Invertebrate Anatomy OnLine
Crassostrea virginica ©
American oyster
25may2007
Copyright 2005 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises, a glossary, and chapters on supplies and laboratory techniques are also available at this site. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Mollusca P, Eumollusca, Conchifera, Ganglioneura, Ancyropoda, Bivalvia C, Metabranchia sC, Filibranchia SO, Pteriomorpha O, Ostreoidea SF, Ostreidae F (Fig 12-125, 12-122)
Mollusca P
Mollusca, the second largest metazoan taxon, consists of Aplacophora, Polyplacophora, Monoplacophora, Gastropoda, Cephalopoda, Bivalvia, and Scaphopoda. The typical mollusc has a calcareous shell, muscular foot, head with mouth and sense organs, and a visceral mass containing most of the gut, the heart, gonads, and kidney. Dorsally the body wall is the mantle and a fold of this body wall forms and encloses that all important molluscan chamber, the mantle cavity. The mantle cavity is filled with water or air and in it are located the gill(s), anus, nephridiopore(s) and gonopore(s). The coelom is reduced to small spaces including the pericardial cavity containing the heart and the gonocoel containing the gonad.
The well-developed hemal system consists of the heart and vessels leading to a spacious hemocoel in which most of the viscera are located. The kidneys are large metanephridia. The central nervous system is cephalized and tetraneurous. There is a tendency to concentrate ganglia in the circumenteric nerve ring from which arise four major longitudinal nerve cords.
Molluscs may be either gonochoric or hermaphroditic. Spiral cleavage produces a veliger larva in many taxa unless it is suppressed in favor of direct development or another larva. Molluscs arose in the sea and most remain there but molluscs have also colonized freshwater and terrestrial habitats.
Eumollusca
Eumollusca, the sister taxon of Aplacophora, includes all molluscs other than aplacophorans. The eumolluscan gut has digestive ceca which are lacking in aplacophorans, the gut is coiled, and a complex radular musculature is present.
Conchifera
Conchifera, the sister taxon of Polyplacophora, includes all Recent molluscs other than aplacophorans and chitons. The conchiferan shell consists of an outer proteinaceous periostracum underlain by calcareous layers and is a single piece (although in some it may appear to be divided into two valves). The mantle margins are divided into three folds.
Ganglioneura
Most Recent molluscs are ganglioneurans, only the small taxa Aplacophora, Polyplacophora, and Monoplacophora are excluded. Neuron cell bodies are localized in ganglia.
Ancyropoda
The mantle cavity, with its gills, is lateral. The calcareous portion of the shell is bivalve, with the valves opening laterally and joined dorsally by a derivative of the periostracum.
Bivalvia C
Bivalvia is a large, successful, and derived taxon. The body is laterally compressed and enclosed in a bivalve shell. The two valves are hinged dorsally. The foot is large and adapted for digging in the ancestral condition. A crystalline style is usually present but never is there a radula. The mantle cavity is lateral and in most bivalves the gills are large and function in respiration and filter-feeding. The head is reduced and bears no special sense organs. The nervous system is not cephalized. The group includes scallops, clams, shipworms, coquinas, marine and freshwater mussels, oysters, cockles, zebra mussels, and many, many more.
Metabranchia sC
Most bivalves are metabranchs. The gills are adapted for filter feeding and water enters the mantle cavity posteriorly.
Filibranchia SO
Filibranchs are suspension-feeding bivalves with filibranch gills.
Pteriomorpha O
Pteriomorph bivalves are benthic and live on the bottomof ther sea where they may be epifauna. They may be attached or unattached, may have a byssus or not, and may cement one valve to the substratum or not. The foot is reduced and the mantle margins are not fused. The gills are large and used for filter feeding. There is a tendency to reduce or loose the anterior adductor muscle. Siphons are absent or reduced. Pteriomorpha includes the well-known arcs, mussels, scallops, pen clams, jingles, and oysters.
Laboratory Specimens
This exercise is written specifically for the American oyster, Crassostrea virginica but it applies equally well to other species of Crassostrea and with a little modification, to Ostrea. Crassostrea has a large promyal chamber on the right side (Fig 6) that is lacking in Ostrea. Crassostrea is oviparous, whereas Ostrea eggs are fertilized in the exhalant mantle cavity and gestated there. Crassostrea is gonochoric, although sometimes they change sex after a breeding season, whereas Ostrea is a simultaneous hermaphrodite.
Oysters are not particularly good examples of metabranch bivalves for they differ from the "typical" condition in several respects. They have only one adductor muscle, have no foot and no siphons and have the enigmatic promyal chamber.
Oysters can be collected along the east coast of the United States from protected, estuarine habitats. They are most abundant on soft bottoms in the intertidal zone where they form reefs composed of countless individuals cemented together to form large clumps exposed at low tide. Live oysters can also be purchased at many fish markets or supermarkets at inland locations.
If possible lean specimens that have just finished spawning and thus have no gametes or glycogen reserves to obscure your view of other anatomical features. Such specimens are available in September, October, and November. Reproductive oysters with ripe gonads have thick mantles with a creamy–yellowish color. Individuals with abundant glycogen reserves also have thick mantle skirts but they are white. Starved individuals that have exhausted their glycogen reserves have thin transparent mantles and watery tissues through which organs are unobscured and readily studied. These are best suited for study. If lean individuals are not available, fat ones can be used and are satisfactory for studying the features described in this exercise.
Living specimens should be anesthetized and relaxed in isotonic magnesium chloride. Use a dissecting microscope as needed.
The edges of oyster shells are very sharp and can easily inflict cuts when handled carelessly. Wear gloves whenever it is necessary to grasp the oyster firmly. Injury from a slipping oyster knife is also possible. Exercise care when opening the shell.
