Invertebrate Anatomy OnLine
Ascaris suum ©
Pig Roundworm
5jul2006
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises, a glossary, and chapters on supplies and laboratory techniques are also available at this site. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
CycloneuraliaSP, Introverta, Nematoida, Nematoda P, Secernentea C, Rhabditida O, Ascaridida, Ascaridina, Ascaridoidea SF, Ascaridae F, (Fig 22-35, 9-26)
Cycloneuralia SP
Cycloneuralia includes Gastrotricha, Nematoda, Nematomorpha, Priapulida, Kinorhyncha, and Loricifera. Most have a secreted cuticle and lack locomotory cilia so that locomotion is accomplished with muscles. The brain is a circular band around the anterior gut composed of forebrain, midbrain, and hindbrain (Fig 22-2A). The pharynx is radially symmetrical (Fig 22-15). In most the body is compact without a body cavity but in large species there may be a spacious hemocoel. Eutely, with about 1000 cells, is common.
Nematoda
The 20,000 known nematode species inhabit terrestrial, marine, and freshwater environments and are found in almost all moist habitats. The taxon includes numerous plant and animal parasites, many of which are of medical or agricultural importance, but most are free-living (non-parasitic). Most nematodes, or roundworms, are long, slender, almost featureless externally, tapered at both ends, and round in cross section (Fig 22-7A). The body cavity, if present, is a hemocoel derived from the blastocoel.
The body is covered with a thick extracellular cuticle secreted by a cellular or syncytial epidermis that is molted during juvenile development (Fig 22-11, 22-9). The epidermal nuclei are sunken below the epithelial layer into four longitudinal epidermal cords that extend the length of the animal. The body wall has well-developed longitudinal but no circular muscles.
The gut is complete with terminal anterior mouth and subterminal posterior anus. It comprises ectodermal foregut and hindgut and an endodermal midgut. The body cavity, or hemocoel, is not lined with mesothelium and there is no muscle, connective tissue, or other mesodermal derivative associated with the midgut.
The nervous system is a ganglionated circumenteric ring, hence the name “cycloneuralia”, with several longitudinal nerve cords, the most important of which is the ganglionated, double, ventral cord (Fig 22-11A). The nerve cords are located in the longitudinal epidermal cords, along with the epidermal nuclei. Cytoplasmic innervation processes from the longitudinal muscles extend to the longitudinal nerve cords and serve the function of motor neurons, which are absent. Sensory equipment may include unique chemosensory amphids and sensory bristles around the mouth.
Most nematodes lack cilia or flagella, even in the sperm. There are, however, ciliary derivatives in the amphids (Fig 22-8B) and cilia are present in the gut epithelium of some nematodes (Fig 22-16A). Roundworms are ammonotelic and nitrogen excretion is mostly by diffusion across the body wall. Osmoregulation is accomplished by an excretory canal system in some and perhaps by excretory glands. No nephridia are present.
Nematodes are typically gonochoric and fertilization is internal with copulation. Sexual dimorphism is common. Nematode sperm have no flagella and probably employ amoeboid locomotion. Nematode development features a phenomenon known as chromosome diminution in which much of the chromosome material of presumptive somatic cells degenerates and is lost (Fig 22-20). Germ cells, however, retain the full complement of genetic material. Development is direct and includes four juvenile and one adult instar separated from each other by molts. Most nematodes are small (<3 mm) and free-living but some of the parasitic species, such as Ascaris, may reach 50 cm.
Secernentea C
Once known as Phasmidia, Secernentea includes terrestrial nematodes and many important parasites. Free-living, non-parasitic species, such as Cephalobus, are usually soil dwellers. Phasmids are present and the amphids are porelike. Excretory canals, and sometimes excretory glands, are present (Fig 22-17). Epidermal cells can be mono- or multinucleated.
Laboratory Specimens
Ascaridae includes the largest nematodes and several members of the family are large enough to be dissected in invertebrate zoology laboratories. Ascaris suum, the pork roundworm, is a large, cosmopolitan species reaching lengths up to 50 cm (Fig 22-21). It is convenient for laboratory studies of nematode anatomy because of its large size and availability. An almost identical species, A. lumbricoides, occurs in humans, sheep, cattle, apes, and squirrels. The two were long thought to be the same species and until recently both went by the name A. lumbricoides. Preserved specimens are available from biological supply companies. Commercially supplied specimens are usually labeled A. lumbricoides but may be either species. Cross section slides are also available commercially.
