Invertebrate Anatomy OnLine
Argopecten irradians
Bay Scallop
with notes on Placopecten magellanicus ©
4jul2006
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises can be accessed by clicking on the links on the left. A glossary and chapters on supplies and laboratory techniques are also available. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
MolluscaP, Eumollusca, Conchifera, Ganglioneura, Ancyropoda, Bivalvia C, Metabranchia sC, Filibranchia SO, Pteriomorpha O, Pectinoidea SF, Pectinidae F (Fig 12-125, 12-122)
Mollusca P
Mollusca, the second largest metazoan taxon, consists of Aplacophora, Polyplacophora, Monoplacophora, Gastropoda, Cephalopoda, Bivalvia, and Scaphopoda. The typical mollusc has a calcareous shell, muscular foot, head with mouth and sense organs, and a visceral mass containing most of the gut, the heart, gonads, and kidney. Dorsally the body wall is the mantle and a fold of this body wall forms and encloses that all important molluscan chamber, the mantle cavity. The mantle cavity is filled with water or air and in it are located the gill(s), anus, nephridiopore(s) and gonopore(s). The coelom is reduced to small spaces including the pericardial cavity containing the heart and the gonocoel containing the gonad.
The well-developed hemal system consists of the heart and vessels leading to a spacious hemocoel in which most of the viscera are located. The kidneys are large metanephridia. The central nervous system is cephalized and tetraneurous. There is a tendency to concentrate ganglia in the circumenteric nerve ring from which arise four major longitudinal nerve cords.
Molluscs may be either gonochoric or hermaphroditic. Spiral cleavage produces a veliger larva in many taxa unless it is suppressed in favor of direct development or another larva. Molluscs arose in the sea and most remain there but molluscs have also colonized freshwater and terrestrial habitats.
Eumollusca
Eumollusca, the sister taxon of Aplacophora, includes all molluscs other than aplacophorans. The eumolluscan gut has digestive ceca which are lacking in aplacophorans, the gut is coiled, and a complex radular musculature is present.
Conchifera
Conchifera, the sister taxon of Polyplacophora, includes all Recent molluscs other than aplacophorans and chitons. The conchiferan shell consists of an outer proteinaceous periostracum underlain by calcareous layers and is a single piece (although in some it may appear to be divided into two valves). The mantle margins are divided into three folds.
Ganglioneura
Most Recent molluscs are ganglioneurans, only the small taxa Aplacophora, Polyplacophora, and Monoplacophora are excluded. Neuron cell bodies are localized in ganglia.
Ancyropoda
The mantle cavity, with its gills, is lateral. The calcareous portion of the shell is bivalve, with the valves opening laterally and joined dorsally by a derivative of the periostracum.
Bivalvia C
Bivalvia is a large, successful, and derived taxon. The body is laterally compressed and enclosed in a bivalve shell. The two valves are hinged dorsally. The the foot is large and adapted for digging in the ancestral condition. A crystalline style is usually present but never is there a radula. The mantle cavity is lateral and in most bivalves the gills are large and function in respiration and filter-feeding. The head is reduced and bears no special sense organs. The nervous system is not cephalized. The group includes scallops, clams, shipworms, coquinas, marine and freshwater mussels, oysters, cockles, zebra mussels, and many, many more.
Metabranchia sC
Metabranch gills are adapted for filter feeding. Water enters the mantle cavity posteriorly.
Filibranchia SO
Filibranchs are suspension-feeding bivalves with filibranch gills.
Pteriomorpha O
Pteriomorph bivalves are epibenthic and live on, rather than in, the bottom. They may be attached or unattached, may have a byssus or not, and may cement one valve to the substratum or not. The foot is reduced and the mantle margins are not fused. The gills are large and used for filter feeding. There is a tendency to reduce or loose one adductor muscle. Siphons are absent or reduced. This group includes the well-known arcs, mussels, scallops, pen clams, and oysters.
Laboratory Specimens
Scallops are easily dissected but are adapted for an epibenthic existence sitting atop the sediment and many aspects of their anatomy are modified accordingly. Some features, such as the loss of one adductor muscle, reduction of the foot, hermaphroditism, lack of siphons, and the ability to swim are not typical of bivalves. Scallops are good examples of filibranch bivalves.
If a more typical species is desired, Mercenaria or Corbicula are recommended. Scallops, however, are easier to dissect than Mercenaria. At least one scallop species is available on most coasts and, as there is little internal variation between species, this account of scallop anatomy can be used with any of them. Differences in shell details must be expected and there is, of course, some variation in internal anatomy. This account is a description of the anatomy of Argopecten irradians, the bay scallop, but includes parenthetical comments for Placopecten magellanicus, the giant sea scallop.
Argopecten irradians (=Pecten irradians) is common in shallow water from New England south through the Caribbean to South America. It is especially common in grass beds and reaches 8 cm in diameter.
Placopecten magellanicus (=Pecten tenuicostatus) is a much larger species, reaching 20 cm in diameter. It lives in deeper, colder water from Labrador south to the middle Atlantic states on the North American east coast. Although Placopecten is large and easily dissected it does not exemplify the filibranch gill condition as it has vascularized, tissue connections between its gill filaments. It is unusual among scallops in this regard. It is also unusual in being gonochoric whereas most scallops are hermaphroditic.
Pecten caurinus is a large, offshore species occurring from Alaska to California on the Pacific Coast. All three species are commercially harvested.
