Invertebrate Anatomy OnLine
Botryllus ©
Compound Tunicate
5jul2006
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises, a glossary, and chapters on supplies and laboratory techniques are also available at this site. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
ChordataP, Tunicata sP, Ascidiacea C, Pleurogona O, Stolidobranchia sO, Styelidae F (Fig 27-12, 29-32)
Chordata P
Chordata is characterized by a suite of apomorphies including a dorsal hollow nerve cord, notochord, pharyngeal gill slits, and a post anal tail (Fig 29-1). The ancestor was a fishlike deuterostome that swam using alternating contractions of right and left longitudinal axial muscles to create undulations of the body. The flexible, incompressible notochord prevented these contractions from compressing the body while allowing lateral deflection. The chordate central nervous system is a hollow, median, longitudinal nerve cord formed in the embryo by an invagination of surface ectoderm whose original function was probably sensory reception. Paired pharyngeal gill slits connect the lumen of the pharynx with the exterior and originally functioned in suspension feeding with respiration being added later. A muscular tail posterior to the anus is, although commonplace in chordates, an unusual feature not found in other taxa. It is an extension of the axial musculature and is the chief locomotory organ. An additional apomorphy is the endostyle, a region of pharyngeal endoderm, that secretes iodated compounds, either mucus or hormones.
Tunicata (= Urochordata) sP
Tunicates are highly derived and less like the ancestral chordates than are cephalochordates or vertebrates. At some time in the life cycle all possess a notochord, dorsal hollow nerve cord, pharyngeal gill slits, postanal tail, and endostyle but only the gill slits and endostyle are present in adults. Tunicates use the pharyngeal gill slits for suspension feeding. The larva is much more chordate-like than the adult and resembles a tadpole or fish, has all the chordate apomorphies, and is known as the tadpole larva. Metanephridia are absent and coelom is reduced to a pericardial cavity and gonads. As in cephalochordates the gut is dominated by an enormous pharynx surrounded by a water-filled atrium but unlike cephalochordates, it is U-shaped with the mouth and anus anterior. Tunicates may be benthic or planktonic and solitary or colonial. All are marine.
Tunicata is traditionally divided into Ascidiacea (the benthic sea squirts in three taxa; Aplousobranchia, Phlebobranchia, and Stolidobranchia), Thaliacea (the pelagic salps), and Appendicularia (the pelagic larvaceans). Recent molecular evidence and reevaluation of morphological evidence, however, suggests that Ascidiacea is paraphyletic and Tunicata should be reorganized into three different higher taxa (Fig 29-32). In this reorganization Stolidobranchia would be one higher taxon. Phlebobranchia plus Thaliacea would be the second taxon. Aplousobranchia plus Appendicularia is the final tunicate taxon. For now, however, the traditional classification will be followed.
Ascidiacea C
Ascidiacea is usually taken as representative of Tunicata, at least for the purposes of introductory laboratory exercises. Ascidians, or sea squirts, are sessile filter feeders that, as adults, bear little resemblance to their chordate relatives. Ascidians have a living, external, cellular exoskeleton, or tunic, underlain by epidermis. The tunic resembles connective tissue, except it isoutside the epidermis, and consists of cells, a secreted extracellular matrix, and ground substance. Much of it is a cellulose-like polysaccharide. In many ascidians blood vessels cross the epidermis to enter the tunic, a feature found in no other animal.
The gut is U-shaped and both openings are anterior, with the anus dorsal to the mouth. The gut is dominated by an enormous pharynx whose wall is perforated by numerous tiny gill slits. The pharynx is surrounded by a water-filled atrium into which the gill slits open and which itself opens to the sea. It is both respiratory organ and filter-feeding device. Water and food particles enter the pharynx and the water passes through the gill slits to the atrium and then out the siphon. Food, entangled in mucus secreted by the endostyle, remains in the gut and passes posteriorly to be digested.