Natural History
Oysters are attached epifaunal bivalves that live with the left valve cemented to the substratum. They have abandoned the ancestral bivalve biotope of burrowing in soft sediments. Consequently the have lost the foot and siphons. They have also lost the anterior adductor muscle, retaining only the posterior adductor. Lacking a foot, they also lack the pedal protractor and retractor muscles.
Oysters typically grow tightly packed in clumps, or reefs, in which they use each other as substrata for the attachment of the left valve. Because of this crowding the shape of the shell is highly variable.
Crassostrea virginica is the commercially harvested oyster of the east coast of North America. It occurs from the Gulf of St. Lawrence south to the Caribbean Sea. Sexually mature individuals vary from 5-35 cm in length.
The small, commensal oyster pea crab, Pinnotheres ostreum, lives in the mantle cavity where it may cause deformation of the gills. Oysters are attacked by several predators including the starfish, Asterias forbesi, and the oyster drill, Urosalpinx cinerea,. The boring sponge, Cliona celata, inhabits the shell and weakens it. Predators and commensals are minimized in intertidal, brackish water habitats and it is here that oysters reach their highest population densities.
External Anatomy
Shell
Make a study of the shell. The exercise is written assuming you will use the shell of your intact specimen but, if available it is better to use a cleaned dry shell from which the animal has been removed. If necessary use a toothbrush to clean sediment and debris from the shell. Rinse the scrubbed shell with tapwater.
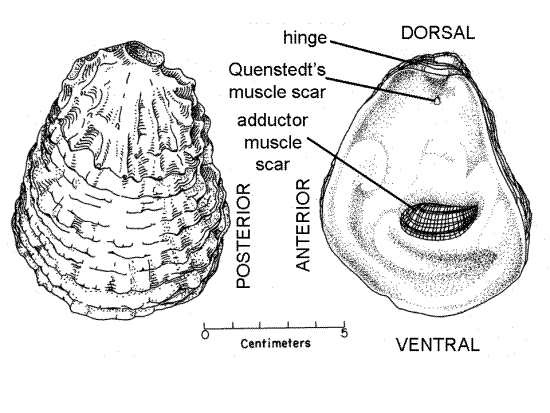
Figure 1. Crassostrea virginica. Outside of left (lower) valve and inner view of right (upper) valve (from Galtsoff, 1964). Bivalve127a.gif
External Shell Features
Study the external features of the shell. One end of the shell is narrow and the other broad. The narrow end is dorsal and the broad end is ventral (Fig 1). The shell is composed of two parts, or valves. Oysters are inequivalve bivalves with a shell composed of two unequal valves. With the exception of the unequal valves oysters, like other bivalves, are bilaterally symmetrical. The plane of symmetry passes between the two valves, one being the right valve and the other the left. The left valve is the larger of the two and is the valve that is cemented to the substratum. It is deeper than the right and forms a cup that cradles the mollusc. The right valve is smaller and flatter, forming a cover that fits over the cup of the left valve (Fig 2). Knowing dorsal-ventral and right-left, determine anterior and posterior (Fig 1, 5). Confirm your determination by referring to the curvature of the two long margins. In most specimens the anterior margin is a relatively smooth convex curve whereas the posterior margin is concave and often irregular (Fig 1). Unfortunately, this is not true of all shells.
Oysters are elongated along the dorso-ventral axis in contrast with the anterior-posterior elongation of most bivalves. This can cause initial (and persistent) orientation confusion for students accustomed to typical bivalves.
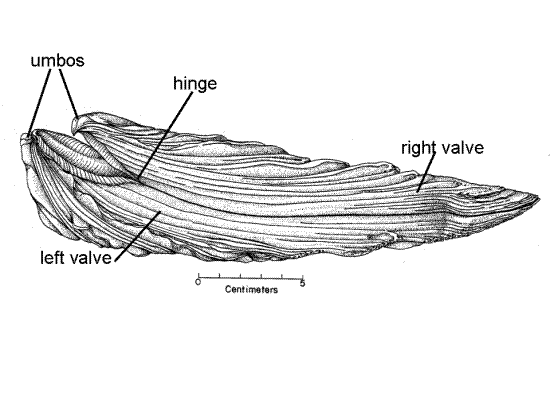
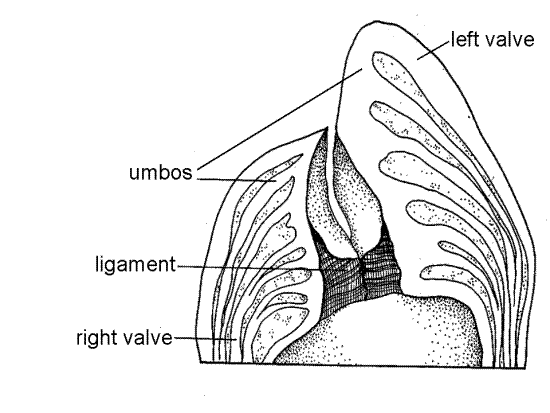
The two valves are joined to each other by a hinge at the dorsal end (Fig 2). An internal protein hinge ligament in the hinge holds the two valves together. The pointed umbo, or beak, of each valve extends dorsally beyond the hinge. The tip of the umbo is the oldest part of the shell (Fig 2). The valves gape along the anterior, ventral, and posterior margins to allow the animal to feed and respire.
Internal Shell Features
" With a cotton work glove protecting your left hand (if right handed) remove the right valve from an oyster. The right valve is the smaller of the two valves. Hold the oyster, right valve uppermost, in your gloved left hand (unless you are left-handed, in which case reverse the instructions). Slip an oyster knife or screwdriver between the two umbos (Fig 2) at the narrow end of the oyster and pry the two valves apart. Do not force the knife toward your hand as it can easily slip and gouge your hand badly. Use the pressure to break the hinge ligament. You may have to try several positions of the knife before you meet with success.
