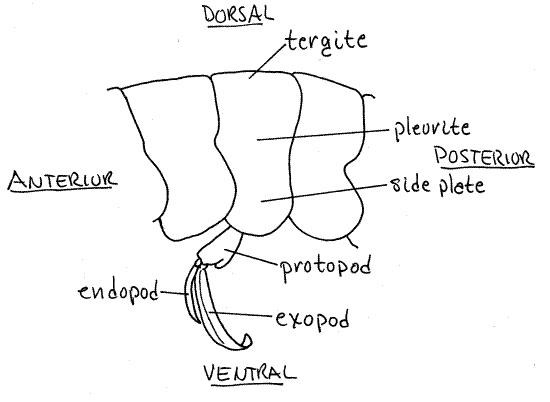
Look at the exoskeleton of an abdominal segment. It forms a skeletal ring composed of the usual sclerites of an arthropod segment although some of them are fused together (Fig 16-1B). The dorsal sclerite is a tergite. It is continuous on each side with a lateral pleurite. The pleurites extend ventrally as side plates (Fig 3), or epimera, and together make a shallow ventral channel in which the pleopods are located. Ventrally the exoskeletal ring is completed by the sternite, which articulates with the pleurites. The sternite is thin and flexible over most of its surface but at the points of articulation of the pleopods it is thicker and stronger. Flex the abdomen with your fingers and note that each segment moves with respect to its neighbors around a central pivot point, or hinge, that is marked on the posterior pleural borders by a small concavity.
Look at the exoskeleton under magnification. You can easily see the small star-shaped chromatophores in the dermis of the integument below the exoskeleton (Fig 19-44A,B). Note that the exoskeleton is thin and flexible. In most shrimps it is not reinforced by deposits of calcium salts and it consists primarily of chitin and protein.
Appendages
The study of appendages is best conducted from posterior to anterior. Appendages can be studied in situ, without removing them, or by removing each one in turn. In most cases it is better to study the appendages without removing them. In either case begin the study at the posterior end and work your way anteriorly, one appendage at a time.
Begin the study of appendages by reviewing the morphology of a typical crustacean limb. It is jointed, or composed of articulated sections called articles and is primitively biramous. Abiramous appendage has a basal article, the protopod that attaches by its proximal end to the body (Fig 2, 19-1). Sometimes the protopod is divided into two articles. From its distal end arise two rami, or branches. The rami are an outer, or lateral, exopod and an inner, or medial, endopod (Fig 2).
The two rami may be composed of any number of articles depending on their function. They may be similar to each other or different. Sometimes only one ramus is present and the appendage is uniramous. Sometimes there are additional branches of the protopod or of the rami. Any additional branch on the lateral side is an exite and any extra medial branch is anendite. Finally, an exite on the base of the appendage is given the special name of epipod.
Uropods
As you have just seen, the sixth abdominal segment bears the telson. It also bears a pair of uropods (Fig 1) attached ventrolaterally beside the base of the telson (uro = tail). The uropods are biramous and each consists of a proximal protopod and two broad, flat, paddlelike rami (Fig 2). The lateral ramus is the exopod and the medial one is the endopod. The flat telson and the two flat rami of each uropod make up the tail fan (Fig 2). The shrimp can expand the tail fan and use the powerful abdominal muscles to flex the abdomen. This causes the animal to move rapidly backwards in the characteristic "escape" response of shrimps, crayfish, and lobsters. With your fingers, expand the tail fan and speculate about its efficiency as a paddle.
Figure 2. Dorsal view of the tail fan of the brown shrimp, Farfantepenaeus aztecus. Redrawn from Farfante (1969).
Pleopods
The anterior five abdominal segments each bear a pair of pleopods, or swimmerets (Fig 1). These are flexible, setose, usually biramous appendages used for swimming forward. In some decapods, but not penaeids, the female carries eggs on her pleopods.
Each pleopod consists of a basal protopod, a small endopod, and a larger exopod (Fig 3). The first pleopod of females has only one ramus as the endopod is missing. In males, the endopods of the first abdominal appendages are fused medially with each other to form a petasma. The petasma is an intromittent organ that transfers sperm from the male gonopores on thoracomere 8 (pereomere 5) to the seminal receptacle (thelycum) of the female.