The hemal system includes a heart, vessels, and blood spaces in the connective tissue. The heart is enclosed in a pericardial cavity derived from the ancestral coelom. The pattern of blood flow resembles that of the cephalochordates and early vertebrates except that the heart reverses direction periodically and the blood thus flows in both directions through the system. Ascidians have no structure recognizable as a kidney.
Ascidians are simultaneous hermaphrodites and the gonoducts open into the atrium. Some ascidians are solitary and may be relatively large. Others are colonial with tiny individual zooids in a common tunic.
Stolidobranchia sO
Phlebobranchia, Aplousobranchia, and Stolidobranchia are the three higher ascidian taxa. In stolidobranchs the gonads are on the inside surface of the body wall beside the pharynx and not in the gut loop. No epicardium is present but Molgulidae has renal sacs, which are derived from the epicardium. The neural gland is dorsal to the cerebral ganglion. The stolidobranch pharyngeal lining is strongly pleated and has transverse and longitudinal blood vessels. Molgulidae has spiral gill slits. Stolidobranchs can be solitary or colonial. The plane of the tadpole tail is vertical.
Laboratory Specimens
The elegantly pigmented colonial tunicate Botryllus can be collected from firm substrata in shallow water on most coasts. Botryllus schlosseri or a similar species can be found on most European and North American coasts. This is a common and widely distributed species whose cosmopolitan distribution is probably the result of transport on ship’s bottoms. Colonies form thin encrustations on a variety of substrata, both living and inert. Pigmentation patterns are highly variable but are often striking. Botryllus is a good choice for the study of colony morphology of a compound ascidian, but study of its individual zooids should be reserved for an advanced course. The small size (<4 mm) and heavy pigmentation of the zooids makes their study difficult.
Colony
Examine a living Botryllus colony in a culture dish of seawater using a dissecting microscope. The colony is composed of clusters of less than 20 individuals, or zooids, embedded in a common tunic (Fig 1, 29-16A,B). Each zooid is a complete sea squirt with its own buccal and atrial siphons, gut, gonads, heart, and nervous system but with its tunic, cloaca, and hemal system shared with the other zooids in its cluster. The clusters, known as systems, are circular or star-shaped (stellate) in which the buccal siphons are peripheral and the atrial siphons at the center. The atrial siphon of each zooid empties into a common central cloaca with a single opening, the common cloacal siphon, to the exterior. The buccal siphon of each zooid is independent of the other zooids and is peripheral in the system.
Each zooid is enclosed in its own mantle consisting of epidermis, connective tissue and muscles, but shares the common tunic (= test) with other members of the colony. The mantle may be heavily invested with pigment granules to the extent that the internal structure of the zooid is obscured and difficult to observe, even when the zooid is removed from the tunic.
Each zooid has an intrazooidal hemal system typical of stolidobranchs but, in addition, the colony has an interzooidal system consisting of vascular stolons extending throughout the common tunic to connect all the zooids. The interzooidal system is pressurized by slow contractions of vascular ampullae concentrated around the periphery of the colony (Fig 1). The colony grows rapidly by asexual budding of the zooids.
Zooids
Removal and study of individual zooids is not recommended for introductory classes. This exercise assumes prior knowledge of the general morphology of a solitary ascidian. Use fine forceps, minuten nadeln, and iridectomy scissors to extract several zooids from the colony. Make wetmounts with them for examination with the compound microscope. Examine the wetmouts with the compound microscope learning as much as you can from each one.
