Invertebrate Anatomy OnLine
Argiope aurantia ©
Garden spider
29may2007
Copyright 2001 by
Richard Fox
Lander University
Preface
This is one of many exercises available from Invertebrate Anatomy OnLine , an Internet laboratory manual for courses in Invertebrate Zoology. Additional exercises can be accessed by clicking on the links to the left. A glossary and chapters on supplies and laboratory techniques are also available through these links. Terminology and phylogeny used in these exercises correspond to usage in the Invertebrate Zoology textbook by Ruppert, Fox, and Barnes (2004). Hyphenated figure callouts refer to figures in the textbook. Callouts that are not hyphenated refer to figures embedded in the exercise. The glossary includes terms from this textbook as well as the laboratory exercises.
Systematics
Arthropoda P, Chelicerata sP, Euchelicerata, Arachnida C, Araneae O, Aranaeomorphae sO, Neocribellitae iO, Araneidae F (Fig 16-15, 18-47)
Arthropoda P
Arthropoda, by far the largest and most diverse animal taxon, includes chelicerates, insects, myriapods, and crustaceans as well as many extinct taxa such as Trilobitomorpha. The segmented body primitively bears a pair of jointed appendages on each segment. The epidermis secretes a complex cuticular exoskeleton which must be molted to permit increase in size. Extant arthropods exhibit regional specialization in the structure and function of segments and appendages but the ancestor probably had similar appendages on all segments. The body is typically divided into a head and trunk, of which the trunk is often further divided into thorax and abdomen.
The gut consists of foregut, midgut, and hindgut and extends the length of the body from anterior mouth to posterior anus. Foregut and hindgut are epidermal invaginations, being derived from the embryonic stomodeum and proctodeum respectively, and are lined by cuticle, as are all epidermal surfaces of arthropods. The midgut is endodermal and is responsible for most enzyme secretion, hydrolysis, and absorption.
The coelom is reduced to small spaces associated with the gonads and kidney. The functional body cavity is a spacious hemocoel divided by a horizontal diaphragm into a dorsal pericardial sinus and a much larger perivisceral sinus. Sometimes there is a small ventral perineural sinus surrounding the ventral nerve cord.
The hemal system includes a dorsal, contractile, tubular, ostiate heart that pumps blood to the hemocoel. Excretory organs vary with taxon and include Malpighian tubules, saccate nephridia, and nephrocytes. Respiratory organs also vary with taxon and include many types of gills, book lungs, and tracheae.
The nervous system consists of a dorsal, anterior brain of two or three pairs of ganglia, circumenteric connectives, and a paired ventral nerve cord with segmental ganglia and segmental peripheral nerves. Various degrees of condensation and cephalization are found in different taxa.
Development is derived with centrolecithal eggs and superficial cleavage. There is frequently a larva although development is direct in many. Juveniles pass through a series of instars separated by molts until reaching the adult size and reproductive condition. At this time molting and growth may cease or continue, depending on taxon.
Chelicerata sP
Chelicerata is a large taxon that includes spiders, scorpions, pseudoscorpions, ticks, mites, horseshoe crabs, sea spiders, and many others. The group originated in marine habitats but almost all modern chelicerates are terrestrial.
The body is divided into an anterior cephalothorax with six pairs of appendages and a posterior abdomen which, in most groups, does not bear appendages or has highly modified appendages. The first appendages of the cephalothorax are the chelicerae. Antennae are not present and the brain has two regions rather than the three found in mandibulates. Appendages are primitively biramous but are uniramous in almost all Recent taxa.
Euchelicerata
The segments of the cephalothorax are fused and covered by a dorsal shield, or carapace. Two median eyes are present.
Arachnida C
Arachnids are the terrestrial descendents of the early aquatic chelicerates. The taxon includes the mites, scorpions, pseudoscorpions, spiders, harvestmen, and several other taxa. All are adapted for a terrestrial existence with internal gas exchange and a waterproof integument. Some are capable of silk production and many use toxins to subdue or kill the prey. Most are carnivores, digestion is usually outside the body, and food is liquefied before ingestion.