Pereopods
Beginning at the posterior end of the pereon, look now at the thoracic appendages. The appendages of the five free thoracomeres (pereomeres) are pereopods, or walking legs. Segments and appendages of the pereon can be named in reference to either the thorax or the pereon, whichever is convenient. Thus pereomere 1 is the same as thoracomere 4.
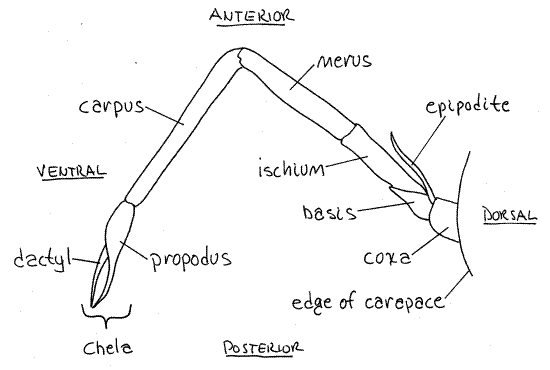
Look at the fifth pereopod (eighth thoracopod). This is the posteriormost thoracic appendage. The typical malacostracan pereopod consists of seven articles. Find them on the 5 thleg. The short proximal article attached to the body is the coxa (Fig 1). Distally the coxa articulates with a shorter, narrower basis. The basis articulates with the ischium. The ischium is longer and narrower than the basis. The remaining articles are also long and narrow and are, in order following the ischium, the merus, carpus, propodus, and dactyl (Fig 4, 19-1, 19-20). The dactyl is sharply pointed. Wiggle the articulations between each pair of articles and note the directions in which the articulation allows movement and the directions in which it resists movement.
Figure 3. The third pleomere and its pleopod of the brown shrimp, Farfantepenaeus aztecus. Redrawn from Farfante (1969).
The first three pereopods of penaeid shrimps are chelate. Examine the first of these. It is thoracopod 4 (pereopod 1). Find the seven articles that contribute to it and note the relationship between the distal two; ie the dactyl and propodus. In a chelate appendage the propodus has a distal process called the immovable finger that forms half of the chela (Fig 4). The dactyl is the movable finger that opposes it and is the other half of the chela.
Most of the walking legs (and the maxillipeds too for that matter) have a small lateral exopod on the basis and a larger, also lateral, epipodite on the coxa (Fig 4). The leg itself is the endopod.
The medial border of the coxa of the female pereopod 3 (thoracopod 6) bears the opening of the oviduct in the center of a low genital papilla. Between the bases of the fifth pereopods, the female has a large, bulbous, median thelycum, which serves as a seminal receptacle.
The male gonopore, which is the opening of the vas deferens, is located on a medial expansion of the coxa of the fifth pereopods. The male first pleopods on the first abdominal segment transfer sperm from the gonopore to the female thelycum. Males have no thelycum.
Branchial Chamber and Gills
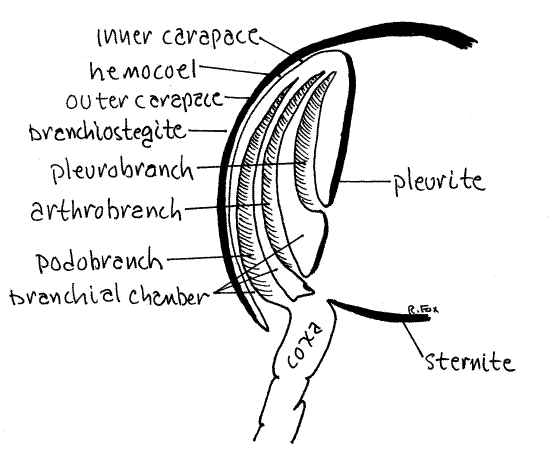
The two sides of the carapace are the branchiostegites. The space between the branchiostegites and the thorax is the branchial chamber (Fig 5). Lift the posterior corner of the left branchiostegite and look under it. You are looking into the branchial chamber and the grayish white tissues you see are the gills. The gills are outgrowths, or epipods, of the thoracopods.