Figure 2. Side view of a large, old specimen (from Galtsoff, 1964). The angle between the umbos determines the maximum movement of the right valve. Bivalve129a.gif
Breaking the hinge allows the valves to gape so you can slip the blade of a sharp scalpel between the two valves at the posterior edge of the shell (Fig 1, 5). Insert the blade until it encounters a rubbery resilient adductor muscle close to the middle of the posterior edge of the valve (Fig 5). Be careful you do not damage the soft tissue in the vicinity of the muscle. The rubbery texture of the muscle is unlike that of any other tissues and can be recognized by feel. You may also be able to look through the gape and see the muscle. When you find it, cut the adductor muscle with the scalpel. Without further cutting, use the scalpel to push all soft tissue away from the right valve as you gently lift the right valve away from the left. Detach the right valve and set it aside. All soft tissues should remain in the left valve. Place the left valve in a dish of isotonic magnesium chloride. Be sure all soft tissues are immersed in the anesthetic. Set the dish aside to allow the animal to relax while you study the interior surface of the right valve.
The oyster shell consists of three layers. Outermost is the organic periostracum composed of a thin layer of the protein conchiolin. In Crassostrea this layer is weakly developed but can be seen as a thin, transparent, glistening, yellowish film adhering to the outer surface of the valve. It is inconspicuous in young specimens and often absent in old. The prismatic layer, composed of calcium carbonate on a protein matrix, is the middle layer. Because the periostracum is thin or absent, it is the prismatic layer that is seen when viewing the outside of an oyster shell (through the periostracum). The inner layer, also composed of calcium carbonate on a protein matrix, is the lamellar layer. This is the layer you see when viewing the inside surface of the shell.
A large purple adductor muscle scar is a conspicuous feature of the inner surface of both valves (Fig 1). The scar is much closer to the posterior margin than the anterior and is ventral of the middle of the valve. The scar marks the sites of attachment of the powerful posterior adductor muscle whose action is to pull the two valves together and close the gape. It is antagonized by the hinge ligament. There is no anterior adductor muscle. Quenstedt’s muscle scar is a tiny indistinct spot just ventral to the hinge (Fig 1). Sometimes it is obvious, sometimes it cannot be found.
Find the hinge at the dorsal (narrow) end of the valve. That of the right valve includes a slight elevation, known as the buttress, of the shell margin. The buttress fits into a correspondingdepression in the hinge of the left valve (Fig 3). With magnification inspect the hinge region and note that other than the buttress, it is similar to the remainder of the valve margin. Oysters completely lack any hinge teeth but the irregular curvature of the edges of the valves prevents the adducted valves from shearing past each other. If possible, compare the hinge of your oyster with one from another bivalve such as the quahog, Mercenaria, which has well developed hinge teeth.
Figure 3. Cross section below the hinge. The buttress of the right valve fits into the depression of the left valve (from Galtsoff, 1964). Bivalve128a.gif
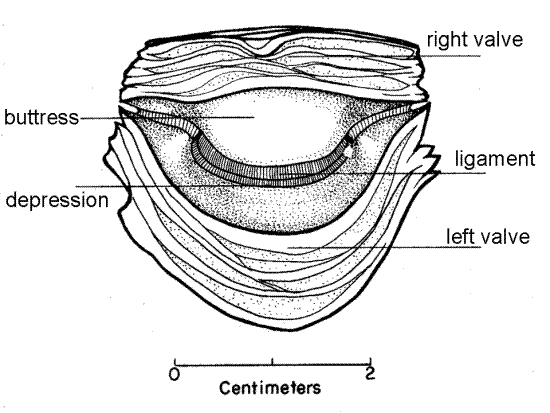
The two valves are held together by a dark, proteinaceous hinge ligament, usually greenish brown in life. The ligament lies between the buttress and depression of the two valves. It is located inside the hinge and is thus an inner ligament, or resilium. (In bivalves in general, the more common position of the ligament is outside the hinge where it is known as a tensilium, or outer ligament.) The ligament will necessarily have been torn when you broke the right valve away from the left but you should be able to find its remains in the hinge at the dorsal end of the valve. The ligament is composed of the protein conchiolin (as is the periostracum) and is elastic. It, along with the adductor muscle, holds the two valves together. Contraction of the adductor muscle adducts the valves and simultaneously compresses the elastic ligament. When the muscle relaxes, elastic recoil of the compressed ligament abducts the valves, opens the gape, and stretches the adductor muscle.
Figure 4. Cross section through the hinge (from Galtsoff, 1964). Bivalve131a.gif
>a. If shells of the quahog, Mercenaria mercenaria, are available, study them using the description at the OnLineLab Web page for Mercenaria . Compare the Mercenaria shell with that of Crassostrea.
Soft Anatomy
Look at the oyster you set aside in magnesium chloride. Support the left valve so the surface of the soft tissue is horizontal. Touch the oyster gently with a teasing needle. If it is relaxed, it will not respond when touched. External anatomy can be studied before complete relaxation occurs. Be careful that magnesium chloride does not spill on the stage of the microscope. In reproductive individuals the diffuse hypertrophied gonads will cover most of the visceral mass and mantle in a thick layer that will obscure the kidney which could otherwise be seen through the body wall.
Preview
Removal of the right valve exposed the right side of the oyster to view. Arrange the oyster in the dish with the right side up and the dorsal end away from you, as in Figure 5. The oyster body consists chiefly of a large visceral mass, two mantle skirts, a mantle cavity, a large adductor muscle, a pair of gills, and a pair of labial palps (Fig 5). There is no foot, no anterior adductor muscle, and no siphons. The head is so weakly developed as to be indistinguishable from the visceral mass.