Scallops are unattached surface dwellers living on soft ocean bottoms. They do not burrow or attach, rather lie with the right valve down on the surface of the sediment (Fig 12-115A). They are active and mobile animals with well developed sensory organs and musculature. They swim by ejecting jets of water from between the valves. They do this for ordinary locomotion and as an escape response from predators such as starfish. Scallops are a valued seafood and several species support commercial fisheries.
This exercise can be conducted using living or preserved specimens. As usual, living specimens are preferred but preserved animals are entirely acceptable. Living specimens can be relaxed rapidly in isotonic magnesium chloride (see Techniques chapter).
External Anatomy
If your scallop is living, place it in a dish of isotonic magnesium chloride solution. Cover the dish to prevent splashing of nearby equipment. The scallop will relax soon. If it is preserved, place it in tapwater instead of magnesium chloride.
Shell
The shell consists of two valves which are similar but not identical. The right valve of Argopecten is deeper than the left (The left valve of Placopecten is the deeper). A shell with two valves of different shapes is said to be inequivalve. One with identical valves is equivalve. Most scallops have slightly inequivalve shells.
The two valves are held together dorsally at the hinge (Fig 1). The hinge of scallops is straight. The raised umbo of each valve arches medially straight toward the middle of the hinge. The umbo is the oldest part of the valve.
At each end of the hinge of each valve is a projecting wing, or ear (Fig 1, 12-115B). One is anterior, the other posterior. The two wings are similar but not identical and can be used to tell anterior from posterior. The anterior wing, especially that of the right valve, projects from the hinge, with a slight overhang whereas the posterior wing does not project, is blunt, and slopes gradually to the shell. You should now be able to determine dorsal, ventral, anterior, posterior, right and left.
Figure 1. The inside of the right valve of Argopecten irradians, from Beaufort, North Carolina. A = anterior, P = posterior. Bivalve81La.gif
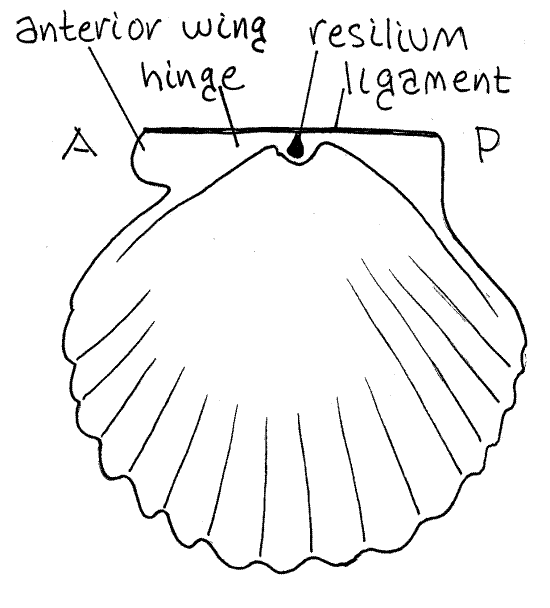
Curved, concentric growth lines can be seen on the surface of the valves. Those of the left valve are easiest to see and are enhanced by raised ridges of periostracum. The left valve, remember, is the upper one when the scallop is resting on the bottom in its life position. The right valve rests against the substratum.
The two valves are not attached to each other on the anterior, ventral, and posterior margins. The space between the open valves is the gape. Scallops do not have siphons.
Press the ventral edges of the valves together and notice that, whereas the shell closes around almost all of its margin, two small gaps are left open, one below each wing. These are the dorsal exhalant apertures through which water is jetted when the valves are closed during swimming. Scallops swim by rapidly closing the valves to force water out between the valves.
Scallops use two different swimming mechanisms. Normal locomotory swimming is accomplished by emitting jets of water from the two dorsal exhalant apertures. This propels the animal with the ventral end (gape) first. The more rapid escape response is accomplished by violent ejection of water around the ventral margin of the valves and sends the animal backwards, with the dorsal edge (hinge) leading.
Viewed from the side, each valve is nearly symmetrical. Valves whose anterior half is a mirror image of the posterior half are said to be equilateral. The valves of Argopecten are equilateral except for the slight dissimilarities of the two wings. The mussels Mytilus and Geukensia, which you may have studied, are good examples of strongly inequilateral species. (The left valve of Placopecten is equilateral but its right valve is not quite so because of the differences in the two wings.)
Note the heavy fluting of the valves. These ridges and grooves increase the strength of the shell without increasing its weight. This is an advantage for a swimming animal, such as a scallop, which needs a strong shell but cannot afford extra weight. (The valves of Placopecten bear fine radial ridges but are not fluted).
Relaxation
If you have a living scallop, it should be relaxed by now but if it is not, do the following. Remove your scallop from the dish and hold it in your hand until it opens its valves. When it does, quickly slip the handle of a teasing needle crosswise into the gape to prevent the valves from closing again. Use a rubber band around the handle of the needle and the ears of the shell to hold the handle in place, much like a bit in a horse's mouth. Return the scallop to its culture dish of isotonic magnesium chloride for a few minutes.
With the scallop relaxed, look into the gape and locate the thick white column of muscle extending from the middle of one valve to the other. This is the (only) adductor muscle. Also note that the inside surface of each valve is covered by a thin sheet of tissue, the mantle skirt, or mantle lobe. The margin of the skirt is thicker around the edge of the valve.