Figure 4. Lateral view of the third pereopod of the brown shrimp, Farfantepenaeus aztecus. Redrawn from Farfante (1969).
" Remove the left branchiostegite. Do this by carefully cutting (with scissors) along an oblique line sloping anteroventrally (down) from a starting position high on the posterior lateral edge of the carapace. Do not cut into the attached portion of the carapace. You can feel the line of attachment with the point of your scissors and should avoid it. Removal of the carapace opens the branchial chamber and reveals its many gills.
Remember that the carapace is a fold of body wall and as such has two layers of exoskeleton and epidermis with hemocoel in between. Notice that the outer of these two layers of exoskeleton is relatively hard, whereas the inner is very thin and soft.
>1a. Make a wet mount of a piece of the inner wall of the carapace and examine it with the compound microscope (400X). Look for chromatophores (Fig 19-44A,B). These are star- or amoeba-shaped pigment-containing organs capable of changing the color of the integument. <
Examine the branchial chamber and find the bases of the five pereopods. Study the base of pereopod 5 (5 th walking leg) prior to examining the gills. Observe that the coxa of each leg articulates with the pleurite of its segment via a long white, thin, flexible articulating membrane extending obliquely, posteriorly, and dorsally from the coxa to the pleurite. The pleurite is harder than the articulating membrane.
There are three regions, the coxa, articulating membrane, and pleurite, where gills could be attached (Fig 5). Gills can be outgrowths (epipods) of any of these three areas and are classified according to their site of attachment. Podobranch gills are attached to the coxa. Arthrobranch gills are attached to the articulating membrane around the coxa. Pleurobranch gills are attached to the pleurite.
Thoracopod 8 (pereopod 5) has no gills. Pereopod 4 has one pleurobranch and one arthrobranch gill. Pereopods 1, 2, 3 have one pleurobranch and two arthrobranch gills each. So far we have accounted for 11 gills (on each side) but there are more.
Figure 5. The branchial chamber of a generalized decapod in cross section. shrimp20L.gif
" Snip the end from one of the gills, place it in a small dish (6-cm culture dish) of water and examine it with the dissecting microscope.
Penaeid shrimps have dendritic (= dendrobranchiate) gills in which the respiratory surface consists of numerous long branched filaments radiating from a central axis (Fig 19-37A,B).
Look at the cut surface of the gill axis to find two blood channels, cut in cross-section, that extend the length of the gill (Fig 19-37A). One is the afferent channel that delivers unoxygenated blood to the gill and the other is the efferent vessel that drains oxygenated blood away from the gill.
Maxillipeds
Continue examining the appendages without removing them from the body. The remaining three thoracopods are maxillipeds (instead of pereopods) and belong to the three thoracomeres that are fused with the head to contribute to the cephalothorax. Thoracopod 3 (maxilliped 3) looks much like a walking leg (pereopod) (Fig 1, 19-1). It is not chelate but has a protopod with a long stenopodous (leg-like) endopod. The endopod arises from the basis of the protopod and consists of an ischium, merus, carpus, propodus, and dactyl as is typical of thoracopod endopods. The protopod also bears a narrow, setose, phyllopodous exopod that is much larger than, but similar to, the exopods on the pereopods. The coxa bears an epipod. Maxilliped 3 has one pleurobranch and two arthrobranch gills. (Now we have accounted for 14 gills.)
Maxilliped 2 (thoracopod 2) does not look so much like a walking leg. It has a narrow, setose, phyllopodous exopod as does maxilliped 3, but its endopod is shorter and its articles are flattened somewhat. The seven articles of the typical thoracic endopod are present however. Its coxa has an epipod like that of most thoracopods. Its gills are a podobranch and two arthrobranchs. (That’s the last of the gills for a total of 17 pairs in Farfantepenaeus.)
Maxilliped l (thoracopod l) looks nothing like a walking leg but closely resembles a maxilla. Its exoskeleton is thin, flexible, and delicate. It is biramous but bears no gills. Its structure is complicated and it has several processes from the basal region. The exopod is easiest to recognize. It is the long narrow ramus near the center of the appendage. Laterally are two broad thin epipods. There is an endite medial to the narrow exopod. The medial border of the endite is heavily setose. There is no endopod.