The uppermost layer of tissue arrayed before you is the right mantle skirt. It may have been damaged a little when you removed the right valve. It will probably be thick and white with glycogen reserves and gametes in the gonad. Its border is marked by a dark brown or gray line. The adductor muscle can be seen penetrating the mantle skirt in the posterior ventral region of the valve. It is white and does not contrast strongly with the mantle so it may not be immediately apparent. The visceral mass, mostly covered by the right mantle skirt, occupies the space between the hinge and the adductor muscle. The open space under the right mantle skirt is the mantle cavity. If you lift the anterior edge of the right mantle skirt, the conspicuous gills will be revealed. The pericardial cavity and heart lie on the dorsal edge of the adductor muscle.
Adductor Muscle
The large adductor muscle can be seen in the posterior ventral region. Its fibers run transversely from valve to valve so you are seeing it now in cross section. It may appear a bit ragged since it was cut by the scalpel. The muscle may be easier to see on the right valve where the mantle has been removed.The function of the muscle is, of course, to pull the two valves tightly together, thus closing the gape and isolating the oyster from external threats. The muscle is divided into a large, pale grey, dorsal quick muscle and a smaller, bright white, ventral and posterior catch muscle whose fibers differ in structure and function (Fig 8). The quick muscle is composed of cross-striated fibers capable of rapid response but incapable of prolonged contraction. Catch muscle, on the other hand, is composed of smooth fibers that react more slowly but that can remain contracted for long periods.
Figure 5. An oyster viewed from the right side with the right valve removed but with the right mantle skirt still in place (from Galtsoff, 1964). Bivalve132a.gif
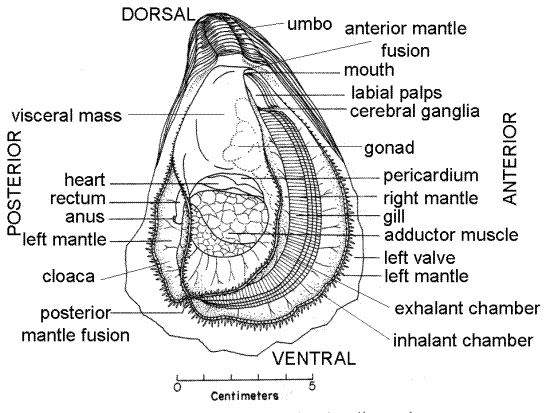
Visceral Mass
The visceral mass fills most of the space between the hinge and the adductor muscle (Fig 5). A large, thin fold of the dorsal body wall extends laterally from each side of the visceral mass. These are the right and left mantle skirts and together they enclose a large water space, the mantle cavity, ventral and anterior to the visceral mass. The dark greenish brown digestive ceca can be seen at the surface the visceral mass posterior to the gills. Sometimes the kidneys, which are paler yellowish brown, may also be visible through the body wall. A pair of large crescent-shaped gills protrude from the anterior and ventral border of the visceral mass. In reproductive individuals the gonads fill much of the space in the visceral mass between the stomach and digestive ceca.
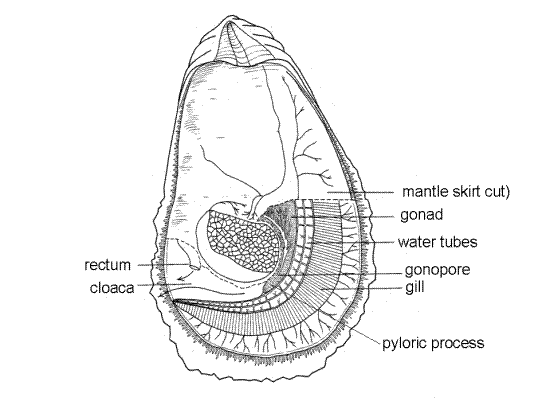
The rectum and anus are located in the cloaca, which is part of the exhalant chamber of the mantle cavity. Lift the right mantle skirt between the adductor muscle and the edge posterior edge of the valve. This will expose the cloaca (Fig 5). The rectum can be seen as a thick tube curving around the posterior border of the adductor muscle. It ends at the anus, whose border is flared outward.
The pointed, conical, ventral tip of the visceral mass extends on the anterior side of the adductor muscle beside the exhalant chamber. It is known as the pyloric process (Fig 11). The common openings of the kidneys and gonads are on the pyloric process. The heart is in the pericardial cavityon the dorsal margin of the adductor muscle.
Mantle and Mantle Cavity
As you now view the specimen the tissue closest to you is the right mantle skirt (Fig 5). It is penetrated by the adductor muscle. The skirt is thin and transparent and its pigmented border bears abundant sensory tentacles. The skirt is immediately inside the right valve and the valve was secreted the skirt. The edge of an identical left mantle skirt can be seen lying against the left valve (Fig 5, 7) but most of this skirt is now hidden by the visceral mass and gills. Blood vessels can be seen in the right mantle skirt. The two mantle skirts enclose a water space, the mantle cavity (Fig 6). In life the mantle cavity is filled with seawater and is divided into inhalant and exhalant chambers by the gills (Fig 5). The feeding and respiratory current enters the inhalant chamber and then passes through pores, or ostia, in the walls of the gills. Once through the ostia, the water is in the exhalant chambers above the right and left gills. These chambers coalesce in the vicinity of the anus to form the cloaca, which empties to the exterior.
Each mantle skirt is a fold of the dorsal body wall and arises from the dorsal part of the visceral mass. Dorsally the right and left skirts are joined to each other and to the visceral mass. Trace the edge of the right skirt dorsally along the anterior border of the valve until it joins the edge of the left skirt. This broad junction is the anterior fusion (Fig 5). Follow the edge of the right skirt ventrally and posteriorly until you find the delicate posterior fusion where it joins with the left mantle skirt (Fig 5). The posterior fusion of the mantle skirts marks the line of separation of the inhalant chamber of the mantle cavity from the cloaca, which is part of the exhalant chamber. Lift the right mantle skirt anterior to the posterior fusion to reveal the spacious inhalant chamber. Lift the right mantle skirt posterior to the posterior fusion to reveal the cloaca. The rectum and anus can be seen in the cloaca.