External Anatomy
Place a preserved adult Ascaris in a long narrow dissecting pan of tapwater. While not essential, the dissection is best conducted with a dissecting microscope. Preserved worms are delicate and should be handled carefully.
Look at the surface of the worm with the dissecting microscope and note that it is firm and resists deformation. It is covered with a thick proteinaceous cuticle which plays an important role, in the absence of circular muscles, in containing the high hydrostatic pressure of the hemocoel. Look for the characteristic ornamentation of the cuticle, which in this species consists of fine circumferential ridges. The cuticle of preserved specimens is fragile and rough handling will cause it to break or peel away.
Figure 1. View of the left side of a male Ascaris. Nematode54L.gif
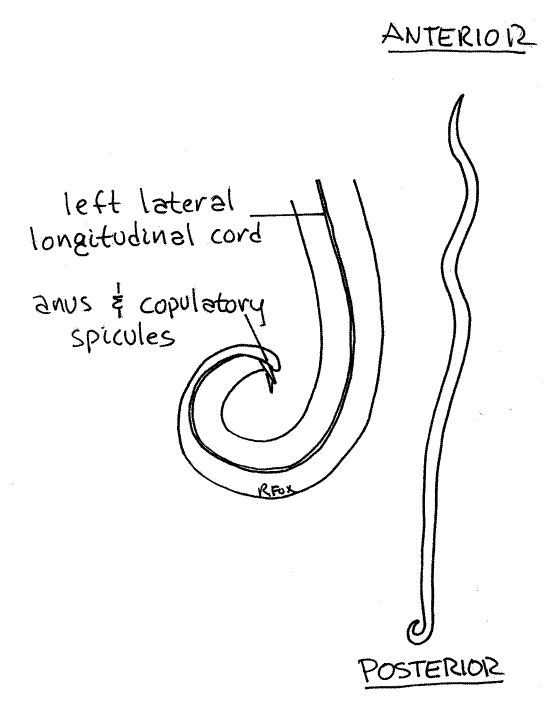
Determine the sex of your specimen. Females reach larger sizes than males. The posterior end of males is curved ventrally and looks like a shepherd’s crook (Fig 1). Two tinycopulatory spicules may be visible protruding from the anus on the inside curve of the crook. The posterior end of females is not noticeably curved.
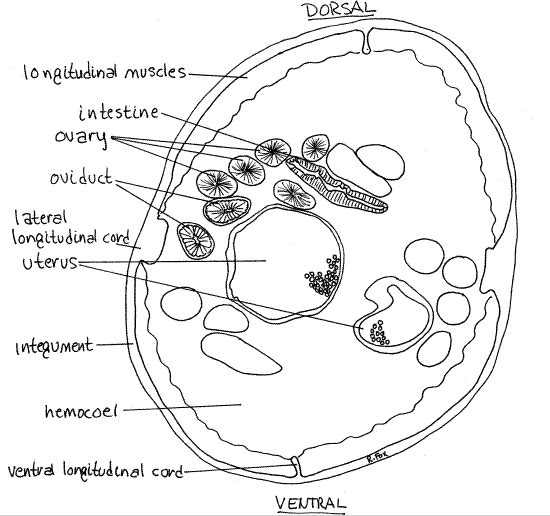
Figure 2. Dorsal dissection of a female Ascaris. The right side of the reproductive system is omitted and the left side has been moved to the left to expose the gut. The reproductive system has been simplified and untangled for clarity. Nematode55La.gif
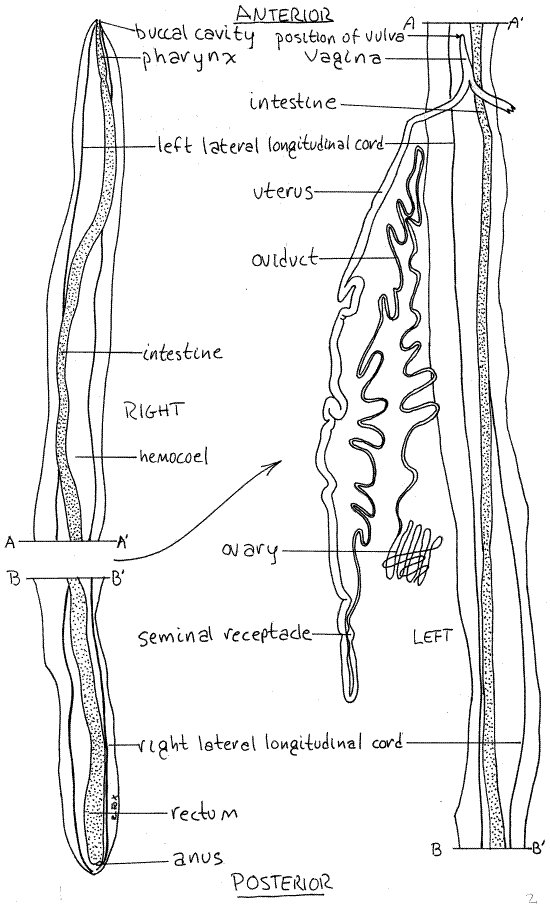
Distinguish between the anterior and posterior ends. The curled posterior end of males makes this easy for that sex but females are straight with similar anterior and posterior ends. In both sexes, however, the mouth is terminal at the anterior end but the posterior end has no terminal opening. Viewed head-on with the help of a hand lens, the mouth can be seen to be surrounded by three small lips. (It is easier to view the mouth with a hand lens than with the dissecting microscope because of the problem of orienting the long worm vertically on the stage.)