" Carefully reach into the gape with a scalpel and scrape (not cut) the left mantle skirt away from the left valve. When you reach the adductor muscle, scrape it away from the left valve. Continue until you have separated the left mantle skirt and the adductor muscle from the left valve. Be careful you do not cut any other tissues. Gently pull the two valves apart and detach the left valve from the right. Place the right valve, with the animal in it, in the dish of magnesium chloride. Set the empty left valve aside.
Look at the animal, still in the right valve, in your culture dish. The hinge has a long, narrow, brown, external, proteinaceous ligament extending anterior to posterior for the length of the hinge (Fig 1). It is stretched and under tension when the valves are closed. At the middle of the hinge there is a compact, thick brown, proteinaceous resilium inside the hinge. When the valves are closed the resilium is squeezed and under compression. Elastic recoil of the ligament and resilium open the valves when the adductor muscle is relaxed. The adductor muscle closes the valves.
Mantle and Mantle Cavity
Mantle Skirts
The mantle consists of two large lateral folds, lobes, or skirts, of body wall that enclose the animal on the right and left. The large space between these two skirts is the mantle cavityand the body and gills lie within it. The body of your specimen now lies surrounded by the spacious mantle cavity between the right and left mantle skirts (unless you accidentally removed the left skirt with the left valve).
The mantle skirt is a thin double layer of body wall lying on the inside of each valve. Each mantle skirt arises from and is attached to the dorsal body wall. The heavily vascularized mantle of scallops is thought to be a major respiratory organ, perhaps more important than the gills. Over most of its surface it is very thin and transparent but along its periphery it is thicker, pigmented, and muscular. Its inner surface is ciliated.
Mantle Folds
The mantle margin along the edge of the valve is much thicker than the rest of the mantle. This part of the mantle has several functions and is elaborated into three parallel folds, or ridges, that follow the edge of the valve (Fig 12-91). Look at the margin of the left mantle skirt with the dissecting microscope. This skirt has been detached from its valve and its three folds are easy to see.
The outer fold (=shell fold) secretes two layers of the shell and, it in an intact specimen, lies against the shell. It is the smallest and least evident of the three folds. It is a narrow, unpigmented ridge of tissue running around the mantle margin. It is separated from the next fold by a shallow furrow, the periostracal groove, from which the periostracum is secreted. The outer surface of the outer fold secretes the middle, prismatic layer (= ostracum) of the shell and lies against it (Fig 12-91). The inner surface of the outer fold, adjacent to the periostracal groove, secretes the periostracum, which is the outermost layer of the shell. The entire outer surface of the mantle skirt secretes the lamellar layer (hypostracum).
The very thin, shiny periostracum is secreted from the periostracal groove between the middle and outer mantle folds and can be seen intact on the undisturbed right mantle skirt extending from the mantle margin over the edge of the valve. It is transparent and very thin but can be seen with good light and careful focus.
The middle fold (or sensory fold ) is much thicker, is pigmented, and is easily seen. It bears stalked green or blue eyes and a band of contractile sensory tentacles (Fig 12-115A).
The eyes are complex and have many of the features of vertebrate eyes. The pale cornea is underlain by a pigmented, blue iris is visible from the outside. Inside, below the iris, is a lens, then a photosensitive retina, a pigmented layer, a reflective tapetum, and a nerve. The eyes in the region of the dorsal exhalant apertures remain exposed and functional, even when the valves are closed. The tentacles of the middle fold are muscular, extensible, and sensitive to touch.
The inner fold (or muscular fold ) of the mantle margin is the largest of the three. Most of it forms a veil, or velum, that hangs away from the mantle into the gape. The velum forms a curtain across the gape and is a valve to help channel water currents during swimming. Guard tentacles of various sizes extend from the margin of the velum across the gape to interdigitate with guard tentacles from the velum on the opposite side. The guard tentacles are thought to be chemosensory and responsible for initiating the characteristic escape response of scallops. The inner fold is muscular and contractile. Its muscles originate on the shell along the pallial line paralleling the margin of the valve (Fig 12-91). The pallial line of scallops is indistinct.
The posterior margin of the mantle of scallops does not form a pair of siphons as it does in most bivalves.
Body
The visceral mass occupies most of the interior of the mantle cavity anterior and dorsal to the adductor muscle. Look below the anterior end of the hinge to find the white, wormlike foot(Fig 2, 12-115B). It extends anteriorly from visceral mass. The scallop foot is reduced in comparison with that of most bivalves. As surface dwellers, scallops have no need for the well- developed digging foot of typical bivalves. A byssal groove is present along its posterior edge but most adult scallops do not regularly produce a byssus although juveniles often do (Fig 12-110B).
Figure 2. The left side of Argopecten irradians. The left valve, mantle skirt, and gill have been removed to reveal the visceral mass, adductor muscle, foot, and right gill. The lateral palp has been deflected to reveal the medial palp and the sorting field. Bivalve82La.gif
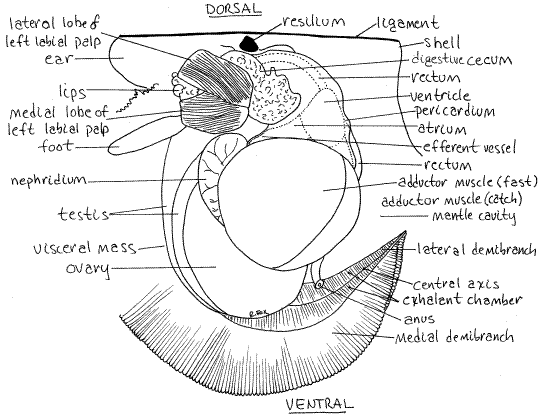
Adductor Muscle
Find the large, white adductor muscle located close to the center of the valve (Fig 2). This, the only adductor muscle present, is homologous to the posterior adductor of other bivalves. Scallops have no anterior adductor muscle.