Araneae O
Spiders are arachnids with unsegmented cephalothorax and abdomen (in most). The cephalothorax bears six pairs of jointed segmental appendages whereas the abdomen bears none, although it has structures derived from them. The anteriormost appendages are chelicerae used to inject poison into the prey. Respiration is via book lungs and/or trachea and excretion is via Malpighian tubules and saccate nephridia known as coxal glands. The heart is a dorsal tube in the abdomen. Abdominal silk glands spin silk for many purposes including the construction of a web for prey capture. All spiders are carnivores that capture living prey. Poison is used to subdue the prey, which is then digested externally by enzymes from the gut, usually following thorough external mastication. The nervous system is highly cephalized and consists of a large nerve ring in the middle of the cephalothorax from which nerves arise. Spiders are gonochoric and sexually dimorphic. Fertilization is internal but sperm transfer is indirect and spermatophores are not used. Males are usually smaller than females.
Aranaeomorphae sO,
Araneomorphae is by far the largest spider taxon with 90 families and over 30,000 recent species. The fangs are either diagonal or labidognath, not orthognath. The abdomen is not segmented. Most taxa have one pair of book lungs
Neocribellatae Io
These are the spiders sometimes known as labidognaths in recognition of their labidognath fangs that move at right angles to the long axis of the body. There may be one or no pairs of book lungs. One pair of tracheae is present. Females usually have an epigynum.
Laboratory Specimens
This exercise is written for preserved female garden spiders, Argiope aurantia , but can be used for most other members of the large family Araneidae. The araneids are orb weavers that build the quintessential, flat, circular spider web. Argiope aurantia, the North American garden spider, is a common, large, distinctively colored (black and yellow) species often seen in the autumn in suburban and rural yards, roadsides, and gardens. Females of this species include a conspicuous zig-zag band of white silk in the center of the orb and because of this are known colloquially as "writing" spiders. The male is a small inconspicuous animal but a careful observer can sometimes find one in its diminutive, imperfect web near the female's much larger web.
Argiope is the genus usually supplied by supply companies but identification is not always accurate. Males are not usually included in commercially supplied jars of preserved spiders. Jars of preserved "garden" spiders from commercial suppliers are likely to include other species in addition to, or instead of Argiope aurantia. Argiope trifasciata, the banded garden spider, is similar to A. aurantia and almost indistinguishable from it morphologically. Nephila clavipes, is a large species that builds gigantic webs in forests across the southern portion of the North American continent. It has an elongate, angular abdomen and long legs with distinctive tufts of black setae on some of the articles. Species of Araneus have fat, almost spherical, abdomens. All belong to the family Araneidae and are similar. Excellent photographs of Argiope bruennichi can be found at www.xs4all.nl/~ednieuw/Spiders/spidhome.htm
External anatomy should be studied with the animal moist but in air, whereas the animal should be immersed in water for dissection and study of internal anatomy. A dissecting microscope or hand lens should be employed for the study of external anatomy and a dissecting microscope for internal anatomy. If living specimens are used they may be anesthetized or killed, as desired, in a jar containing a wad of cotton dampened with chloroform or ethyl acetate respectively. Preserved specimens used for external anatomy can be saved and, with care, used for many years.
External Anatomy
Tagmata
Examine a female Argiope, or similar species, using magnification as needed (Fig 1, 2, 18-18). If the specimen is preserved, handle it carefully as it will be brittle.
The body is covered by an exoskeleton, or cuticle. Some regions of the cuticle are hardened, or sclerotized, whereas others are soft and flexible but, in general, the exoskeleton tends to be softer than that of most insects and crustaceans.