The three lips of Ascaris are formed by fusion of the six lips of the ancestral nematodes (Fig 22-8A). One of the lips is dorsomedian in position whereas the other two are ventrolateral. The arrangement of the mouth and its lips is radially symmetrical. The subterminal anus of both sexes is located slightly anterior to the posterior tip of the worm (Fig 2). It is a transverse ventral slit and is the best landmark for recognizing the ventral surface. Its position just behind the tip on the ventral surface is referred to as subterminal. (The mouth, which is at the extreme tip of its end, is terminal.)
The female gonopore, known as the vulva, is located on the midventral line about 1/3 of the animal’s length posterior to the mouth. It is a small pore best found with magnification. The female reproductive system opens to the exterior independently of the gut and there is no cloaca in this sex. The male reproductive system does not have its own external gonopore and sperm exit the animal via the anus (= vent). Two protrusible copulatory spicules, which are part of the male copulatory apparatus, may extend from the anus in some specimens.
Knowing ventral, anterior, and posterior, you can now find dorsal, right, and left. Find the plane of symmetry and the dorsal midline.
The four longitudinal cords in the body wall are visible from the exterior as thin, pale stripes Fig 1, 2, 3, 4, 5, 22-10B,C, 22-11). These are the dorsal, ventral, and two lateral longitudinal epidermal cords. They are faint, but discernable with good light, and are concentrations of the epidermal nuclei. The two lateral cords are easiest to see. A tiny pore belonging to the excretory canal system is located immediately posterior to the mouth on the ventral midline but is usually not visible.
Internal Anatomy
" Position a worm in a dissecting pan of water with its ventral side down. Males must be rotated a little to accommodate the curl of the tail.
Hold the worm gently with thumb and forefinger of one hand and use a # 1 insect pin to scrape a longitudinal middorsal incision through the cuticle and longitudinal muscles of the body wall in the anterior third of the body. Do your best to keep the incision on the dorsal midline even though there are not many landmarks to guide you. The two lateral epidermal cords, which are revealed by your incision are large and conspicuous longitudinal ridges on the inside of the body wall that can be used for orientation. You should make the dorsal incision so a lateral epidermal cord is equidistant from either side of it. You will not be able to see the cords at first and must wait until the incision is long enough to spread the walls apart. In the anterior end of the body there are few organs to damage but you should be careful nevertheless. You can develop your skill with the insect pin in this region where the possibility of damage is lessened.
The cuticle tends to resist the cutting motion of the pin so that it separates unevenly but several scrapes in the same place will penetrate it. The longitudinal muscles inside the cuticle, on the other hand, help guide the pin in the correct direction and there is no connective tissue or circular muscles to impede the passage of the pin.
Extend the incision anteriorly to the mouth. Deflect the cut edges of the body wall and pin them to the wax using # 1 insect pins inserted at 45 ° angles. Be careful as you pin the body wall aside. The internal organs, especially the gut, are very delicate and break easily. Further, the gut often adheres to the body wall and is easily pulled past its breaking point by movement of the body wall.
When you have opened and pinned the anterior third of the worm, extend the incision posteriorly to the end of the body, deflecting and pinning the walls. Opening the middle region of the worm is a bit more difficult because it is packed with the reproductive system (Fig 2).