Note that the muscle is divided into two regions. The largest is antero-dorsal and is the fast muscle (= quick muscle). It is composed of striated fibers that contract rapidly but fatigue easily (Fig 2, 12-115B). The posteroventral border of the muscle is a much smaller catch muscle of smooth fibers and abundant collagen. These fibers react slowly but do not fatigue. Which part of the muscle do you suppose is used for escape swimming? Which would be used to hold the valves closed for long periods?
In most bivalves the chief function of the adductor muscles are to hold the valves tightly together, perhaps for long periods, and the catch muscle is larger than it is in scallops. Scallops, on the other hand, use the muscle more for locomotion and have more need for fast muscle than for catch. They rely on escape rather than closing the shell for protection. Consequently, the fast portion is the larger. Scallops typically do not remain closed for long periods. If you are studying a living specimen you probably noticed that it opens its valves soon after any disturbance and does not remain closed for long periods as would a quahog (clam), for example.
Gills
Next look at the large crescent-shaped gills. There is one gill on each side of the body. The gills are, in addition to being respiratory organs, the food gathering device of these filter-feeding animals. The gills extend the length of the mantle cavity and divide it into a small dorsal exhalant chamber (= suprabranchial chamber), which you cannot see at present, as it is dorsal to the gills, and a large inhalant chamber (= branchial chamber), which is the space you see surrounding the gills (Fig 12-93B).
Bivalve gills are composed of a central axis from which arise two rows of long slender filaments (Fig 3). Each of the two gills is a holobranch, or whole gill, and is composed of twodemibranchs, or half gills, one lateral and one medial (Fig 3). The holobranch is attached to the body by the central axis running longitudinally along the roof of the mantle cavity and the two demibranchs are composed of the combined filaments on the two sides of the axis. There is a demibranch on each side of the axis. The surfaces of the filaments are covered with cilia, some of which generate the feeding current and others that move trapped food particles and mucus anteriorly to the mouth.
Figure 3. Diagrammatic view of a portion of a scallop holobranch. The mantle, which is not figured, is to the left and the visceral mass to the right. The angle between lamellae is exaggerated in the drawing to the extent that interlamellar junctions are not possible. Bivalve83La.gif
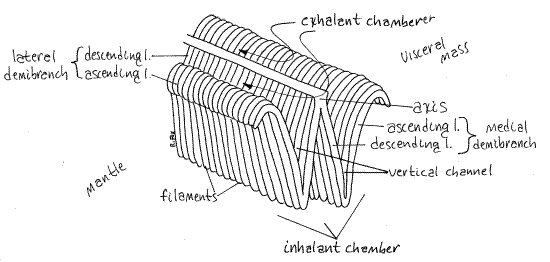
Gill Detail
This section is a detailed discussion of the anatomy and function of bivalve gills (Fig 12-96D). It may be omitted at the instructor's discretion.
Lamellae
" Scallops are good subjects for a careful study of filibranch gill structure and function. To undertake such a study, deflect or remove the two left demibranchs and the left mantle so you can see the right gill more clearly.
Examine the medial demibranch of the right gill with the dissecting microscope. It, like the other demibranchs, has two surfaces; an ascending lamella and a descending lamella(Fig 3). Each holobranch is composed of two demibranchs and four lamellae. Demibranchs (and lamellae) are composed of closely-spaced, contiguous filaments attached to the central axis (Fig 3, 12-96D). The descending lamellae drop ventrally from the central axis. At their ventral extremity, they reverse direction and climb dorsally as the ascending lamella. Each holobranch thus has two descending lamellae and two ascending lamellae.
The ascending lamella of the medial demibranch is facing you now and its descending lamella is hidden from your view by the ascending lamella. The ascending lamellae of all four demibranchs are about half the width of the descending lamellae. The descending lamellae are attached to the central axis but the ascending lamellae are unattached unlike the situation in most lamellibranchs.
Filaments
The lamellae are composed of hundreds of narrow filaments (Fig 3, 12-96D) that run from the central axis of the gill down the descending lamella then reverse direction at the ventral to edge of the demibranch and run up as part of the ascending lamella. Each filament begins at the central axis and descends, bends, and ascends. Each is thus bent near its middle with part of it contributing to the ascending lamella and the other part to the descending lamella.
The gills of scallops, arcs, and mussels (Filibranchia) are filibranch in that the filaments are independent of each other. Adjacent filaments are held together by specialized cilia and there are few or no tissue connections between them. More advanced gills, such as the pseudolamellibranch gills of oysters and pen clams, have adjacent filaments held together by occasional tissue interfilamentar junctions. Even more advanced gills, such as the eulamellibranch gills of the Venus clams, have extensive interfilamentar tissue junctions. So extensive are the junctions that most of the space between filaments is filled with tissue leaving only small pores, or ostia, through an otherwise unbroken lamella.