Figure 1. Botryllus sp.
from Coos Bay, Oregon. A, A small colony showing several stellate
systems. B, A single system of eight zooids. Uro96L.gif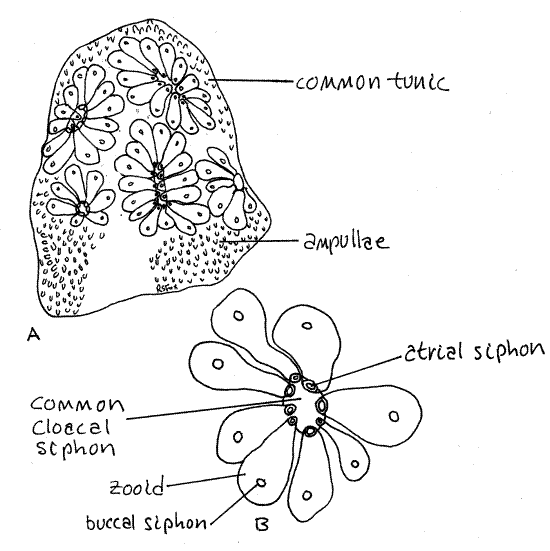
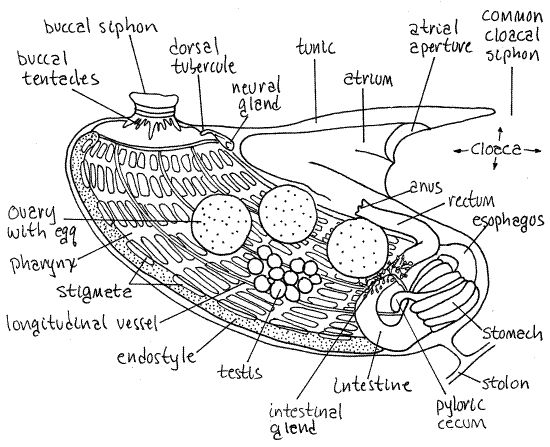
Botryllus zooids are miniature replicas of solitary stolidobranch sea squirts (Fig 2, 29-16B). The body consists solely of the thorax with no abdomen or post abdomen. The digestive system consists of the buccal siphon, buccal cavity, pharynx, esophagus, stomach, intestine, rectum, and anus. The buccal cavity has a ring of 16 buccal tentacles. The pharynx has a ventral endostyle, dorsal lamina, longitudinal blood vessels, and about nine rows of rectangular stigmata. The pleats characteristic of the walls of stolidobranch pharynges are reduced to three longitudinal bars over the longitudinal vessels on each side. The wall of the stomach has about 10 longitudinal glandular folds, one of which is lengthened to form a pyloric cecum. The anus opens into an atrium that is proportionately larger than that of solitary ascidians. A ventral heart is present between the endostyle and stomach. The hermaphroditic gonad consists of one testis and a 1-3 ovaries on each side. The testes are grape-like clusters of lobes and each testis empties into the atrium by a short vas deferens. Each ovary contains a single large spherical egg and is connected with the atrium by its own short oviduct. A dorsal tubercle, neural gland, and cerebral ganglion are present on the dorsal midline above the junction of the buccal cavity and pharynx.
Figure 2 The left side of a zooid of Botryllus schlosseri. The center of the system is to the right. Redrawn from van Name (1945). Uro95L.gif
References
Alder J, Hancock A. 1907. The British Tunicata, vol II. Ray Society, London. 164p, 50pl.
Barrington , E.J.W. 1965. The Biology of Hemichordata and Protochordata . Oliver and Boyd, Edinburgh. 176p.
Berrill NK. 1950. The Tunicata, with an account of the British species. Ray Society, London. 354p.
Burighel P, Cloney RA. 1997. Urochordata: Ascidiacea, pp 221-347 in Harrison FW, Ruppert
EE (eds.) Microscopic anatomy of invertebrates. Wiley-Liss, New York. 537pp.
Kleinholz LH. 1950. Molgula manhattensis. in F. A. Brown, Selected Invertebrate Types. Wiley, New York. 597p.
Parker TJ, Haswell WA. 1921. A Text-book of Zoology, vol II. MacMillan. London. 714p.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Van Name WG. 1945. The North and South American ascidians. Bull. American Mus. Nat. Hist. 84:1-476, 31 pls.
Supplies
Dissecting microscope
Compound microscope
8-m culture dish
Seawater
Living Botryllus colony
Slides, coverslips
Dissecting set with microdissecting tools