Figure 1. Dorsal view of a female garden spider, Argiope aurantia. Spider48L.gif
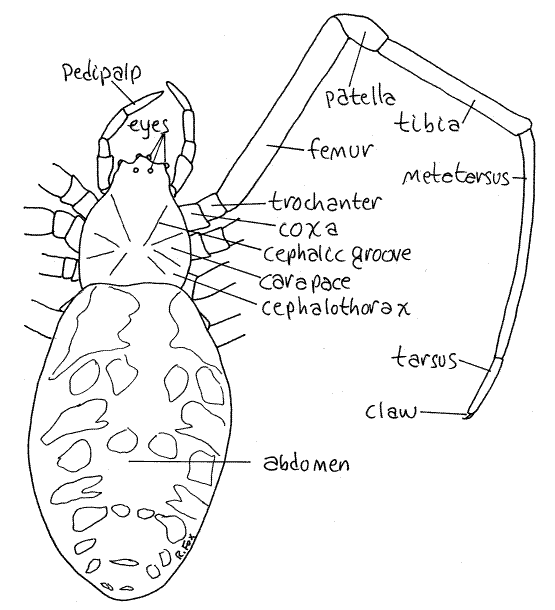
The body is composed of two tagmata; the anterior cephalothorax, or prosoma, and the posterior abdomen, or opisthosoma (Fig 1). The two are connected by a narrow waist, orpedicel (Figs 2, 5). The bulbous ovoid abdomen is much larger than the cephalothorax.
Cephalothorax
The cephalothorax consists of the fused head and thorax of the proarachnid ancestors. It is flattened dorsally and covered by a broad, flat, sclerotized, exoskeletal plate known as thecarapace (Fig 1). A pair of oblique cephalic grooves on the carapace marks the line of fusion of the ancestral head and thorax. The head is anterior to the cephalic grooves and the thoraxposterior to them. The carapace is covered by a pelt of fine setae visible only with magnification.
The anterior end of the cephalothorax bears four pairs of large eyes, some of which are elevated on small tubercles (Fig 3). Each possesses a single large, transparent lens bulging above the carapace surface. The anterior median eyes, or main eyes, are homologous to simple ocelli of other arthropods. They are anteriormost and face forward (Fig 4, 18-27). The remaining three pairs are secondary eyes derived from compound eyes of chelicerate ancestors and include, from anterior to posterior, the anterior lateral eyes, posterior lateral eyes, andposterior median eyes. The eyes point in different directions and are simple, rather than compound.
Figure 2. Ventral view of a female garden spider, Argiope aurantia. Spider49L.gif
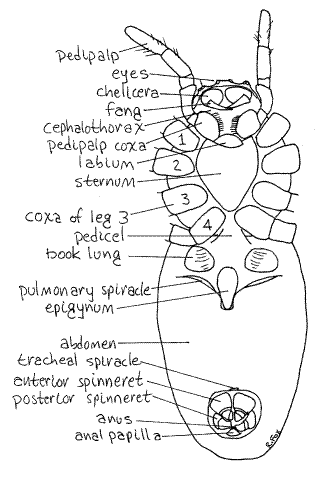
Hold the specimen so you can view the anterior end with the dissecting microscope. The anterior end of the cephalothorax, or face, of the spider is composed of the anterior carapace and the broad basal articles of the chelicerae (Fig 3, 18-27A).
The chelicerae are the first appendages of the cephalothorax and are composed of two articles. The proximal article is the large basal piece attached to the anterior end of the cephalothorax under the edge of the carapace. It contains powerful muscles and a duct from a poison gland in the cephalothorax.
The second, or distal, article of the chelicera is a slender, sharp, hollow fang at whose tip the poison duct opens. Spider venom is a mixture of neurotoxins and proteolytic enzymes. Almost all spiders have poison glands but few are harmful to humans.
The second cephalothoracic appendages are the pedipalps (Figs 3, 1, 2). These long, slender, leglike appendages are composed of six articles. The enlarged proximal article, orcoxa, attaches to the ventral side of the anterior thorax immediately posterior to the fangs (Fig 2). The median process of the coxa is a setose plate known as the pedipalpal endite, or gnathobase, that masticates food and mixes it with digestive enzymes (Fig 2, 18-18B). Distally the pedipalps are sensory whereas proximally they are used to manipulate food. Those of males are modified to transfer sperm to the female.