Continue following the right mantle skirt dorsally along the posterior margin until you find where it arises from the visceral mass. The origin of the mantle skirt anteriorly is hidden from view.
The edges of the mantle skirts are lobed to form three longitudinal ridges, or folds, which can be seen with magnification. With the dissecting microscope study the edge of the left mantle skirt and find the three folds. The outer fold secretes the shell, the middle fold is sensory and bears numerous tentacles, and the inner fold is muscular, but also has tentacles (Fig 12-91). The outer surface of the outer fold secretes the prismatic layer. The outer surface of the entire mantle skirt secretes the lamellar layer. The groove between the outer and middle folds is the periostracal groove from which the periostracum is secreted. Find a region of the left valve with the left mantle still intact and adhering to the valve. Study the mantle edge with magnification to find the outer fold and the periostracal groove. The outer fold, being the same color as the shell and covered by the middle fold, is difficult to see. Adjust the light (reduce it), lift the middle fold with a microneedle, and focus on the outer fold. Look for the thin, glistening, newly secreted sheet of periostracum emerging from the groove and extending over the outer surface of the left valve. New periostracum is difficult to demonstrate in oysters as it is thin and weak and tears free of the valve.
Gills
" Use fine scissors to remove the right mantle skirt by cutting along the line where the skirt joins the right gill for the entire distance between the anterior and posterior fusion points. Set the mantle aside. Two large crescent-shaped gills protrude from the anterior and ventral edge of the visceral mass into the mantle cavity (Fig 5). Each gill is known as a whole gill, orholobranch, and is composed of two half gills, or demibranchs, one of which is lateral and one medial. The four demibranchs give the impression that four gills are present but there are actually only two (Fig 5, 7).
The four conspicuously ridged demibranchs are stacked on top of each other in the inhalant chamber of the mantle cavity (Fig 5). Find the lateral and medial demibranchs of the right holobranch and the lateral and medial demibranchs of the left holobranch. In oysters there is no foot to divide the mantle cavity into right and left sides and separate the two gills.
Each demibranch is composed of long narrow gill filaments which cannot be seen without magnification and then only by careful observation and sharp focus. The conspicuous ridges oriented perpendicular to the long axis of the gill are plicae, or plications, and each is composed of numerous filaments.
Lift the lateral demibranch of the right gill so you can see the line along which it joins the medial demibranch. This line is the central axis of the gill (Fig 6) and is the attachment of the gill to the body. You can see the plicae arise from the central axis and then extend down into the mantle cavity to form one side of the demibranch. At the free edge of the demibranch the filaments are bent almost 180 ° so they extend up again to form the other side of the demibranch before they end at the body. The resulting two-sided sheet is the demibranch. Each side of a demibranch is known as a lamella and is composed of side-by-side plicae (made of filaments). The lamella joined to the central axis is the descending lamella (= descending limb) whereas the opposite side of the demibranch is the ascending lamella (Fig 6, 12-96). The spaces inside the demibranchs, between the lamellae, are water tubes, which are part of the exhalant chamber.
The free edge of each demibranch bears a conspicuous longitudinal food groove (Fig 6). You may need to use fine forceps to bend the free edge of the demibranch toward you to reveal the groove. Cilia in this groove generate a current that moves a mucous string with trapped food particles anteriorly to the labial palps for sorting.
The gill filaments of which the plicae are composed have lateral cilia between the filaments to generate the ventilating and feeding current and frontal cilia on the outer surface to move particles off the lamellae. As the ventilating current moves through the lamella from the inhalant to the exhalant side, particles too large for the ostia are caught on the surface of the lamella. Ciliary currents move them ventrally to the food groove along the free margin of the demibranch. The food groove extends dorsally to the labial palps to which it transports food.
Figure 6. A cross section made just ventral to the labial palps. (from Galtsoff, 1964). Bivalve134a.gif

>b. Place a little carmine-seawater or a sprinkle of finely divided carmine powder or chalk dust on the surface of the ascending lamella of the lateral demibranch and watch as it is transported anteriorly by the frontal cilia. Watch for particles to arrive at the food groove and then change directions 90 ° and move dorsally on the way to the mouth. <
>c. With the highest power of the dissecting microscope (40X) study the surface of the ascending lamella. The unrelentingly white tissue of most bivalves provides little contrast and makes it difficult to resolve objects such as gill filaments. Slip a small square of black paper under the demibranch to provide a contrasting background . Use fine forceps as necessary to manipulate the demibranch and tug gently on the plicae as necessary. Confirm that the plicae are composed of numerous gill filaments, which are much smaller than the plicae. They are almost too small to resolve at 40X. Adjacent filaments are held permanently together by tissue interfilamentar junctions that cannot be broken without tearing the tissue. The interfilamentar junctions are penetrated periodically by small pores, or ostia. It is through the ostia that the feeding current passes through the lamella on its way from the inhalant to the exhalant chamber. Between the plicae the ascending lamella is joined to the descending lamella on the other side of the demibranch by tissue interlamellar junctions (Fig 6, 12-98D). These junctions divide the demibranch into vertical water tubes that extend up into the exhalant chamber. The interior of each plica is a water tube. <
>d. Use fine scissors to remove a 3x3 mm square of the lateral demibranch including a section of the food groove. Make a wet mount with the square being careful that it is not folded on itself. Examine it with 400X of the compound microscope with the light carefully adjusted. Focus on the food groove and look for beating cilia in the groove. Find the parallel gill filaments, which also have cilia that may still be beating.