Mouthparts
The crustacean head consists of five fused segments, each with a pair of appendages. These are two pairs of antennae anterior to the mouth, a pair of mandibles flanking the mouth, and two pairs of maxillae posterior to the mouth. The mandibles and maxillae are the mouthparts. Because the posterior appendages tend to overlie and obscure those anterior to them, it is best to begin their study with the posteriormost (maxilla 2) and work anteriorly. It is better to study the mouth parts in place in the animal rather than remove them.
The posteriormost appendage of the crustacean head is the biramous second maxilla. It is situated just anterior to the first maxilliped, which you just examined. Its central coxa bears two medial endites. Each one is subdivided into two lobes. Endite l is posterior, endite 2 anterior. The medial borders are setose. Laterally there is a large flat leaf-like scaphognathite, or gill bailer. The unsegmented endopod is a short blunt ramus between the scaphognathite and the endites. The coxa is not recognizable as a separate entity, but is the central region to which the above processes attach.
The scaphognathite is formed of the exopod and an epipodite. Its lateral border is curved and bears a fringe of setae. The motion of the scaphognathite pumps water through the branchial chamber (Fig 19-38A).
Examine the body wall beside the second maxilla. Notice that it is very thin, scarcely more than a lightly cuticularized membrane, yet it is the complete body wall that separates the interior of the animal from the exterior. The cavity you see on its opposite side of it the hemocoel (Fig 19-3A). This thin membrane is all that separates the seawater environment from the blood inside the body.
The first maxilla (= maxillule) lies anterior to the second maxilla and is the smallest and most delicate of the mouthparts. It has no exopod or epipods. Medially it bears two largeendites. Endite 1 is posterior and endite 2 is anterior to it. Both have setose medial margins. The lateral region of the first maxilla is a long, delicate, stenopodous endopod.
Anterior to the first maxilla and covering the medial surface of the hard, smooth, rounded dome of the mandibles is a pair of fleshy lobes, the paragnaths. They are unsegmented outgrowths of the body wall and are not segmental appendages.
The heavily cuticularized mandibles are covered by the curved anterior surface of the first maxillae. Lift these maxillae to expose them. The coxa of the mandible is greatly enlarged to form a smooth, heavy, hard jaw which bears teeth on its medial border. Emerging from the anterior lateral corner of the coxa is a broad, thin, leaflike mandibular palp. It is segmented and is the endopod of this appendage. Its proximal article is the basis which articulates with the coxa.
The mouth is located ventrally between the medial borders of the mandibular coxae. Anterior to the mouth and mandibles is a low conical outgrowth of the body wall called theepistome.
Antennae
The remaining head appendages are two pairs of antennae. The second antennae are located just anterior to the mouth and mandibles (Fig 1). They are biramous with a single basalprotopod from which arises a long sensory flagellum and a short, broad, flat antennal scale. The protopod bears a small urinary papilla medially on its anterior margin. Thenephridiopore is located on the urinary papilla. This is the opening of the antennal gland, or kidney.
The first antennae (= antennules) are relatively short anterior sensory appendages located just ventral to the eyestalks. Each consists of a basal portion with three articles and two small distal flagella. The basal article has a small endite. This article has a broad dorsal concavity to accommodate the cornea at the end of the eyestalk. There is a statocyst for gravity detection in the basal article (Fig 19-7B).
Internal Anatomy
Dissection and study of the internal anatomy is difficult, especially with fish market specimens of dubious freshness. It is best to forego this part of the exercise unless living or preserved specimens are available. If you wish to continue with the dissection, proceed as follows. Living specimens should be in isotonic magnesium chloride. Dead fresh or preserved animals should be in tapwater.
" Lift the lateral edges of the remains of the carapace and use a large teasing needle (not a minuten nadel) to separate the carapace from the underlying body wall. Mostly, this is simply a matter of moving the needle between the hard outer cuticle and the soft underlying body wall. Anteriorly, however, there are powerful muscles that insert on the inner surface of the carapace and these must be separated from the carapace. When all the tissue connections are severed the carapace will remain attached to the exoskeleton of the front of the head from which it must be cut or carefully torn with forceps. Remove the carapace and set it aside. Place the shrimp in a culture dish of isotonic magnesium chloride if it is not already in one.