Body Wall
The body wall will be studied in more detail later using cross section slides (Fig 3, 4, 5, 22-11) but some of its features are visible in gross dissection. The heavy, transparent cuticle is its outermost layer. Immediately inside the cuticle is the inconspicuous, thin epidermis. Inside the epidermis is a thick, white sheath of longitudinal muscles composed of a single layer of cells which protrude into the hemocoel (Fig 4).
The hemocoel, or body cavity, is filled with fluid under exceptionally high pressure (higher than that of any other animal) and is a hydrostatic skeleton. Virtually all other organ systems are affected by this pressure and must be able to function under its influence. The pressure maintains the body shape and acts as a hydrostatic skeleton against which the body wall muscles act to accomplish locomotion.
The nuclei of the epidermal cells are concentrated in the four longitudinal epidermal cords (Fig 1,2,3,4,5, 22-11A). The two lateral epidermal cords are large and conspicuous and protrude into the hemocoel. The dorsal and ventral cords are much less evident and the dorsal cord is usually destroyed by the middorsal incision. You must push the surrounding muscle cells aside to see the ventral and dorsal cords. Each epidermal cord includes at least one longitudinal nerve cord and an excretory canal is present in each lateral cord. The epidermal cords divide the somatic musculature into dorsal and ventral fields and make convenient landmarks.
Digestive System
The gut is a long, straight tube running from mouth to anus (Fig 2, 22-10). It is composed of an anterior, ectodermal foregut, endodermal midgut, and ectodermal hindgut.
Relocate the terminal mouth. The foregut, or stomodeum, comprises the buccal cavity and pharynx, which, consistent with their ectodermal origins, are lined with cuticle. The mouth opens into the small, inconspicuous, thin-walled buccal cavity (Fig 2, 22-13A). Immediately posterior to the buccal cavity is the longer, thicker-walled pharynx whose heavily muscularized walls are used to suck food into the gut in opposition to the high hydrostatic pressure of the hemocoel (Fig 2, 22-14B). The posterior end of the pharynx is swollen slightly and the pharyngeal lumen is triangular in cross section as is that of other cycloneuralians (Fig 22-15).
Look at a commercially prepared slide of a cross section made through the pharynx (Fig 22-15). Note the thick muscular pharyngeal walls and the triradiate lumen with its cuticular lining. When filled with food, the lumen expands and becomes circular.
The midgut, or intestine, begins immediately posterior to the pharynx (Fig 2, 22-10A). It is a long dorsoventrally flattened tube that extends posteriorly almost to the anus. Unlike the ectodermal foregut, its walls consist solely of a simple columnar or cuboidal epithelium and its basal lamina. There is no associated muscle, connective tissue, or mesothelium.
The intestine is the region of hydrolysis and absorption. In the middle of the body your view of the intestine is probably obscured by the reproductive system but you can find it again posterior to this region. Ascaris subsists chiefly on monomers (sugars and amino acids) from the intestinal contents of its host. These are absorbed by the microvilliated midgut epithelium.
The intestine extends posteriorly to join the short ectodermal hindgut, or rectum (Fig 2). In females, the rectum is difficult to differentiate from the intestine but in males the rectum is acloaca which receives the male gonoduct and the intestine before opening to the exterior via the anus (Fig 22-18A). Being ectodermal, the rectum is lined with cuticle.
Respiratory System
Energy metabolism is anaerobic and there are no special gas exchange structures.
Fluid Transport System
The unpartitioned hemocoel (= pseudocoel) of nematodes obviates the need for a hemal system and there is none. Transport is thought to be accomplished by diffusion in small species and by movement of the hemocoelic fluid (blood) in large species.
Excretory/osmoregulatory System
The excretory system consists of an enormous H-shaped canal system contained within a single cell (Fig 22-17A,B). The uprights of the “H” are longitudinal canals located in the lateral epidermal cords and extend over the entire length of the worm. The two longitudinal canals connect with each other via a transverse canal near the anterior end of the worm. A short excretory duct leads from the transverse canal to the excretory pore on the anterior ventral midline. The system is thought to be chiefly osmoregulatory. The excretory canal system is difficult to observe in gross dissection of preserved whole specimens. The excretory pore is located immediately posterior to the mouth on the ventral midline but it is difficult to find.