Plicate Gills
The lamellae are corrugated so that the gill consists of parallel ridges and grooves. Such a gill is said to be plicate and this is a common condition among the bivalves. The ridges areplications (Fig 4). The gills of mussels are not plicate. Each plication is made of many filaments. The filaments are perpendicular to the central axis of the gill.
Plicate gills are composed of two types of filaments and are said to be heterohabditic. Mussels, with only one type of filament, are homorhabditic.
Most of the filaments are very small in diameter and together make up the plications of the lamellae. They are called the ordinary filaments (Fig 4). The ordinary filaments are slender and consist of a chitinous central tube surrounded by a thin ciliated epithelium.
The bottoms of the valleys between the plications are made of principal filaments. This second type of filament is larger than an ordinary filament (Fig 4). Principal filaments provide most of the structural support to maintain the shape of the gill. The corresponding principle filaments of adjacent lamellae are held together by tissue interlamellar junctions (Fig 4).
The interior of each demibranch is the exhalant chamber and is hollow (Fig 3, 4). This chamber is imperfectly divided into vertical channels by contact between the principal filaments of the two lamellae of the demibranch. Each ridge, or plication, is made of l6 ordinary filaments (Fig 4) and encloses a vertical water channel of the exhalant chamber.
Each of the two walls of the tube is made of 16 filaments so it might seem that there should be 32 filaments in each tube but on reflection you will recall that the 16 filaments on the two sides are only opposite ends (descending and ascending) of the same 16 filaments (compare Fig 3 and 4).
Figure 4. A frontal section of two plications (vertical channels) of a scallop gill. Bivalve84La.gif
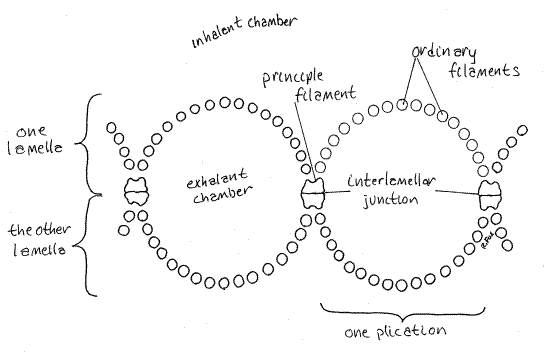
At the bottom of each groove is located a principal filament. Principal filaments are larger and stronger with a heavier chitinous skeleton. The two edges of opposing principal filaments are joined to each other, especially ventrally. Interlamellar junctions extend from the medial limb of each principal filament to its lateral limb. In this manner the two lamellae of a demibranch are held together and the grooves between the ridges are created. This is what makes the gill plicate. This also subdivides the interior of the demibranch into the vertical channels that communicate with the exhalant suprabranchial chamber above the gills.
Attachments of Lamellae to Body
In scallops and mussels the filaments are attached to the body only at the central axis (Fig 3). This means that the demibranchs themselves are attached along only one edge and that to the central axis. Only the descending lamellae are attached to the body. The dorsal edges of the ascending lamellae are not attached to the body and as a consequence, tend to separate from their descending lamellae so that the exhalant chamber and vertical channels are wider dorsally than ventrally. In eulamellibranch bivalves the demibranchs are attached at the central axis, foot, and mantle skirt. Each holobranch thus has three attachments. The lamellae are parallel rather than diverging.
Filibranch Gills
Scallops, like mussels and arks, have filibranch gills. This is the primitive condition of lamellibranch gills and in it adjacent filaments are not joined to each other by tissue connections. (Placopecten has pseudolamellibranch gills with tissue and vascular connections between adjacent filaments. It differs from other scallops in this regard.) There are no solid interfilamentar junctions in most scallops. The filaments are held together only by special ciliary junctions.
Run your minuten nadel between two adjacent secondary filaments and "unzip" the ciliary connections between the two. You can now look inside the demibranch into the vertical channels of the exhalant chamber. You can also look across the chamber and see the lamella on the other side of the demibranch. Follow the "unzipped" filament down the ascending lamella to the free ventral to edge of the demibranch to demonstrate that it is continuous with the descending limb in the descending lamella.
Inside the exhalant chamber look for regularly spaced connective spurs on the edges of the filaments. These are not tissue connections. The spurs are muscular, ciliated, and occur at regular intervals along the ordinary and principal filaments. The cilia of the spurs hold adjacent filaments together. Your use of the nadel broke the ciliary connections and separated the filaments from each other. Through their movement, the spurs can vary the interfilamentar space and change the pore size of the filter. Their cilia make the ciliary interfilamentar junctionswith adjacent filaments.
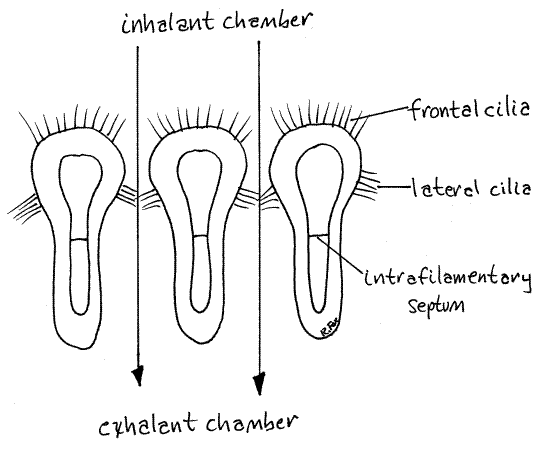
Cilia
The epithelium of the gill filaments bears two types of cilia with two different functions. Frontal cilia are located on the outer surface of the filaments and lateral cilia are on the surfaces facing other filaments (Fig 5). (There are no laterofrontal cilia in scallops.)