Find ostia between the filaments. <
" Use fine scissors to cut longitudinally along the line of fusion of the lateral demibranch with the body. Extend the incision for the entire length of the gill. This will open the right exhalant chamber and reveal the water tubes and interlamellar junctions in the interior of the demibranch. Trace the chamber ventrally and posteriorly to the cloaca just posterior to the adductor muscle. Find the anus and rectum on the circumference of the muscle if you have not already done so. Find the opening from the cloaca into the left exhalant chamber. It will be to the left of the visceral mass. Confirm that the cloaca is formed by the junction of the right and left exhalant chambers.
In Crassostrea, the dorsal end of the right exhalant chamber is expanded to form a large promyal chamber (Fig 6). There is no such modification of the left exhalant chamber nor is it present in Ostrea.
Heart and Pericardial Cavity
If it is still present, remove the right mantle skirt from the region immediately dorsal to the adductor muscle. This will reveal the thin walled pericardial cavity on the dorsal border of the muscle (Fig 5, 7). The wall of the cavity is the thin, translucent pericardium. With your finest scissors cut through the pericardium to open the cavity. The pericardial cavity is all that remains of the mollusc coelom. Inside it is the heart consisting of a ventricle and two atria. The ventricle is a large, white or gray, ovoid chamber in the posterodorsal quadrant of the cavity. The atria are smaller than the ventricle and are in the anteroventral region of the cavity. The atria drain oxygenated from the gills via the efferent branchial vessels and send it to the ventricle to be pumped to the various hemocoels via a system of arteries (Fig 7). The brownish right atrium is closer to you and the only one you can get a good look at without further dissection. Watch the ventricle carefully for a minute or so and you will probably see it contract, even if your specimen has been in magnesium chloride for an hour or two. If it does not contract give the heart a gentle squeeze with forceps and watch for a contraction in response.
The conspicuous posterior aorta exits the dorsoposterior corner of the ventricle and curves ventrally, still within the pericardium, to supply the adductor muscle (Fig 7). The less obvious, but larger, anterior aorta exits the same end of the ventricle dorsal to the posterior aorta. The anterior aorta disappears immediately into the pericardial wall but can be followed a short distance by cutting into the wall with fine scissors. It quickly gives off a large visceral aorta before itself extending anteriorly towards the head. The visceral aorta runs anteriorly and dorsally to the gonad and visceral mass. Notice that the rectum does not pass through the ventricle, or even enter the pericardium as it does in many bivalves.
>e. If you wish to observe the heartbeat, replace the magnesium chloride with seawater and let the oyster sit for about 15-30 minutes. Stimulate the ventricle with a gentle pinch and watch the resulting contractions. Eventually (about 30 minutes) the heart will resume beating spontaneously. When finished with your observations, replace the water with magnesium chloride. <
Figure 7. The arterial system of Crassostrea (from Galtsoff, 1964). a = artery. Bivalve140a.gif
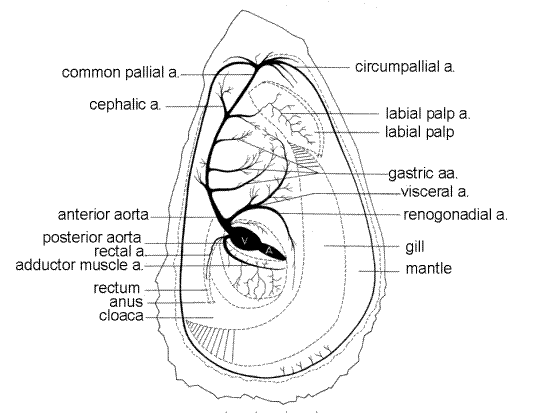
The accessory heart is a conspicuous Y-shaped tube on the left wall of the cloaca ventral to the adductor muscle. Handling it with dissecting tools may cause it to collapse, in which case it cannot be seen. The accessory heart is muscular and beats, although not as rapidly as the heart itself. Its function apparently is to assist in the movement of oxygenated blood from the mantle to the efferent vessels draining the gills.
Labial Palps
" Use the scalpel to separate the left end of the adductor muscle from the left valve. Carefully remove the oyster from the left valve and transfer it to a small dissecting pan of isotonic magnesium chloride. Place the right side uppermost, facing you, as it was while in the shell. Examine your oyster and find landmarks to orient yourself so you still know dorsal, ventral, anterior, posterior, right, and left.
The two labial palps are located in the mantle cavity at the anterior end of the gills. Each gill is associated with a labial palp. Like the gills, the labial palps are bilobed giving the appearance of two palps on each side (Fig 5, 12-113). Each lobe, known as a lamella, is flat and leaflike. The two lamellae of each palp face each other. The palps are ciliated and are used to transfer food from the gills to the mouth.
Find the right labial palp at the dorsal end of the right gill. Find its two lamellae. The one closest to you is the lateral lamella of the right palp and the other is the medial lamella. Each demibranch is associated with a palpal lamella; the lateral demibranch with the lateral lamella and the medial demibranch with the medial lamella.
Determine that the lateral surface of the lateral lamella is smooth as is the medial surface of the medial lamella. In contrast, the medial surface of the lateral lamella and the lateral surface of the medial lamella bear conspicuous ridges and grooves. In other words, the surfaces of the lamellae that face each other are ridged and grooved and the outside surfaces are not. The ridges and grooves are ciliated and are presumed to function as a sorting field to separate food and mineral particles although that may not be the case for Crassostrea.
Observe the relationship between the lamellae of the right palp and the demibranchs of the right gill. The food grooves on the edges of the demibranchs end at the crease at the junction of the right and left lamellae of the palp. This crease is the ciliated oral groove and it leads to the mouth. Its cilia generate a current in the direction of the mouth. The food string of mucus and food particles from the food groove passes along the oral groove into the mouth.