Nervous System
Study of the nervous system of Ascaris requires specially prepared material and will not be attempted. The central nervous system consists of a characteristic cycloneuralian brain which is a tripartite nerve ring around the pharynx (Fig 22-11A). Try to find the brain. Dorsal, ventral and lateral longitudinal nerves arise from the brain and extend posteriorly in the epidermal cords. Of these the ventral nerve cord is most important and is a double ganglionated cord. The dorsal cord is single and unganglionated. A small lateral nerve cord is present in each lateral epidermal cord. Ascaris has no amphids.
Locomotion
The locomotory system comprises the pressurized hemocoel, which is a hydrostatic skeleton, the antagonistic dorsal and ventral longitudinal muscle fields of the body wall, and the elastic cuticle, which contains the hydrostatic pressure and opposes the longitudinal muscles. When one muscle field contracts, the opposite side of the body lengthens to relieve the hydrostatic pressure and the cuticle on that side stretches (Fig 22-12A). Alternate contractions of dorsal and ventral muscle fields result in sinusoidal waves in the dorso-ventral plane passing along the length of the body. The arrangement of the protein fibers in the cuticle allow changes in the length of the worm but not its diameter. This results in the characteristic dorso-ventral thrashing motion that can be translated to efficient forward movement in a viscous medium or an environment, such as wet sand or the walls of a host’s intestine, with solid or resistant surfaces to push against. Nematodes (except for the very small) are relatively helpless in pure water and are not capable of directed motion but have efficient locomotion when there is a substratum to push against.
>1a. If living nematodes are available in the laboratory, place some in a Petri dish of water and watch their motion with a dissecting microscope. Add some sand grains to the dish and note the difference in the effectiveness of locomotion. <
Reproductive System
The reproductive system is a tube with the gonads continuous with the gonoducts so gametes are not released into the hemocoel (Fig 22-18A, 22-19A). Both male and female systems are long tapered tubes lying coiled in the hemocoel. The upstream, solid, free ends of the tubes are small in diameter but expand and become hollow as they extend downstream toward the gonopore. The solid upper ends are the gonads, ovaries or testes. The hollow, larger regions are specialized for various purposes, including transport and storage of gametes. Mitotic divisions of primordial germ cells in the gonad produce diploid gonial cells which move down the gonoduct undergoing gametogenesis on the way. Study the reproductive system of your specimen and then look at a dissection of the opposite sex. You should be familiar with both sexes.
Female
Female ascarids have a Y-shaped reproductive system consisting of two tubes, each with an ovary, oviduct, and uterus forming an arm of the Y (Fig 2, 22-10). The two arms join to form a common (unpaired) vagina which is the stem of the “Y”. The vagina empties to the exterior via the single gonopore. It is convenient to trace the system backwards beginning at the gonopore but keep in mind that female gametes travel in the opposite direction. The upper, small diameter ends are referred to as “ upstream”.
Relocate the approximate position of the vulva on the midventral line and look on the inside surface of the body wall to find a short tube attached to the body wall. This tube is the vaginaand the vulva opens into it (Fig 2, 22-10A). The vagina is formed by the union of two large, convoluted, tubular uteri (Fig 2, 22-19A). Each uterus extends posteriorly, decreasing in diameter, almost to the posterior end of the hemocoel.
Upstream of each uterus is the small-diameter oviduct (Fig 2). A small swelling, the seminal receptacle, is situated at the junction of the oviduct and uterus (Fig 2, 22-19A). The oviductsextends anteriorly, without much change in diameter, to about the level of the vagina where it turns and runs posteriorly again. The two oviducts are coiled around the uteri, the gut, and themselves.
Near the middle of the body each oviduct decreases in diameter to become an ovary. The ovaries are solid, not tubular, and form a mass of small-diameter threads in the middle of the worm.
Oogonia are produced by mitotic divisions of primordial germ cells in the upper ovary. The oogonia move downstream to the upper oviduct where additional mitotic divisions produce primary oocytes. The cell is fertilized in the seminal receptacle before meiotic divisions begin, while it is still a diploid oocyte. Once fertilized, it enters the uterus where it develops a chitinous eggshell and undergoes oogenesis followed by embryonic development. The uterus contains shelled "eggs" in all stages of oogenesis and embryonic development.
Male
The reproductive system of male ascarids resembles that of females but is only one tube rather than two (Fig 22-18A). The solid free upstream end of the tube is the testis. It is a small, white, solid thread coiled in the posterior third of the hemocoel. As it twists back and forth it gradually increases in diameter. Eventually it turns anteriorly and becomes the vas deferens, or sperm duct. This region of the male duct is hollow and contains male sex cells undergoing spermatogenesis. It makes several loops in the middle of the hemocoel, some of which extend posteriorly to the region of the testis.