The lateral cilia generate the feeding current that passes between adjacent filaments from the inhalant to the exhalant chamber. The filaments produce mucus when struck by particles. Particles attempting to pass between filaments are trapped by mucus and then moved, with the mucus, over the inhalant surface of the lamella by the frontal cilia.
On the surface of the lamella frontal cilia beat in different directions depending on their location on a plication. Cilia on the ridges of plicae generate a current that moves ventrally whereas cilia in the grooves beat dorsally. Thus particles may be carried dorsally in the grooves or ventrally on the ridges.
At the dorsal or ventral edge of the demibranch the food particles and mucus are transported anteriorly to the labial palps by other frontal cilia. There are no well-developed food grooves in Argopecten such as are present in mussels but the dorsal lip of the ascending lamellae are curved over to form a trough in which there is a strong anteriorly directed current (Fig 3, 12-99C).
Figure 5. Frontal section of three ordinary filaments of a scallop gill lamella. The arrows indicate the direction of ciliary currents generated by the lateral cilia. Bivalve85La.gif
>1a. Orient the specimen (if living) so the medial demibranch is horizontal . Place chalk dust or carmine-seawater suspension on the surface of the demibranch. Can you see motion? In which direction(s)? Look on the edges of the lamellae for transport anteriorly. Don’t confuse ciliary currents with density currents. <
>1b. Remove a few complete filaments from a demibranch and place them on a slide. This is best accomplished by selecting one plication and freeing it from its attachment at the central axis. Examine the slide with the dissecting microscope and study the relationship between the ordinary and principal filaments, the water tube, the ridges and grooves, the lamellae, demibranchs, and the entire gill. You should now understand the structure of bivalve gills. If your specimen is living, look for beating cilia on the filaments. Try to distinguish lateral from frontal cilia. <
Labial Palps
Find the flat labial palps at the anterior end of each gill (Fig 2). There is one on each side and each resembles a small gill. Each consists of a lateral lamella and a medial lamella. The ridged and ciliated surfaces of the two lamellae are close together and face each other.
The palps of the right and left sides are connected with each other across the middle of the head (Fig 12-100). The two lateral lamellae, one left and one right, are united with each other by the upper lip which runs transversely across the midline above the mouth (Fig 2). Similarly, the two medial lamellae are connected to each other by the lower lip which crosses the midline below the mouth. The frilly lips are tightly intertwined with each other and must be carefully separated in order to see the mouth.
The opposed (facing) surfaces of the two lamellae on each side are covered by deep, closely spaced, ciliated ridges and grooves extending outward from a central oral groove at the junction of the two lamellae. Particles (entrapped in mucus) move to the mouth from the gills along the oral groove. Flocculent organic particles are moved toward the mouth by the cilia of the ridges and oral groove whereas mineral particles are moved by the groove cilia to the edge of the palp where they drop into the mantle cavity as pseudofeces.
>1c. Separate the two lamellae of a palp so you can see the opposed surfaces. Place some carmine of chalk powder in seawater on the surface of each in turn. Watch the ciliary currents. <
Pseudofeces
Suspended particles striking the surface of lamellibranch gills cause the immediate local release of mucus which binds the particle and holds it to the gill. Large numbers of particles impinging on the gills cause the release of proportionately more mucus. Normal feeding involves relatively small numbers of (mostly organic) particles and correspondingly small amounts of mucus. Under these circumstances the ciliation of the gills and palps eventually carries the food and mucus to the mouth and gut.
In water with a heavy silt load something else happens. The large number of particles causes the release of a copious mucus which forms longitudinal strings across the face of the gill lamella. These strings are transported by the ridge cilia and hence move to the ventral to edges of the lamella. The food grooves here are weakly developed and cannot accommodate these large, bulky strings which consequently fall off the gill into the mantle cavity. They are known as pseudofeces and are eliminated by water currents from the mantle cavity. They never enter the mouth.
Hemal System
The hemal system consists of heart, vessels, blood, and hemocoel. The heart is located in the pericardial cavity enclosed in the membranous, triangular, white, translucent pericardiumposterior to the digestive ceca and dorsal to the adductor muscle (Fig 2, 6, 12-115).
" Open the pericardial cavity carefully, by removing the pericardium, if it is not already open.
The heart is penetrated by the median, tubular rectum which runs longitudinally through the pericardial cavity. Inside the pericardial cavity is a single large, white, muscular ventricle. (It may resume beating if you transfer your living specimen to seawater.) The ventricle surrounds the rectum. The surface of the ventricle is smooth and its two lobes extend far to the right and left of the rectum.
The two atria enter the ventricle on its two antero-ventral corners, one on each side (Fig 2, 6). The atria are elongate and triangular with irregular surfaces. They extend from the base of the gills from which they receive oxygenated blood via the efferent branchial vessel. The atrial walls are slightly yellowish or brownish because of the pericardial glands located there. These glands bear podocytes and produce an ultrafiltrate of the blood into the pericardial cavity (Fig 12-118).
An anterior aorta exits the ventricle dorsally and runs anteriorly (Fig 12-115B). A posterior aorta exits the ventricle immediately ventral to the rectum and runs posteriorly.