Look deep under the right palp to find the left labial palp. Each lamella of the right palp, medial and lateral, is connected physically with its counterpart on the left palp. Trace the connections from right to left from right lateral lamella to left lateral lamella and from right medial lamella to left medial lamella. These transverse connections form a pair of lips above and below the mouth (Fig 12-100). Thus the right and left lateral palps are connected with each other by the dorsal lip above the mouth and the right and left medial palps are connected by the ventral lipbelow the mouth.
Grasp the oyster with thumb and forefinger of one hand and hold it above the dissecting pan with the dorsal end up, so you can focus on the head. Use a blunt probe to explore the region at the dorsal end of the visceral mass and locate again the upper and lower lips. With the probe push the two lips apart and find the mouth. It is a fairly large horizontal slit. It opens into a short esophagus which itself opens into the stomach. If you press the visceral mass gently with your fingers, stomach contents may be regurgitated out of the mouth, thus confirming its presence. The stomach contents will probably be brownish green. You should squirt this material out of your field of vision with jets of magnesium chloride from a plastic Pasteur pipet.
>f. Reflect the lateral right lamella to the side and arrange the oyster so the ridged surface of the medial lamella of the right palp is horizontal. Place a little carmine-seawater or a sprinkle of carmine powder on the surface of the palp and watch as the particles are transported by the cilia. <
Internal Anatomy
Digestive System
The digestive system consists of mouth, esophagus, stomach, midgut and style sac, descending intestine, ascending intestine, rectum, and anus.
" Hold the oyster with thumb and forefinger of one hand so you can focus on the region of the mouth and lips. Insert one blade of your fine scissors into the right side of the mouth and cut along the right side of the visceral mass back to the pericardial cavity. The incision should be deep enough to open the lumen of the gut, which in this region will be the stomach. Use a pipet to blow away debris and cloudy fluids. The incision will open the short esophagus and the large, irregularly lobed stomach (Fig 8). Extracellular digestion, using enzymes released from a rotating crystalline style, occurs in the stomach.
Figure 8. The digestive system of an oyster as revealed by latex injection (from Galtsoff, 1964). Bivalve139a.gif
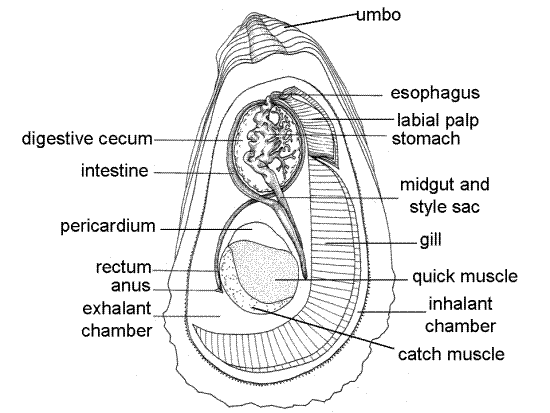
The crystalline style is a long, soft, gelatinous, protein rod whose distal end extends into the stomach lumen (Fig 12-102, 12-89B). It is probably homologous to the protostyle of the ancestral molluscs. The style is composed of digestive enzymes that are released into the stomach lumen as the style rotates. The style originates in a deep diverticulum of the ventral stomach wall known as the style say. The glandular epithelium of the sac secretes the enzymes and it s cilia cause the style to rotate. The free end of the style protrudes from the sac into the stomach lumen. The style is reabsorbed when the oyster has not fed recently. The style will probably not be present in your specimen.
The stomach is embedded in, and surrounded by, the greenish-brown digestive ceca (Fig 8, 12-89B). The ceca are diverticula of the stomach and are the site of enzyme synthesis, intracellular digestion, and absorption. Part of the digestive cecum is usually visible on the surface of the visceral mass, even in reproductive individuals. On the surface the digestive cecum appears as an irregular, dark greenish-black spot. The kidney, on the other hand, when visible on the surface is a pale yellowish brown.
" Trace the remainder of the gut as far as you can. This is best accomplished by using fine scissors to open its lumen as you did the esophagus and stomach. Use the Pasteur pipet freely to wash away debris that interferes with your view of the lumen. Tracing the gut is destructive of the remaining soft anatomy.
Figure 9. Cross section of the excretory system of Crassostrea virginica (from Galtsoff, 1964). Bivalve142a.gif
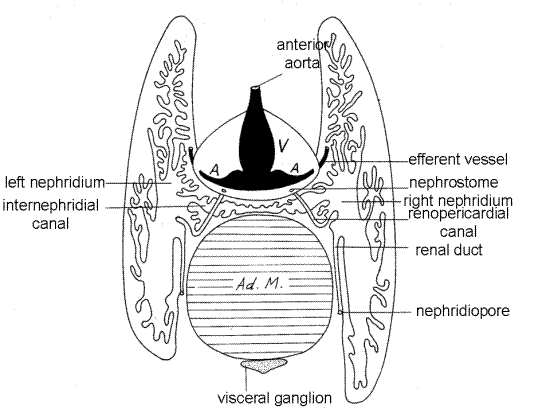
Locate the opening into the combined midgut and style sac at the ventral end of the stomach. Use the scissors to open the midgut for its entire length. It extends ventrally to the level of the adductor muscle. The style sac and midgut are coalesced side by side and share a common lumen partially partitioned by a pair of opposing ridges (Fig 12-103A). In freshly caught, recently fed individuals a crystalline style may be present in the style sac. Together the two extend ventrally a position near the pyloric process (Fig 11) on the anterior edge of the adductor muscle.