Look at the thorax with the dissecting microscope. The thin, transparent body wall should still be intact.
" Make a shallow longitudinal cut, with fine scissors, through the lateral body wall from posterior to anterior on the left side. Anteriorly you will have to cut around the large, white, hardapodeme of the mandible. Note the large masses of muscle in the anterior thorax and head. Remove the second antenna by cutting through the muscle and cuticle at its base. Do not remove more than the appendage itself. Separate the remaining muscle to expose completely the conspicuously pigmented esophagus which extends almost vertically up from the mouth. Be careful that you do not damage the large white nerve (circumesophageal connective) that crosses the base of the esophagus laterally.
Digestive System
Locate the mouth and esophagus. Dorsally the esophagus turns posteriorly where it joins the anterior (or cardiac) region of the large two-part stomach.
The stomach and esophagus are part of the foregut and are lined inside by exoskeleton. In the cardiac stomach the cuticle is elaborated to form a complex and intricate gastric mill to grind food (Fig 19-34).
Posterior to the cardiac stomach is a smaller stomach region, the pyloric stomach which contains a sieve, or filter press, made of cuticular setae. The sieve separates fine food particles from coarser wastes and shunts the fine material into the openings of the digestive ceca and allows the coarse material to pass into the intestine from which it eventually exits via the anus. The coarsest particles are probably regurgitated from the cardiac stomach out the mouth.
The digestive ceca are large, yellow, highly branched organs that fill most of the hemocoel (unless the specimen is reproductively active and has large gonads, in which case the gonads will be larger). The pyloric stomach and anterior intestine quickly deteriorate in dead unpreserved specimens. They may not be present if you have a shrimp from the fish market.
Find the intestine where it exits the pyloric stomach and trace it into the muscle mass of the abdomen. The intestine is the midgut and the digestive ceca connect to its extreme anterior region. It courses dorsally through this mass to terminate at the anus on the ventral surface of the telson.
" Open the abdominal muscle mass by making two longitudinal cuts (use scissors) through the exoskeleton of the abdomen. These cuts should be located dorsolaterally so that a median strip of dorsal exoskeleton about 1 cm wide can be lifted from the top of the abdomen. You need not be particularly careful of the underlying muscle as you make these cuts. If you cut too deeply however, you could damage the intestine.
After you have made the lateral cuts, remove the median strip of exoskeleton and deepen the cut through the muscle on the left. There is a natural division between right and left muscle masses in this region and you need only accentuate it. When you have done so, you can lift the median dorsal muscle mass away from the lateral masses and reveal the intestine on the midline between the two masses. Its thin walls bear chromatophores dorsally. Dorsal to it are the two dorsal abdominal arteries whose walls also bear chromatophores. Trace the intestine to the anus.
Nervous System
Return to the anterior end of the animal and relocate the left circumesophageal connective lying atop the ventral extremity of the esophagus. Follow it anteriorly to the brain, cutting or teasing your way through muscle and cuticle as necessary. The brain is located between the bases of the eyestalks. Notice that it, and the nerves, bear black chromatophores on their surfaces. The tripartite nature of the crustacean brain is not readily apparent but the three major pairs of sensory nerves that enter it are easily seen.
Anteriormost is the pair of large optic nerves extending from the eyestalks to the anterior brain (protocerebrum) (Fig 16-10A). Next, is a smaller antennular nerve extending from the first antennae (antennules) to the deutocerebrum. Finally, is a pair of large antennal nerves from the second antennae to the posterior tritocerebrum.
The right and left circumesophageal connectives exit the posterior brain and make a ring around the esophagus. They reconnect well posterior to the esophagus at thesubesophageal ganglion from which exits the double, ganglionated, ventral nerve cord.