Digestive System
Find the mouth on the midline, between the upper and lower lips. It is obscure and difficult to find, even when not covered by the lips. It opens into the esophagus. Dorsal and posterior to the mouth are the two large, dark brown or greenish-brown digestive ceca (=liver). In gross dissection the ceca appear to be a single organ. Parts of it is very close to the surface and is visible through the body wall. The mouth and esophagus are surrounded by the digestive ceca and the labial palps are attached beside it. It connects by ducts to the stomach. The stomach is located in the visceral mass dorsal to the adductor muscle.
" Insert a blunt probe into the mouth and esophagus and cut along the probe to reach the stomach. Make the cut on the left side.
As you cut through the dark digestive ceca, note the numerous branching ducts that extend through its tissue. These ducts lead to the stomach. The relatively long esophagus passes posteriorly through the dorsal part of the digestive ceca and joins the stomach dorsally (Fig 6). The stomach lies a little to the left of the esophagus.
" Open the stomach. Its inner walls are complexly folded (Fig 12-102). Food is moved through the gut by cilia and the stomach walls have a conspicuous ridged and grooved sorting field. There is an opalescent, chitinous gastric shield on the posterior wall.
A very long gelatinous crystalline style rotates against the gastric shield and releases digestive enzymes into the stomach lumen. At present you can see only a short piece of the proximal end of the style. The large openings of the ducts of the digestive ceca can be seen in the walls of the stomach.
" Follow the intestinal lumen by cutting through the viscera with fine scissors. Working under magnification and under liquid, insert the point of a fine scissors in the stomach and follow the gut lumen posteriorly by cutting through the intervening tissue. Periodically blow the debris away with gentle jets of water from a pipet. Cut along the length of the intestine, exposing its lumen as you go. Refer to Figure 6 for help following the lumen.
The intestine loops through the visceral mass. In Argopecten there are many loops but in Placopecten the situation is simpler and the intestine consists of three regions. First is a descending region that runs ventrally from the stomach, then an ascending region that climbs back to the original dorsal position, and finally the rectum which extends posteriorly and ventrally to the anus (Fig 6, 11-115).
In both species the first region, the descending intestine, actually has a double lumen consisting of the intestine and the adjacent tubular style sac (Fig 12-103A). The two curve ventrally and slightly anteriorly from the stomach through the visceral mass anterior to the adductor muscle. The combined intestine and style sac exit the anteroventral corner of the stomach. The lumina of the style sac and intestine are continuous over the length of the style sac. They are incompletely separated from each other by two ciliated ridges, or typhlosoles. The style sac ends a little before the descending intestine reverses direction and becomes the ascending intestine.
Figure 6. Diagram of a dissected Placopecten from Walpole, Maine opened and viewed from the left side. Bivalve86La.gif
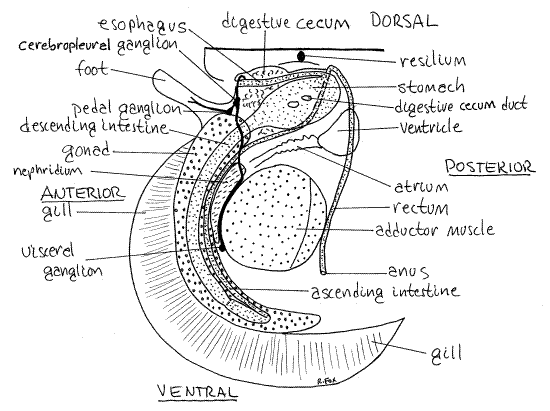
The style sac contains, secretes, and rotates the crystalline style. The style sac is much larger in diameter than the intestine beside it. Together they curve anteriorly and ventrally, embedded in the visceral mass, more or less paralleling the anterior edge of the adductor muscle. The style sac and style end when they reach a position opposite at about 7:00 o'clock on the adductor muscle.
The intestine continues a little farther without the sac and then reverses direction sharply, moves a little to the right and extends dorsally as the ascending intestine. The ascending intestine may make several loops in the visceral mass before ending up in the dorsal visceral mass on the right side of the stomach. Here it turns posteriorly and ventrally and becomes the rectum.
" It is not required that you trace the ascending intestine but if you wish to do so it is best to wait until you have completed the remainder of the exercise and then return to this section. Skip this part now and go on to the rectum. When and if you are ready to trace the ascending intestine, remove the animal from the right valve, cut away the right mantle and gill, and then follow the intestine as it ascends on the right side. The ascending intestine is best dissected from the right side. It parallels the route of the descending limb but is closer to the adductor muscle, reaches the posterior end of the digestive cecum, and then arches to the dorsal midline and turns posteriorly and becomes the rectum.
The rectum lies on the midline for its entire length. Shortly after becoming the rectum the gut enters and passes through the pericardial cavity (Fig 2, 12-115B) and can be traced from here to the flaring anus without dissection (Figs 2, 6). The terminal portion of the rectum occupies a tubelike extension of the body wall and is unattached distally. It lies on the posterior curve of the adductor muscle.
Excretory System
The two nephridia, or kidneys, are large, brownish or pinkish organs on the anterior edge of the adductor muscle at the base of the central axis of the gill (Fig 2, 12-115B). They are easy to see if the gill has been removed. Each is ventral to the digestive ceca and adjacent to the efferent branchial artery.