The remaining cephalothoracic appendages are four pairs of similar walking legs (Figs 1, 2). Each is composed of a linear series of seven articles named, in order from proximal to distal; coxa, trochanter, femur, patella, tibia, metatarsus, and tarsus (Fig 1). The distal tip of each tarsus bears tiny claws.
Figure 3. En face view of a female garden spider, Argiope aurantia. Spider50L.gif
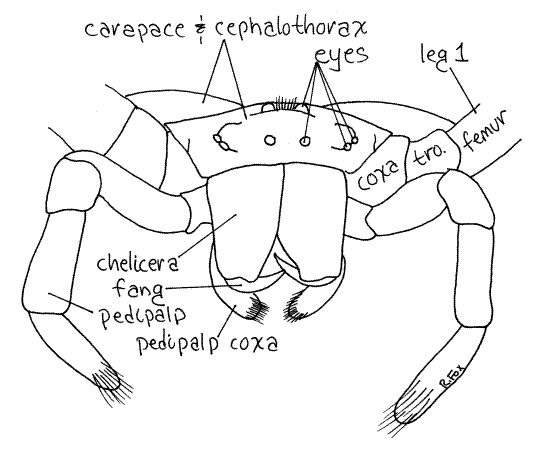
The legs have abundant setae, most or all of which are sensory receptors, chiefly mechanoreceptors sensitive to vibrations, touch, and air movements. Slit sense organs sensitive to strain in the cuticle are present at various places on the body, especially on the legs. Trichobothria, which are special, long, slender setae, are sensitive to very weak air movements. Chemoreceptors are present on the tarsi, especially those of the first legs.
Most of the ventral surface of the cephalothorax is protected by a large, heavily sclerotized sternum formed by the fusion of four embryonic sternites (Fig 2, 18-18B). Anterior to the sternum is a small triangular labium which extends anteriorly between the bases of the pedipalpal coxae. The labium is movable along its articulation with the sternum. It is the sternite of the pedipalpal segment.
The chelicerae, pedipalpal coxae, and labium surround the mouth and enclose the preoral cavity that precedes the mouth. Within the preoral cavity is a prominent conical labrumlocated on the midline. Push the chelicerae and pedipalpal coxae aside to reveal the labrum. The mouth is a triangular opening on the posterior surface of the labrum and is covered and closed by the labium. It can be seen by moving the labium posteriorly on its hinge. The heavy fringes of setae on the pedipalpal coxae and labium filter the slurry resulting from external predigestion before it enters the mouth. Large particles are excluded and only liquid and very small particles enter the mouth.
Figure 4. Enlarged en face view showing the four pairs of eyes. Spider137L.gif
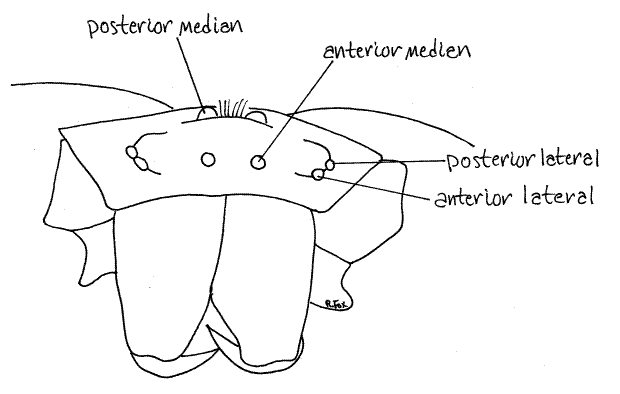
Laterally the dorsal carapace and ventral sternum are joined by the pleural region (pleuron) of the exoskeleton. The pleuron is the soft, flexible and unsclerotized area surrounding the coxae. Little of it is visible because most of it is covered by the coxae of the walking legs.
Abdomen
The abdomen of Argiope is connected to the cephalothorax by a short slender pedicel (Figs 2, 5, 18-20), which is visible ventrally. Because the cephalothorax overhangs the anterior abdomen the pedicel is not visible from above. The pedicel originates on the cephalothorax between the coxae of the fourth pair of walking legs and attaches to the anterior end of the abdomen (Fig 2). Dorsally, the abdomen is featureless except for its striking yellow and black pigmentation (in life).