There is some tendency to cephalization in these primitive decapods and the subesophageal ganglion, while small, nevertheless contains the segmental ganglia for the mouth parts and maxillipeds. If you lift the ganglion in this region you will see the nerves to these appendages exiting the cord ventrally. The ganglia of the pereon and abdomen, however, are not included in the subesophageal ganglion and remain segmentally distributed along the ventral nerve cord.
Trace the nerve cord posteriorly (after you have finished with the intestine and dorsal abdominal artery). Sever the transverse connective tissue connections holding the right and left muscle masses together in the floor of the thorax and abdomen. Push the two masses apart and follow the nerve cord posteriorly through the thorax and abdomen, cutting transverse connective tissues as necessary. The nerve cord diminishes in diameter in the abdomen. Find the segmental nerves exiting the cord at each segmental ganglion.
Below the nerve cord can be seen the thin transparent sternites of the abdominal exoskeleton. At about the level of the 2 nd pereopod, the nerve cord divides to pass around the sternal artery from the dorsal abdominal artery.
Hemal System
The open spaces in the body are the hemocoel and contain blood (hemolymph). The blood is pumped by the heart through a complex array of arteries to the hemocoel (Fig 19-3A). The heart is surrounded by a compartment of the hemocoel, the pericardial sinus. The sinus is not a coelomic space and hence is not a true pericardium.
The heart lies in pericardial sinus in the roof of the posterior thorax (Fig 19-2). It is short and wide, and tapering anteriorly and posteriorly. It is pale and nearly colorless. It is suspended in the cardiac hemocoel by alary ligaments and muscles and bears ostia in its lateral walls.
A pair of large posterior dorsal abdominal arteries run longitudinally dorsal to the intestine and give off segmental branches to the abdominal muscles and intestine. It is difficult to trace the blood vessels in fresh specimens but you should make an attempt. The connections with the heart are especially fragile and easily broken.
The sternal artery exits the dorsal abdominal artery near the heart and slopes steeply anteriorly to penetrate the ventral nerve cord at about the level of the 2 nd pereopod.
Ventral to the nerve cord the sternal artery bifurcates to form the ventral thoracic artery and the ventral abdominal artery running anteriorly and posteriorly respectively. Lateral segmental branches of these vessels can be seen extending to the structures in their segments.
Anteriorly from the heart there exits a median unpaired ophthalmic artery to the brain, eyestalks and first antennae. At a position above the anterodorsal surface of the cardiac stomach it swells to form the cor frontale, which is an accessory heart. A pair of antennary arteries also exit the heart anteriorly and run to the carapace, most of the viscera and much of the thoracic musculature.
Excretory System
The decapod excretory organs are a pair of antennal glands (= saccate nephridia) located on the floor of the head and emptying by a duct to the nephridiopores on the coxae of the 2nd antenna (Fig 19-6B). The left antennal gland may have been inadvertently destroyed during dissection of the left side of the cephalothorax.
" Examine the right side of the cephalothorax beside the esophagus in the ventral part of the head. Cut through the large muscles in this region to reach the floor of the head.
The antennal gland is a small white pad of tissue on the floor of the head just anterior to and lateral to the esophagus. It looks very much like the muscles in the region and is difficult to locate. It is not fibrous like the muscles however.
The antennal gland is a saccate nephridium derived from the ancestral metanephridium. In the decapods it is also known as the green gland or coxal gland but it is not green in shrimps.
Reproductive System
Unless your specimen is in reproductive condition the reproductive system will be difficult to study. The gonads (ovaries or testes) are paired tubular structures in the thorax. They connect with the gonopores via paired gonoducts (oviducts or vasa deferentia. The female gonopores are on thoracomere 6 (on the coxae of pereopod 3) and the male pores are on thoracomere 8 (on the coxae of pereopod 5).
References
Farfante IP. 1969. Western Atlantic shrimps of the genus Penaeus. Fishery Bulletin 67(3):461-591.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Williams AB. 1984. Shrimps. lobsters, and crabs of the Atlantic coast of the eastern United States, Maine to Florida. Smithsonian Institution Press, Washington.
Supplies
Dissecting microscope
20-cm culture dish
Isotonic magnesium chloride
Seawater
Living, fresh, or preserved Farfantepenaeus