Two nephridia, one right and one left open into the exhalant chamber via nephridiopores atop urinary papillae. A renopericardial canal connects the pericardial cavity with the nephridium but it is difficult to demonstrate.
Reproductive System
Most scallops are hermaphroditic and the two gonads, ovary and testis, are located in the visceral mass anterior to the adductor muscle. They are obscured, or partly obscured, by black pigment in the body wall. The testis is creamy white and occupies the anterior dorsal margin of the visceral mass (Fig 2, 12-115B). The ovary is pink or orange and is adjacent to the adductor muscle and posterior to the testis. Gametes are released into the exhalant chamber through the nephridium and nephridiopore. Fertilization is external.
(Placopecten is gonochoric and has a large, fat, crescent-shaped gonad, either ovary or testis, on the anterior face of the adductor muscle (Fig 6). The ovary is coral pink and the testis is cream or white.)
Nervous System
" Study the nervous system under magnification (Fig 6). Cut off the left labial palps. Admire the lips again and find the mouth and esophagus.
The cerebropleural ganglia lie just beneath the integument ventral to the esophagus at the base of the anterior edge of the foot (Fig 6, 12-119). Carefully remove the integument from this region. It may also be necessary to pick away a little of the gonad and digestive ceca. Work with the animal immersed in liquid and under magnification. It will help visibility to flush the working area with fluid from a pipet occasionally to remove bits or clouds of material.
Each elongate cerebropleural ganglion is composed of the fused cerebral and pleural ganglia and is connected to the other cerebropleural ganglion by a long cerebral commissure that arches dorsally, just under the integument, over the top of the esophagus. The two cerebropleural ganglia are widely separated dorsally but are closer together ventral to the esophagus.
The cerebropleural ganglia are closely associated with the pedal ganglia. The two pedal ganglia are joined to each other by the short pedal commissure (Fig 6, 12-119). The pedal and cerebropleural ganglia on each side are connected by the short cerebropedal connective. These ganglia, commissures, and connectives form the circumesophageal nerve ring around the esophagus.
The cerebrovisceral connective exits the posteroventral corner of each cerebropleural ganglion and extends under the gonad to the anterior edge of the adductor muscle (Fig 6). It curves around this edge, hidden by the base of the kidney, to the visceral ganglion located at the ventral end of the kidney at about 8:00 on the margin of the adductor muscle (viewed from the left side).
" The two visceral ganglia lie close together near the median plane and are connected by a short visceral commissure. Expose the cerebrovisceral connective and visceral ganglion on the left side by carefully following the connective posteriorly. Cut through the gonad with your fine scissors and remove the kidney. The connective is easily seen at the base of the kidney on the border of the adductor muscle.
>1d. If small living bivalves such as Donax or Tellina are available in the laboratory they can be used to demonstrate the layout of the bivalve nervous system. Relax such a clam in magnesium chloride, remove it from its shell, remove the mantle skirts and gills, and place the clam between two slides to compress it. Examine the squeeze preparation with the dissecting microscope.
The ganglia of most such bivalves are orange, yellow, or pink with neuroglobin and can be seen clearly against the otherwise white tissue background. The cerebropleural ganglia are located in the anterior end dorsal to the esophagus. The pedal ganglia are ventral and posterior to the border between the visceral mass and the foot. The visceral ganglia are posterior, near the posterior adductor muscle. <
Behavior and Biology
>1e. If you have a living specimen, place it in a small aquarium of seawater and watch it swim by rapidly and forcefully clamping its valves together. Manipulate the animal so that it comes to rest with the gape of the shell facing you through the glass wall of the aquarium.
Look into the gape between the valves to see the elaborate mantle edge. Part of it forms a curtain, the velar fold, around the gape (Fig 12-115B). The numerous beautiful, iridescent, blue eyes and sensory tentacles of the mantle are easily seen (Fig 12-115A). On which valve does your scallop prefer to rest? The left valve is dark and overgrown with fouling organisms whereas the right is pale and mostly unfouled. <
>1f. Look for other organisms on the upper (left) valve. A tiny, white, pyramidellid snail, Boonea seminuda, is often found in its feeding position along the margin of the gape ofArgopecten. It curves its proboscis around the edge of the shell and sucks blood from the mantle. <
>1g. Touch the mantle margin gently with the arm of a starfish or squirt a drop or two of starfish extract into the gape. (Starfish extract is prepared by grinding the arm of a starfish in a mortar, filtering and recovering the filtrate for use.) Observe and record the response of the scallop. <
References
Dakin ML. 1906. Habits, anatomy, and embryology of the giant scallop (Pecten tenuicostatis, Mighels). Univ. Maine Stud. 6:1-71, 17 pls.
Dakin WJ. 1909. Pecten. Liverpool Marine Biology Committee Mem. 17:1-114, 9 pls.
Gutsell J. 1930. Natural history of the bay scallop. Bull. U. S. Bur. Fish. 46:569-632.
Kellogg JL. 1915. Ciliary mechanism of lamellibranchs with descriptions of anatomy. J. Morph. 26:625-701.
Pierce ME. 1950. Pecten irradians, in F. A. Brown (ed) Selected Invertebrate Types. Wiley, New York. pp 321-324.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Supplies
Dissecting microscope
Compound microscope
Living or preserved scallop
Isotonic magnesium chloride for living specimens
Small dissecting pan
Dissecting set
Chalk dust or carmine/seawater suspension