The style sac ends here, near the adductor muscle, but the gut continues on as the intestine. Posterior to the end of the style sac the midgut becomes the descending intestine which reverses direction and heads dorsally again, back toward the stomach. It reaches the posterior edge of the digestive ceca, loops dorsally then anteriorly around the stomach and ceca. Having completed this loop the intestine turns ventrally again, now as the ascending intestine, and extends to the level of the pericardium where it becomes the rectum before ending at the anus in the cloaca.
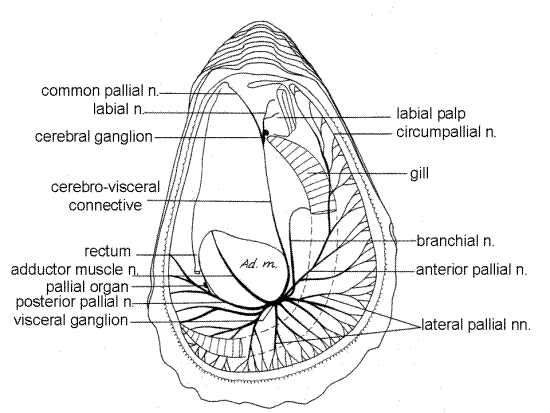
Figure 10. The nervous system of Crassostrea viewed from the right (from Galtsoff, 1964). n = nerve, Ad. M = adductor muscle. Bivalve143a.gif
Trace the gut as far downstream as you can by opening the gut lumen with fine scissors. When you can follow it no farther, redirect your attention to the anus and rectum. Insert one blade of the scissors in the anus and open the lumen of the rectum. Cut as far dorsally as you can, opening the ascending intestine as you go. The ascending intestine will eventually reach the level of the digestive ceca and pass anteriorly to the left of the cecum and stomach. It will cross the descending intestine, which you should watch for (Fig 8). After crossing the descending intestine ventral to the digestive cecum it passes along the anterior border of the cecum until it joins the descending intestine dorsal to the cecum (Fig 8).
Excretory System
Although parts of the excretory system can be seen on the surface of the visceral mass, especially in the vicinity of the pericardium, this system will not be studied in this exercise. Exposed parts of the nephridia are pale yellowish brown.
The excretory system consists of a pair of large metanephridia, or kidneys extending from nephrostomes in the wall of the pericardium to nephridiopores in the atrium (shared with the gonad) leading to the cloaca (Fig 9). The nephrostome opens into the renopericardial duct, which connects with the lumen of the nephridium. The nephridia lie immediately below the surface of the visceral mass where they are visible without dissection in lean oysters without well-developed gonads. In fat oysters with hypertrophied gonads the kidneys will be hidden deep under a thick layer of gonad.
Figure 11. Crassostrea viewed from the right with part of the right mantle skirt removed to reveal the pyloric process and gonad (from Galtsoff, 1964). Bivalve144a.gif
A large, thick-walled metanephridium lies on each side of the visceral mass. The right and left metanephridia are connected across the midline by an internephridial canal between the pericardial cavity and the adductor muscle to form a H-shaped complex (Fig 9, 12-118). One upright of the H lies to the right of the pericardium and adductor muscle and the other to the left. The nephridia extend dorspoventrally from anterior to the heart to posterior to the adductor muscle.
Nervous System
The nervous system, although simple in Crassostrea, is difficult to demonstrate in gross dissection and will not be studied in this exercise (Fig 10, 12-119). It includes a pair of cerebral ganglia, with one ganglion on each side of the esophagus. The two are connected by the cerebral commissure that arches up over the top of the esophagus. The coalesced visceral ganglia are at the ventral end of the visceral mass on the anteroventral border of the adductor muscle. The cerebro-visceral connectives run from the cerebral ganglia to the visceral. There being no foot, there are likewise no pedal ganglia. Pleural ganglia are also absent.
In the absence of cephalic sense organs the cerebral ganglia are weakly developed and small. The visceral ganglia are much larger than the cerebral and nerves from it innervate the mantle, gills, and adductor muscle. In addition to their usual autonomic functions, the visceral ganglia also receive sensory input from the sensory tentacles of the mantle. The tentacles are photoreceptive, mechanoreceptive, and chemoreceptive.
The large white visceral ganglion can be revealed by opening the exhalant chamber and cloaca and looking between the pyloric process and the adductor muscle. The small cerebral ganglia are beside the esophagus at the bases of the labial palps. They are near the surface of the visceral mass but are usually obscured by gonad.
Reproductive System
Crassostrea is gonochoric and at any given time is either male or female (although Ostrea is simultaneously hermaphroditic). In reproductive individuals the gonad is large and occupies most of the space in the visceral mass between the digestive ceca and the surface. It may be several millimeters thick but is an indistinct organ without definite walls. The two gonads of the ancestral bivalves are coalesced to form a single organ with two gonoducts, right and left. Gametes exit the gonad via the gonoduct, which joins the kidney tubule to form a common chamber, the atrium, which opens into the cloaca via a pore on the pyloric process (Fig 11, 12-120B).
>e. Cut a small piece of gonad from the visceral mass beside the digestive cecum and make a wetmount using seawater. Examine the slide with the compound microscope looking for gametes. Eggs are large and irregularly shaped with conspicuous pronuclei, sperm are tiny and monoflagellated. Be sure you see oysters and gamete wetmounts of both sexes. <
References
Galtsoff PS. 1964. The American oyster, Crassostrea virginica Gmelin. US Fish. Wildlife Ser, Fish. Bull 64:1-480.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Supplies
Dissecting microscope
American oysters, Crassostrea virginica, 10-12 cm in length
Empty complete shell (If available)
Isotonic magnesium chloride
Cotton work gloves
Screw driver or oyster knife
Dissecting set with iridectomy scissors and microdissecting forceps
Carmine particles
12 cm culture dish
Canned smoked oysters
black construction paper
Plastic Pasteur pipet