A pair of large, pale areas of wrinkled exoskeleton on the anterior ventral abdomen marks the position of the internal book lungs (Fig 2). The lungs are invaginations of the exoskeleton elaborated to form numerous tiny lamellae that resemble the pages of a book (Fig 18-6).
The external openings of the book lungs are oblique slits, known as lung spiracles (= pulmonary spiracles), just posterior to the lungs themselves. Insert a microneedle into one of these spiracles to demonstrate its presence.
" If instructed to do so, use a pair of fine forceps to peel the exoskeleton off the surface of the lungs to reveal the lamellae. The lamellae expand the surface area for gas exchange.
The female epigynum is a large exoskeletal protuberance lying on the midline between the two lung spiracles (Figs 2, 5). It covers the female gonopores (Fig 5, 18-29).
Near the posterior end of the abdomen, but still on its ventral surface, there is a large circular depression containing several structures. A third spiracle, this one the median unpairedtracheal spiracle, is the opening into the tracheal system. It lies on the anterior border of this depression. The tracheae are derived from a second pair of book lungs which were present in the ancestral spiders.
Also within the depression are three pairs of spinnerets that spin silk (Figs 2, 5, 18-21). The anterior spinnerets and posterior spinnerets are large and easily seen (with magnification) but the median spinnerets are small and hidden by the other two. They can be seen by pushing the anterior and posterior pairs aside and looking between their bases. The spinnerets are derived from abdominal appendages of the ancestor. The spinnerets are connected by ducts to silk glands in the abdomen (Fig 5, 18-20).
The anus is located subterminally on the posterior surface of the anal papilla. The papilla is on the midline at the posterior edge of the depression (Fig 2).
Internal Anatomy
Preserved spiders are inferior to fresh specimens for the study of internal anatomy but a superficial examination of most of the major organ systems can be accomplished fairly easily, even with preserve material.
" Open the animal by cutting it in half along the median sagittal plane. Use a sawing motion by a sharp scalpel or razor blade for this. Make every effort to keep the blade on the median sagittal plane so you produce right and left mirror images. You will find the cut surfaces of both sides to be useful for study. Pin them side by side under water in a small (anchovy can) wax-bottom dissecting pan so the cut surfaces face up and are easily examined with the dissecting microscope. The appearances of structures viewed in these sections will vary, sometimes markedly, depending on the position of your section. Do not expect your sections to look exactly like Figure 5 or like each other, or like your neighbor's.
Structures in the abdomen may be more difficult to identify that those of the cephalothorax. There is very little hemocoelic space in the abdomen and most of it is packed with digestive ceca or silk glands.
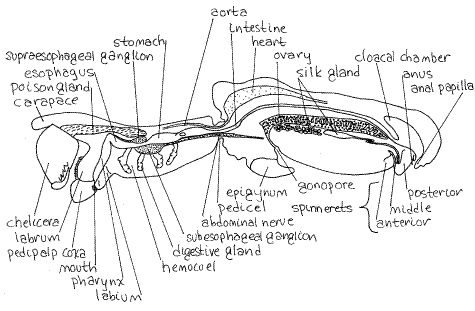
Cephalothorax
Look at the cephalothorax and relocate the external structures in the vicinity of the mouth. You will now have new views of these "mouthparts". Find the mouth on the posterior surface of the labrum and note, once again that it is preceded by a preoral cavity bordered by the chelicerae, pedipalpal coxae, and labium. It is closed posteriorly by the labium.
There is an elongate, hollow poison gland on either side of the midline in the anterior, dorsal cephalothorax (Fig 5, 18-20). The two poison glands are separated by a vertical, partition-like sheet of muscle (the pharynx dilator or levator) lying on the midline. You will probably be able to see the poison gland on one side but not the other (unless you remove this muscle). The anterior end of the poison gland extends into the base of the chelicera and empties by a duct leading to the tip of the fang. The poison glands are muscular and the poison is ejected by contractions of the muscles.