The last loop of the vas deferens turns posteriorly again and expands abruptly to become the seminal vesicle, a large-diameter tube that runs straight posteriorly in the posterior third of the animal. Its diameter increases as it extends posteriorly and spermatozoa are stored in it.
Before reaching the posterior end of the body, the male duct decreases in diameter once again and becomes the ejaculatory duct. This muscular duct extends posteriorly from the end of the seminal vesicle to join the rectum, or cloaca, immediately anterior to the anus. A region with secretory epithelium, the prostate gland, lies between the seminal vesicle and the ejaculatory duct. Two retractile copulatory spicules are located beside the cloaca. The spicules are housed in deep pouches opening from the cloaca (Fig 22-18A,B). During copulation these are extended out of the anus and into the vulva of the female to hold the vulva open (Fig 22-18D).
Cross Sections
Study stained cross sections of the region of the pharynx (Fig 3, 22-15) and more posterior sections showing the male and female reproductive systems (Figs 4, 5). Orienting these sections is sometimes difficult and you cannot rely on position on the slide for clues. The lateral epidermal cords are much larger than the dorsal and ventral cords and can be used to distinguish lateral from dorsal/ventral (Fig 4, 5). Distinguishing dorsal from ventral is difficult but the best landmark is the gut, which is usually (but not always) in the dorsal half of the hemocoel. Fortunately it doesn’t make much difference if you can’t make the distinction between dorsal and ventral but it is important that you distinguish lateral from dorsal and ventral.
Body Wall
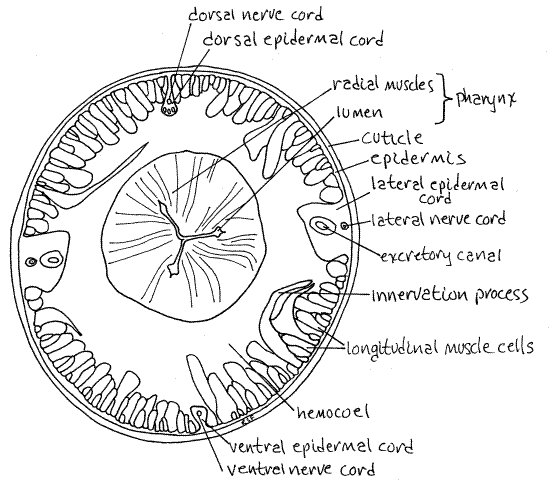
The body wall can be studied on any of your cross sections but, if possible, it is most convenient to begin with the pharyngeal cross section (Fig 3). The outermost layer of the body wallis the thick cuticle. It is a nonliving extracellular secretion that stains pink in most preparations (Fig 3, 4, 22-11).
Immediately inside the cuticle is the thinner epidermis which secretes the cuticle (Fig 3). On most slides the epidermis appears as a thin, pale, pink layer but it may be separated from the cuticle by a white space. If present, this space is an artifact resulting from the slide-making process and in life there is no space between cuticle and epidermis. Together the epidermis and cuticle make up the integument.
The epidermis is a syncytium whose nuclei are located in the four epidermal cords sunken into the body cavity. The right and left lateral epidermal cords are large and easily located (Fig 3, 4). The inconspicuous excretory ducts are in these cords.
The dorsal and ventral epidermal cords are much smaller but can be found by careful inspection. The dorsal and ventral longitudinal nerve cords are usually visible in the dorsal and ventral epidermal cords respectively.
The thickest part of the body wall is the longitudinal muscle layer (Fig 3, 4, 22-11B). This is a single layer of large cells which bulge far into the body cavity and occupy much of it. Each muscle cell comprises an obvious peripheral and a less evident central portion. The peripheral, or fibrillar, region sits on the inside of the basal lamina of the epidermis and contains the contractile fibers of the cell. It is easily recognized because the fibers stain dark pink and form a thick outline around this portion of the cell.
The nucleus and most of the cytoplasm (or sarcoplasm), however, are in a large, but less conspicuous, bulging, centrally located sarcoplasmic region that extends deep into the body cavity. The outlines of this part of the cell are not as apparent as are those of the fibrillar region.
The two lateral epidermal cords divide the longitudinal muscle sheath into antagonistic dorsal and ventral muscle fields. In nematodes the axons of motor neurons do not exit the central nervous system (nerve cords) and do not extend to the muscles. Narrow sarcoplasmic extensions, or innervation processes, arise from the apical ends of the muscle cells and run to a dorsal or ventral nerve cord to synapse with neurons confined to the cord. The innervation processes carry motor commands from the central nervous system to the muscles in the absence of the peripheral axons that perform this function in most animals.
Digestive System
The white space in the interior of the worm is the hemocoel (= pseudocoel), or body cavity, and in it are the digestive and reproductive systems (Fig 3,4). The pharynx (= esophagus) is round in cross section and has thick muscular walls (Fig 3). At rest its lumen is collapsed and is triradiate (= Y-shaped). The lumen is dilated by contraction of the radial muscles in the pharyngeal walls. As part of the foregut, the pharynx is lined by epidermis, which is not visible, and has a thick cuticular lining, which is.
The intestine is usually flattened (Fig 4, 5) but may be dilated and very large. At high power you can see that the intestinal walls are composed of a monolayered columnar epitheliumof very tall cells. The basal ends of the cells rest on a basal lamina, which you can see around the outside of the gut tube. The basal lamina separates the epithelium from the hemocoel and there are no muscles or mesothelium associated with the gut. The apical ends of the epithelial cells are microvilliated and form an absorptive brush border which is visible as a dark line around the midgut lumen. The hemocoel is bounded on the outside by somatic musculature, which is mesodermal, and on the inside by the midgut epithelium, which is endodermal.
Figure 3. Cross section through the pharynx of Ascaris. Nematode71L.gif
Reproductive System
Commercially prepared slides usually have one section each from male and female specimens. These sections are made through posterior regions of the body and include several sections through regions of the reproductive tube(s).
Male
Study the cross section through a male Ascaris (Fig 4). Male cross sections are usually smaller in diameter than female and lack the large egg-filled uteri (Fig 5). The male reproductive system is a long much-coiled tube. The plane of section will have passed through this tube many times and each specialized region of the tube will probably be represented many times. The number of times depends on the level of the section.
Figure 4. Cross section of a male Ascaris. Nematode56La.gif
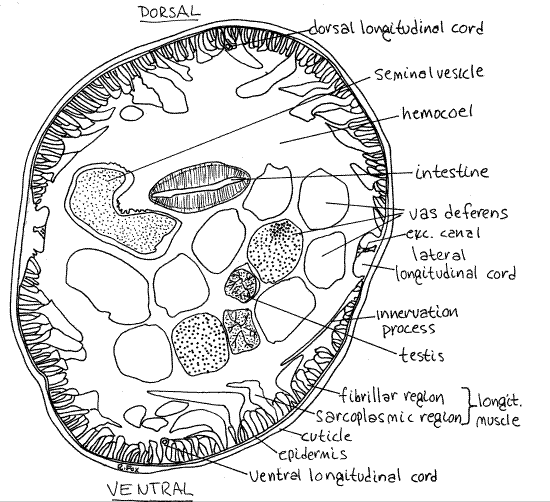
The proximal, upstream end of the tube is the testis which is solid, small in diameter, and enclosed by an epithelium (Fig 4, 22-18A). It is filled with small, spherical primordial germ cells and has no lumen. The primordial germ cells are associated with a branching core, or rachis. Mitotic divisions of the germ cells produce spermatogonia which move downstream to undergo spermatogenesis. Several sections through the testis may be present.
The next region of the male tube is the vas deferens . It is slightly larger in diameter than the testis and is also enclosed by a thin epithelium. Its interior is filled with spermatogoniaand their daughters undergoing spermatogenesis. The organization of the contents of the vas deferens is looser than that of the testis and its sex cells are larger and not attached to a rachis. A lumen is present although it may not be apparent since it is filled with developing germ cells. There should be several sections through the vas deferens in the hemocoel of your specimen.
The next region of the male duct is the seminal vesicle. Amoeboid spermatozoa are stored here (Fig 22-18C). Most slides have a single section through the seminal vesicle but one made too far anteriorly will have none. In life, the seminal vesicle is larger in diameter than the other regions of the male system but in preserved material it often contracts and may be smaller. The epithelial walls of this region are much thicker than those of upper regions of the reproductive tube and this is the best way to recognize it. The ejaculatory duct, which is the downstream-most region of the male system, is too far posterior to be present in the same cross section as the testis and vas deferens.