Digestive System
The pharynx is a long, narrow, almost vertical tube extending dorsally from the mouth (Fig 5, 18-20). The pharynx dilator muscle extends from the pharynx to the midline of the carapace. Contractions of this muscle dilate the pharynx and draw liquid food in through the mouth from the preoral cavity.
A slender esophagus extends posteriorly from the dorsal end of the pharynx (Fig 5). In about the middle of the cephalothorax it widens to form the stomach. The pharynx, esophagus, and stomach are all part of the foregut and as such are derived from ectoderm and lined with exoskeleton.
The stomach dilator muscle extends from the dorsal wall of the stomach to the midline of the carapace. Its action is similar to that of the pharynx dilator. Together the stomach and pharynx suck food into the gut.
A narrow intestine, or midgut, exits the posterior end of the stomach and passes through the pedicel to the abdomen. The midgut is endodermal and has no cuticular lining. A one-way valve prevents the reverse flow of food out of the midgut when the stomach dilates.
Tubular anterior digestive ceca arise from each side of the anterior end of the intestine in the cephalothorax. These twisting, cylindrical glands ramify through the interior of the cephalothorax but lie out of the plane of section and may be difficult to discern.
In the abdomen the intestine passes dorsal to the ovary to reach the anus at the posterior end. The large, conspicuous cloacal chamber (or stercoral pocket) opens into the posterior end of the midgut. The gut posterior to the cloacal chamber is the hindgut, or rectum. The hindgut is ectodermal, like the foregut, and lined with cuticle. Posterior digestive ceca open from the midgut and fill much of the space in the abdomen. Most of the posterior digestive cecum is dorsal to the ovary.
Nervous System
The large nerve ring is the easily recognized white mass in the center of the cephalothorax (Fig 5). It consists of a brain, or supraesophageal ganglion, lying on the dorsal surface of the posterior esophagus and a much larger subesophageal ganglion ventral to the esophagus. The two are connected by paired circumesophageal connectives which lie out of the plane of section.
The segmental ganglia, which in primitive arthropods are arranged along a ventral nerve cord, in spiders have moved anteriorly and are concentrated in the subesophageal ganglion as part of the nerve ring. There is no nerve cord and the many peripheral nerves arise from the nerve ring to serve the body. The optic nerve to the eyes enters the brain anteriorly and a largeabdominal nerve exits the posterior end of the subesophageal ganglion and runs along the ventral edge of the intestine, through the pedicel, to the abdomen (Fig 5).
Hemal System
The hemal system and excretory systems in the cephalothorax are difficult to demonstrate in preserved material. The space surrounding the viscera is the hemocoel.
The heart is a large curved tube in the anterior dorsal region of the abdomen (Fig 5, 18-20). Anteriorly it narrows to form the anterior aorta passing through the pedicel to the cephalothorax. Posteriorly it becomes the posterior aorta opening into the posterior abdomen. Its walls bear ostia and it is supported by alary ligaments.
An aorta extends from the heart in the abdomen to supply the hemocoel with blood but it is usually not evident.
Excretory System
The single pair of small coxal glands are in the cephalothorax but you probably will not see them. The chief excretory organs are a pair of very long, narrow Malpighian tubules in the abdomen but they are usually not demonstrable in these dissections or preserved animals. They open into the cloaca and are midgut (endodermal) derivatives.
Reproductive System
The most conspicuous feature of a sagittal section of the abdomen is usually one of the two ovaries (Fig 5, 18-20). The ovary is a long white organ in the middle of the abdomen. Its walls bear distinctive vertical folds and eggs are clearly visible within it. It connects with the female gonopore at the base of the epigynum via an oviduct (Fig 18-29).
Silk Glands
A wide variety of different kinds of silk glands can be seen ventral to the ovary. Each connects to spigots on the spinnerets via ducts (Fig 18-20). Silk glands are not muscular and is pulled, or drawn, out of the spigots as the abdomen is moved away from an attachment point. Silk is composed of the protein fibroin and is comparable to nylon in strength. It is liquid when extruded but solidifies when placed under tension by the “drawing out” process. An orb weaver, such as Argiope, has many different kinds of silk, each secreted by a different kind of gland. Six types of silk glands are known in spiders.
Natural History
The familiar orb web of the araneid spiders is a complicated construction consisting of five main regions. If you have access to an orb web, use the following description to study its anatomy (Fig 18-24, 18-33C).
1. The central hub consists of an irregular network of threads near the center of the web. The spider spends most of her time in the hub. 2. A set of radial threads radiates outward from the hub to the frame. 3. A frame of single threads attached to vegetation, or other features of the environment, extends around the periphery. The radial threads attach to it. The frame is the part most distant from the hub and the two are connected by the radial threads. 4. The catching spiral is a sticky thread that spirals outward from just outside the hub to the periphery of the web. It crosses the radial threads repeatedly at close intervals. It creates the meshwork of closely spaced threads in which flying insects are caught. The catching spiral is the only part of the web that is sticky. Look closely at the sticky spiral, with a hand lens if possible. The tiny droplets of sticky fluid are easily seen. Gently touch one of the sticky threads and note its interaction with your finger. Try the same with the other parts of the web and note the difference. The spider never touches this thread (after spinning it). She uses the radial threads when she wishes to move across the web. 5. Between the hub and the catching spiral there is afree zone consisting solely of radial threads. There is no sticky spiral here so the spider need not worry about catching herself. In this zone the spider can pass safely through the plane of the web from one side to the other.
Argiope aurantia and a few other species include a distinctive, zig-zag band of white silk threads in the hub and free zone and for this reason is called the "writing spider". This structure is the stabilimentum which has been shown to serve as a conspicuous marker to prevent birds from flying through the web and destroying it. Stabilimenta are found only in the webs of those relatively few spiders, of which, Argiope is one, that leave the web up during the day. Most spiders eat the web before daybreak and spin a new one the following night and thus are not endangered by flying birds. When threatened Argiope displays a characteristic alarm response which in which she rocks the web back and forth, presumably to make capture by a predator more difficult.
The female Argiope prepares a large brown, silken cocoon of eggs in the fall and usually dies shortly thereafter although she may live long enough to produce a second cocoon. The eggs hatch in winter but remain in the cocoon until spring. Juveniles emerge and grow during the summer and are adults by fall. The life span is one year.
Laboratory Behavior
Living Argiope can be maintained in large glass aquaria or other enclosures in the laboratory where some aspects of their behavior including web construction, prey capture and handling can be observed.
References
Brown FA . (ed) 1950. Selected Invertebrate Types. Wiley, New York. 597p.
Buck JB, Keister ML. 1950. Argiope aurantia, pp 382-394 in Brown FA (ed.). Selected invertebrate types. Wiley, New York. 597pp.
Eisner T. 2003. For love of insects. Belknap Press of Harvard University, Cambridge. 448pp.
Emerton JH. 1902. The Common Spiders of the United States. Ginn & Co. 227 pp. (Dover reprint).
Felgenhauer BE. 1999. Araneae, pp 223-266 in Harrison FW, Foelix RF (eds), Microscopic anatomy of invertebrates, vol 8A, Chelicerate Arthropoda. Wiley-Liss, New York, 266pp.
Foelix RF. 1996. Biology of Spiders, 2 nd ed. Harvard University Press, Cambridge. 330pp.
Hickman FM, Hickman CP. 1986 . Laboratory Studies in Integrated Zoology. Times Mosby/Mirror, St. Louis. 414 p.
Savory TH. 1928. The Biology of Spiders. Sidgwick & Jackson, London. 376p.
Savory TH. 1977. Arachnida 2 nd ed. Academic Press, New York. 340p.
Ruppert EE, Fox RS, Barnes RB. 2004. Invertebrate Zoology, A functional evolutionary approach, 7 th ed. Brooks Cole Thomson, Belmont CA. 963 pp.
Vanuytven H. Arachnology Home Page : www.arachnology.be/Arachnology.html
Supplies
Dissecting microscope
Small dissecting pan (anchovy or sardine can)
Preserved (or living) Argiope
Dissecting set with microdissecting tools
# 1 stainless steel insect pins