Female
Study the cross section of a female Ascaris and its reproductive system. It should be recognizable by its two large egg-filled uteri. The female reproductive system consists of two long much-coiled tubes (Fig 2, 22-10A). The plane of section passes through these tubes many times and each region of a tube will probably be represented many times. The number depends on the level of the section. Most sections should include cuts through the ovaries, oviducts, and uteri. The twisting, coiled nature of the reproductive system results in multiple sections through the ovaries and oviducts, but probably not the uteri. Unlike that of the male, the female reproductive system is double so there are two ovaries, two oviducts and two uteri. There is only one vagina and gonopore but these will not be represented on the slide.
The smallest sections are of the ovary (Fig 5, 22-19A). These are easily recognized because they are solid whereas the oviducts and uteri are hollow (albeit so full of eggs they may seem to be solid). The ovary has a central cellular core, known as the rachis, from which radiate a single layer of long narrow primordial germ cells. Their nuclei are usually easy to see. Mitotic divisions of the germ cells produce oogonia which move downstream as they undergo oogenesis. The ovary and oviduct are surrounded by thin epithelia which are often pulled away from the germinal cells leaving a white space between them. This space is an artifact. The rachis may appear to be a tiny lumen but it is not. If you look closely, you will see pale, lightly staining cells in it. (If there are no cells here and the center of the organ is white, you are looking at an oviduct, not an ovary.)
The oviduct has much the same appearance as the ovary but is a little larger in diameter and is hollow (Fig 5). Its epithelium is similar to that of the ovary and is thick but it has a smalllumen instead of a rachis. Around the lumen is a single thick layer of oogonia (upstream) or oocytes (farther downstream).
The uteri, of which there should be two in most cross sections, are much larger than either oviducts or ovaries. They are filled with large, shelled “eggs” in various stages of oogenesis and development and the uterine wall is thick and muscular.
Life Cycle
The ascarid life cycle involves only one host which becomes infected when it ingests Ascaris eggs in its food or water. These hatch in the intestine and juvenile worms migrate to the liver where they enter the host’s hemal system. They are carried in the blood to the lungs where they enter the lumen of the alveoli. From here they crawl to the pharynx, then follow the gut lumen to return to the small intestine where they mature into adult roundworms and feed on chyme.
Figure 5. Cross section of a female Ascaris featuring the reproductive system. The details of the body wall have been omitted. Nematode57La.gif
Tremendous numbers of worms may be present in a single host (Fig 22-21). After maturation, copulation occurs and females produce and release shelled eggs which leave the host in the feces. When fully developed and infective, an ascarid “egg” contains a tiny juvenile worm capable of infecting a new host. Ascarid eggs are difficult to kill and remain viable in soil for as long as 20 years and are widespread in the environment. Development is direct with no distinct larva. The juveniles undergo four molts to become adults (Fig 22-9).
Ingestion of eggs and subsequent infection is most common in children due to their habit of playing in grass and sand and indiscriminately ingesting a variety of potentially contaminated materials.
References
Goodchild CG. 1950. Ascaris lumbricoides , pp 243-251 in Brown FA. (ed). Selected Invertebrate Types. Wiley, New York. 597p.
Crofton HD . 1966. Nematodes. Hutchinson Univ. Library, London. 160p.
Maggenti A. 1981. General Nematology. Springer-Verlag, New York. 372p.
Petrunkevitch A . 1916. Morphology of Invertebrate Types. MacMillan, New York. 263p.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Schmidt GO, Roberts LS . 1989. Foundations of Parasitology, 4 th ed. Times Mirror/Mosby, New York. 750p.
Wells MM. 1924. Ascaris megalocephala, an outline of the life history, anatomy, and cytology of the horse- Ascaris. General Biological Supply (Turtox) Pub. Pp. 1-24.
Wright KA. 1991. Nematoda. Harrison FW, Ruppert EE (eds.). Microscopic Anatomy of Invertebrates vol. 4 Aschelminthes . Wiley-Liss, New York. 424p.
Supplies
Dissecting microscope
Long, narrow dissecting pan (aluminum ice cube tray with wax bottom is ideal)
# 1 stainless steel insect pins
10X hand lens
Cross section slides of male and female reproductive systems
Cross section slide through pharynx
Living small nematodes such as Cephalobus
Dissecting microscope
Beach sand
Cross section slides of the reproductive region of male and female Ascaris are available from Carolina Biological and Ward’s Natural Science. Cross section slides of the pharyngeal region are available from Ward’s